Abstract
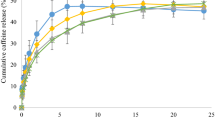
Ginoid hydrolipodystrophy (HDLG) or “cellulite” involves alteration of the cutaneous relief and occurs in 80-90% of the female population. Several topical treatments are available with the use of substances capable of stimulating lipolysis, such as caffeine. However, the effectiveness of topical therapy is related to the processes of release and permeation of the active in skin cells. In this sense, ionic liquids, such as choline geranate, are considered to facilitate topical permeation agents. In this way, the aim of this research was to develop and evaluation of the effectiveness of a cosmetic product for topical treatment of cellulite with caffeine in association with choline geranate. The choline geranate was synthesized by the reaction between geranic acid and choline hydroxide [1: 2]. The gel was prepared using 2% Carpobol 940®, 5% caffeine, and 1% choline geranate. Preliminary and accelerated stability tests were performed by checking pH, spreadability, and organoleptic characteristics. The transdermal permeation capacity of caffeine in vitro was evaluated by the Franz cell permeation assay, and the gel cytotoxicity by the MTS method. To prove the efficacy in the treatment of cellulite, a pilot type 1 clinical trial was carried out. The formulation was considered stable and the product maintained your characteristics during 180 days of storage. The product showed moderate cytotoxicity and high skin permeation capacity. In the clinical trial, it showed results superior to the caffeine gel without ionic liquid. The developed gel favored the cutaneous permeation of caffeine, showing a promising product in the treatment of cellulite.






Similar content being viewed by others
Change history
28 April 2021
A Correction to this paper has been published: https://doi.org/10.1208/s12249-021-02015-9
References
Cunha MG, Cunha ALG, Machado CA. Physiophatology of gynoid lipodystrophy. Surg Cosmet Dermatology. 2015;7:98–103.
Costa A, Alves CRT, Pereira ESP, Cruz FAM, Fidelis MC, Frigerio RM, et al. Gynoid lipodystrophy and clinical therapy: a critical analysis of scientific papers. Surg Cosmet Dermatology. 2012;4:64–75.
Friedman DP, Vick GL, Mishra M. Cellulite: a review with a focus on subcision. Clin Cosmet Investig Dermatol. 2017;10:17–23.
Freire TB, Dario MF, Mendes OG, Oliveira AC, Vetore Neto A, Faria DLA, et al. Nanoemulsion containing caffeine for cellulite treatment: characterization and in vitro evaluation. Brazilian J Pharm Sci. 2019;55:1–11.
Altamoros FMP, Pérez DA, Sigall DA, Romay AAA, Gastelum JAB, Salcedo JAP, et al. Evidence-based treatment for gynoid lipodystrophy: a review of the recent literature. J Cosmet Dermatol. 2018;17:977–83.
Sadik N. Treatment for celulite. Int J Woman Dermatology. 2019;5:68–72.
Visconti MJ, Haidari W, Feldman SR. Therapeutic use of caffeine in dermatology: a literature review. J Dermatology Dermatologic Surg. 2020;24:1–7.
Ruela ALM, Perissinato AG, Lino MES, Mudrik PS, Pereira GR. Evaluation of skin absorption of drugs from topical and transdermal formulations. Brazilian J Pharm Sci. 2016;52:527–44.
Carvalho FC, Bruschi ML, Evangelista RC, Gremião MPD. Mucoadhesive drug delivery systems. Brazilian J Pharm Sci. 2010;46:1–17.
Zakrewsky M, Lovejoy KS, Kern TL, Miller TE, Le V, Nagy A, et al. Ionic liquids as a class of materials for transdermal delivery and pathogen neutralization. Proc Natl Acad Sci. 2014;111:13313–8.
Jorge LR, Harada LK, Silva EC, Campos WF, Moreli FC, Shimamoto G, et al. Non-invasive transdermal delivery of human insulin using ionic liquids: in vitro studies. Front Pharmacol [Internet]. 2020;11:1–17 Available from: https://www.frontiersin.org/article/10.3389/fphar.2020.00243/full.
Campos WF, Silva EC, Oliveira TJ, Oliveira JM Jr, Tubino M, Pereira C, et al. Transdermal permeation of bacteriophage particles by choline oleate: potential for treatment of soft-tissue infections. Future Microbiol. 2020;15:881–96.
Monti D, Egiziano E, Burgalassi S, Chetoni P, Chiappe C, Sanzone A, et al. Ionic liquids as potential enhancers for transdermal drug delivery. Int J Pharm [Internet]. Elsevier B.V.; 2017;516:45–51. Available from: https://doi.org/10.1016/j.ijpharm.2016.11.020, 2017
Zakrewsky M, Banerjee A, Apte S, Kern TL, Jones MR, Sesto RE, et al. Choline and geranate deep eutectic solvent as a broad-spectrum antiseptic agent for preventive and therapeutic applications. Adv Healthc Matherials. 2016;5:1282–9.
Chaugule A, Patil H, Pagariya S, Ingle P. Extraction of caffeine. Int J Adv Res Chem Sci. 2019;6:11–9.
Veryser L, Boonen J, Mehuys E, Roche N, Remon J, Peremans K, et al. Transdermal evaluation of caffeine in different formulations and excipientes. J Caffeine Res. 2013;3:1–6.
Bhawani SA, Fong SS, Ibrahim MNM. Spectrophotometric analysis of caffeine. Int J Anal Chem. 2015;2015:170239.
Pires VGA, Moura MR. Preparation of new nanocomposites containing nanoemulsions of melaleuca, copaíba and lemon oil for application as biomaterial. Quim Nova. 2017;40:1–5.
Bhagyashree P, Karishma G, Sampada A, Ankita P, Pratibha C, Kailash V. Recent trends in stability testing of pharmaceutical products: a review. Res J Pharm Biol Chem Sci. 2015;6:1557–69.
Dantas MGB, Reis SAGB, Damasceno CMD, Rolim LA, Rolim Neto PJ, Carvalho FO, et al. Development and evaluation of stability of a gel formulation containing the monoterpene borneol. Sci World J. 2016:1–4.
Ministry of Health B. Ministry of Health, BRAZIL, RDC No 318, 6 NOVEMBER 2019 [Internet]. 2019 [cited 2021 Feb 4]. Available from: http://abracro.org.br/pdfs/RDC-318-06Nov2019-estudos-estabilidade.pdf
Salerno C, Carlucci AM, Bregni C. Study of in vitro drug release and percutaneous absorption of fluconazole from topical dosage forms. AAPS PharmSciTech. 2010;11:986–93.
Giannelli M, Chellini F, Margheri M, Toneli P, Tani A. Effect of chlorhexidine digluconate on different cell types: a molecular and ultrastructural investigation. Toxicol Vitr. 2008;22:308–17.
Ulrich W. A celulite é curável. Rio de Janeiro: Tecnoprint; 1982.
R Core T. A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2020.
Harada LK, Pereira JFB, Campos WF, Silva EC, Moutinho CG, Vila MMDC, et al. Insights into protein-ionic liquid interactions aiming at macromolecule delivery systems. J Braz Chem Soc. 2018;29:1983–98.
Vizak VP, Calada MS, Vuksanovic JM, Ivanis GR, Brasnco ASH, Grozdani ND, et al. Solutions of ionic liquids with diverse aliphatic and aromatic solutes – phase behavior and potentials for applications: A review article. Arab J Chem. 2019;12:1628–40.
Djajadisastra J, Hadyanti S. Percutane transport profile of caffeine and aminophyllin as anticellulite and the influences of other substances on in vitro penetration. Int J Pharm Pharm Sci. 2014;6:532–8.
Amnuaikit T, Ingkatawornwong S, Maneenuan D, Worachotekamjorn K. Caffeine gel formulation. Indian J Pharm Sci. 2008;4:16–24.
Islam R, Chowdhury R, Wakabayashi R, Kamiya N, Moniruzzaman M, Goto M. Ionic liquid-in-oil microemulsions prepared with biocompatible choline carboxylic acids for improving the transdermal delivery of a sparingly soluble drug. Pharmaceutics. 2020;12:1–18.
Saeedi M, Rafati MR, Morteza-Semnani K, Rostam AY, Kelidari HR. Evaluation of effect of tween 80 on characteristics of tadalafil 0.1% suspension. Pharm Biomed Res. 2015;1:35–43.
Prieto C, Calvo L. Performance of the biocompatible surfactant tween 80, for the formation of microemulsions suitable for newpharmaceutical processing. J Appl Chem. 2013:1–10.
Wagemaker TAL, Rijo P, Rodrigues LM, Maia Campos PMBG, Feranades AS, Rosado C. Integrated approach in the assessment of skin compatibility of cosmetic formulations with green coffee oil. Int J Cosmet Sci. 2015;37:505–10.
Sowmya KV, Ravishankar K, Basha DP, Kiranmayi GVN. Estimation of caffeine and sodium benzoate in caffeine and sodium benzoate injection by isoabsorption method (isobestic method). Int J Pharm Chem Biol Sci. 2011;1:26–31.
Herman A, Herman AP. Caffeine’s mechanisms of action and its cosmetic use. Skin Pharmacol Physiol. 2013;26:8–14.
Banerjee A, Ibsen K, Iwao Y, Zakrewsky M, Mitragotri S. Transdermal protein delivery using choline and geranate (CAGE) deep eutectic solvent. Adv Healthc Mater. 2017;6:1–11.
Luebberding S, Krueger N, Sadick NS. Cellulite: an evidence-based review. Am J Clin Dermatol. 2015;16:243–56.
Nikolis A, Enright KM. Methods of standardizing photography for cellulite in the buttocks and thighs. Dermatologic Surg. 2018;4:1208–10.
Funding
This work was funded by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, São Paulo, Brazil) (FAPESP Refs. No. 2016/08884-3 (Project PneumoPhageColor) and 2016/12234-4 (Project TransAppIL)). Funding for Victor M. Balcão through a BPE grant from FAPESP (São Paulo, Brazil) (Ref. No. 2018/05522-9, Project PsaPhageKill) is hereby gratefully acknowledged. This work also received support from CNPq, National Council for Scientific and Technological Development Brazil, in the form of Research Productivity (PQ) fellowships granted to Victor M. Balcão (Refs. No. 306113/2014-7 and 308208/2017-0).
Author information
Authors and Affiliations
Contributions
All authors participated in the conception and design of the experiments and analyzed the resulting data; A.N.H and R.B. performed the experiments; V.M.B. and M.M.D.C.V. wrote the paper. M.M.D.C.V. supervised the work and contributed with reagents and analysis tools. V.M.B. performed the image analysis and revised the paper. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: An English translation error occurred with the word usage of “cellulitis” in the title and throughout the article. The correct usage is “cellulite”.
Rights and permissions
About this article
Cite this article
Hernandes, A.N., Boscariol, R., Balcão, V.M. et al. Transdermal Permeation of Caffeine Aided by Ionic Liquids: Potential for Enhanced Treatment of Cellulite. AAPS PharmSciTech 22, 121 (2021). https://doi.org/10.1208/s12249-021-01956-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-021-01956-5