Abstract
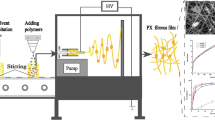
The antihypertensive drug felodipine (FD) is a typical biopharmaceutics classification system (BCS) II drug; thus, improving the dissolution rate of FD is very important to enhance its bioavailability. Besides, according to the in situ “close loop” perfusion assay, we found that the jejunum is the main absorptive site, then the duodenum and ileum. Consequently, a novel micron-size particulate of FD in a core-shell structure was fabricated by a coaxial electrospray technique; within the drug delivery system, Hypromellose K4M (HPMC K4M) was selected as a sheath material to prolong the retention time in the upper GI tract, while povidone K30 (PVP K30) was mixed with FD in the inner layer. The dissolution study in three different media (0.02% Tween-80 solution; phosphate buffer containing 0.02% Tween-80, pH 6.8; and HCl solution containing 0.02% Tween-80, pH 1.2) demonstrated that FD-loaded coaxial electrospray particles (F-COES) could greatly improve the dissolution of FD. Furthermore, in vivo pharmacokinetics revealed that F-COES emerged no changes in the half-life but significantly prolonged the tmax and increased the oral bioavailability. Collectively, this work supplies a promising drug release system that will improve the dissolution and enhance the bioavailability simultaneously for those poorly water-soluble drugs mainly absorbed in the upper GI tract.






Similar content being viewed by others

References
Elvelin L, Jönsson L. The effect of dihydropyridine calcium antagonists on heart rate: studies of felodipine. Curr Ther Res. 1994;55(7):736–46.
Saltiel E, Ellrodt AG, Monk JP, Langley MS. Felodipine. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic use in hypertension. Drugs. 1988;36(4):387–428.
Kumar D, Thipparaboina R, Shastri NR. Can vacuum morphologies predict solubility and intrinsic dissolution rate. A case study with felodipine polymorph form IV. J Sports Sci. 2015;10:178–85.
Caiqin Y, Wei G, Yulong L, et al. Experimental and DFT simulation study of a novel felodipine cocrystal: characterization, dissolving properties and thermal decomposition kinetics. J Pharm Biomed Anal. 2018;154:198–206.
Kim EJ, Chun MK, Jang JS, et al. Preparation of a solid dispersion of felodipine using a solvent wetting method. Eur J Pharm Biopharm. 2006;64(2):200–5.
Chen Y, Huang W, Chen J, et al. The synergetic effects of non-polar and polar protic solvents on the properties of felodipine and soluplus in solutions, casting films and spray dried solid dispersions. J Pharm Sci. 2018:S0022354918300881.
Wu C, Zhao Z, Zhao Y, Hao Y, Liu Y, Liu C. Preparation of a push-pull osmotic pump of felodipine solubilized by mesoporous silica nanoparticles with a core-shell structure. Int J Pharm. 2014;475(1–2):298–305.
Gonçalves A, Nikmaram N, Roohinejad S, et al. Production, properties, and applications of solid self-emulsifying delivery systems (S-SEDS) in the food and pharmaceutical industries. Colloids Surf A Physicochem Eng Asp. 2017:S0927775717309767.
Shah U, Joshi G, Sawant K. Improvement in antihypertensive and antianginal effects of felodipine by enhanced absorption from PLGA nanoparticles optimized by factorial design. Mater Sci Eng C. 2014;35(1):153–63.
Pi C, Feng T, Liang J, Liu H, Huang D, Zhan C, et al. Polymer blends used to develop felodipine-loaded hollow microspheres for improved oral bioavailability. Int J Biol Macromol. 2018;112:1038–47.
Palem CR, Dudhipala N, Battu SK, Goda S, Repka MA, Yamsani MR. Combined dosage form of pioglitazone and felodipine as mucoadhesive pellets via hot melt extrusion for improved buccal delivery with application of quality by design approach. J Drug Deliv Sci Technol. 2015;30:209–19.
Sarode AL, Malekar SA, Cote C, Worthen DR. Hydroxypropyl cellulose stabilizes amorphous solid dispersions of the poorly water soluble drug felodipine. Carbohydr Polym. 2014;112(21):512–9.
Leuner C, Dressman J. Improving drug solubility for oral delivery using solid dispersions. Eur J Pharm Biopharm. 2000;50:47–60.
Moneghini M, Carcano A, Zingone G, Perissutti B. Studies in dissolution enhancement of atenolol. Part I. Int J Pharm. 1998;175:177–83.
Chakraborty S, Liao I, Adler A, et al. Electrohydrodynamics: a facile technique to fabricate drug delivery systems. Adv Drug Deliv Rev. 2009;61(12):1043–54.
Zhang S, Kawakami K, Yamamoto M, Masaoka Y, Kataoka M, Yamashita S, et al. Coaxial electrospray formulations for improving oral absorption of a poorly water-soluble drug. Mol Pharm. 2011;8(3):807–13.
Kawakami K, Zhang S, Chauhan RS, Ishizuka N, Yamamoto M, Masaoka Y, et al. Preparation of fenofibrate solid dispersion using electrospray deposition and improvement in oral absorption by instantaneous post-heating of the formulation. Int J Pharm. 2013;450(1–2):123–8.
Wang M, Wang Y, Omari-Siaw E, Wang S, Zhu Y, Xu X. Reduced burst release and enhanced Oral bioavailability in shikimic acid-loaded polylactic acid submicron particles by coaxial electrospray. J Pharm Sci. 2016;105(8):2427–36.
Jiao Y, Pang X, Liu M, et al. Recent progresses in bioadhesive microspheres via transmucosal administration. Colloids Surf B. 2015;140:361.
Liu K, Zhou X, Xu Z, Bai H, Zhu J, Gu J, et al. Anhydrates and hemihydrate of tasimelteon: synthesis, structure, and pharmacokinetic study. J Pharm Biomed Anal. 2018;151:235–43.
Zhang J, Liu D, Huang Y, Gao Y, Qian S. Biopharmaceutics classification and intestinal absorption study of apigenin. Int J Pharm. 2012;436(1–2):311–7.
Gustafsson AB, Gottlieb RA, Granville DJ. TAT-mediated protein transduction: delivering biologically active proteins to the heart. Methods Mol Med. 2005;112(112):81.
Chen Y, Yuan L, Zhou L, et al. Effect of cell-penetrating peptide-coated nanostructured lipid carriers on the oral absorption of tripterine. Int J Nanomedicine. 2012;7(4):4581–91.
Jian Mao Y, Liu-sheng Z, Deng-Guang Y, Jianyun L. Coaxial electrospinning with acetic acid for preparing ferulic acid/zein composite fibers with improved drug release profiles. Colloids Surf B. 2013;102:737–43.
Sultanova Z, Kaleli G, Kabay G, Mutlu M. Controlled release of a hydrophilic drug from coaxially electrosprayed polycaprolactone nanofibers. Int J Pharm. 2016;505(1–2):133–8.
Rui M, Xin Y, Ran L, et al. Targeted biomimetic nanoparticles for synergistic combination chemotherapy of paclitaxel and doxorubicin. Mol Pharm. 2016;14(1):107.
Li H, Dong WF, Zhou JY, Xu XM, Li FQ. Triggering effect of N-acetylglucosamine on retarded drug release from a lectin-anchored chitosan nanoparticles-in-microparticles system. Int J Pharm. 2013;449(1–2):37–43.
Chen J, Cui Y, Xin X, et al. Direct and effective preparation of core-shell PCL/PEG nanoparticles based on shell insertion strategy by using coaxial electrospray. Colloids Surf A Physicochem Eng Asp. 2018;547:1–7.
Cao X, Deng W, Fu M, Zhu Y, Liu H, Wang L, et al. Seventy-two-hour release formulation of the poorly soluble drug silybin based on porous silica nanoparticles: in vitro release kinetics and in vitro/in vivo correlations in beagle dogs. Eur J Pharm Sci. 2013;48(1–2):64–71.
Zhu Y, Zhang J, Fu M, et al. Calcium carbonate nanoparticles templated by mixed polymeric micelles: characterization, in vitro drug release and oral bioavailability in beagle dogs(J). Lat Am J Pharm. 2014;33(33):1106–13.
Wang G, Wang JJ, Chen XL, du L, Li F. Quercetin-loaded freeze-dried nanomicelles: improving absorption and anti-glioma efficiency in vitro and in vivo. J Control Release. 2016;235:276–90.
Zhen OY, Cao X, Wei Y, et al. Pharmacokinetic study of rutin and quercetin in rats after oral administration of total flavones of mulberry leaf extract. Rev Bras Farmacogn. 2013;23(5):776–82.
Zhu Y, Wang M, Zhang J, Peng W, Firempong CK, Deng W, et al. Improved oral bioavailability of capsaicin via liposomal nanoformulation: preparation, in vitro drug release and pharmacokinetics in rats. Arch Pharm Res. 2015;38(4):512–21.
Feng Y, Sun C, Yuan Y, Zhu Y, Wan J, Firempong CK, et al. Enhanced oral bioavailability and in vivo antioxidant activity of chlorogenic acid via liposomal formulation. Int J Pharm. 2016;501(1–2):342–9.
Nguyen DN, Clasen C, Mooter GVD. Pharmaceutical applications of electrospraying. J Pharm Sci. 2016;105(9):2601–20.
Shah VP, Konecny JJ, Everett RL, McCullough B, Noorizadeh AC, Skelly JP. In vitro dissolution profile of water-insoluble drug dosage forms in the presence of surfactants. Pharm Res. 1989;6(7):612–8.
Pezzini BR, Beringhs AO, Ferraz HG, Silva MAS, Stulzer HK, Sonaglio D. Liquisolid technology applied to pellets I: evaluation of the feasibility and dissolution performance using felodipine as a model drug. Chem Eng Res Des. 2016;110:62–9.
Parhizkar M, Reardon PJT, Knowles JC, Browning RJ, Stride E, Pedley RB, et al. Performance of novel high throughput multi electrospray systems for forming of polymeric micro/nanoparticles. Mater Des. 2017;126:73–84.
Deng-Guang Y, Jiao-Jiao L, Williams GR, et al. Electrospun amorphous solid dispersions of poorly water-soluble drugs: a review. J Control Release. 2018:S0168365918304826.
Herpin MJ, Smyth HDC. Super-heated aqueous particle engineering (SHAPE): a novel method for the micronization of poorly water soluble drugs. J Pharm Invest. 2018;48(1):135–42.
Edgar B, Hoffmann KJ, Lundborg P, RegÅrdh CG, Rönn O, Weidolf L. Absorption, distribution and elimination of felodipine in man. Drugs. 1985;29(2):9–15.
Palugan L, Cerea M, Zema L, Gazzaniga A, Maroni A. Coated pellets for oral colon delivery. J Drug Deliv Sci Technol. 2015;25:1–15.
Strugala G, Elsenhans BW. Active transport inhibition in rat small intestine by amphiphilic amines: an in vitro study with various local anaesthetics. Biochem Pharmacol. 2000;59(8):907–13.
Suvannasara P, Juntapram K, Praphairaksit N, Siralertmukul K, Muangsin N. Mucoadhesive 4-carboxybenzenesulfonamide-chitosan with antibacterial properties. Carbohydr Polym. 2013;94(1):244–52.
Satish P, Talele GS. Gastroretentive mucoadhesive tablet of lafutidine for controlled release and enhanced bioavailability. Drug Deliv. 2015;22(3):312–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yin, X., Pan, H. & Liu, H. A Novel Micron-Size Particulate Formulation of Felodipine with Improved Release and Enhanced Oral Bioavailability Fabricated by Coaxial Electrospray. AAPS PharmSciTech 20, 282 (2019). https://doi.org/10.1208/s12249-019-1495-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-019-1495-8