Abstract
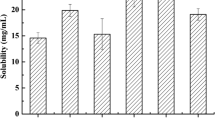
To improve the absorption of poorly water-soluble 20(S)-protopanaxadiol (20(S)-PPD), novel 20(S)-PPD-loaded redispersible dry suspension and dry emulsion were developed in this study. 20(S)-PPD dry suspension (PPD-DS) was prepared by enabling drug fully dispersed with suspending agent Avicel CL611 and solubilizer Poloxamer 188. 20(S)-PPD dry emulsion (PPD-DE) was prepared by employing oleic acid as oil phase, Cremophor RH-40 as surfactant, and n-butyl alcohol as co-surfactant. Both PPD-DS and PPD-DE were evaluated for their physicochemical characterization after being dispersed in distilled water. The in vivo pharmacokinetics was evaluated by UPLC-MS/MS. The droplet size of PPD-DS and PPD-DE was in the scope of 1446–1653 nm and 652.8–784.5 nm. The sedimentation volume ratios of PPD-DS and PPD-DE were both at value of 1. The zeta potential of PPD-DS and PPD-DE were from − 53.7 to − 70.4 mV and − 27.5 to − 34.5 mV, respectively, which indicated stable systems. PPD-DS and PPD-DE both achieved dramatically enhanced aqueous solubility and higher perfusion of 20(S)-PPD in rats’ intestine. Although statistically, no oral bioavailability enhancements of 20(S)-PPD were achieved in PPD-DE and PPD-DS, there were some improvements in the pharmacokinetic behaviors. Especially, PPD-DS could be a promising drug delivery carrier for 20(S)-PPD with the advantages of long-term stability, dosing flexibility, and the convenience of administering to infants and to those who have difficulty swallowing tablets or capsules.







Similar content being viewed by others
References
Wang BX, Cui JC, Liu AJ, Wu SK. Studies on the anti-fatigue effect of the saponins of stems and leaves of Panax ginseng (SSLG). J Tradit Chin Med. 1983;2:89–94.
Surh YJ, Lee JY, Chun KS, Na HK. Anti-tumor promoting effects of selected ginsenosides and their underlying molecular mechanisms. J Ginseng Res. 2002;10:343–50.
Popovich DG, Kitts DD. Structure–function relationship exists for ginsenosides in reducing cell proliferation and inducing apoptosis in the human leukemia (THP-1) cell line. Arch Biochem Biophys. 2002;406:1–8.
Usami Y, Liu YN, Lin AS, Shibano M, Akiyama T, Itokawa H. Antitumor agents. 261. 20(S)-protopanaxadiol and 20(S)-protopanaxatriol as antiangiogenic agents and total assignment of 1H NMR spectra. J Nat Prod. 2008;71:478–81.
Wang W, Zhao YQ, Rayburn ER, Hill DL, Wang H, Zhang RW. In vitro anti- cancer activity and structure–activity relationships of natural products isolated from fruits of Panax ginseng. Cancer Chemother Pharmacol. 2007;59:589–601.
Ben-Eltriki M, Deb S, Adomat H, Guns ES. Calcitriol and 20(S)-protopanaxadiol synergistically inhibit growth and induce apoptosis in human prostate cancer cells. J Steroid Biochem. 2015;158:207–19.
Xu C, Teng J, Chen W, Ge Q, Yang Z, Yu C, et al. 20(S)- protopanaxadiol, an active ginseng metabolite, exhibits strong antidepressantlike effects in animal tests. Prog Neuro-Psychopharmacol Biol Psychiatry. 2010;34:1402–11.
Chen GT, Yang X, Li JL, Ge HJ, Song Y, Ren J. Biotransformation of 20(S)-protopanaxadiol by Aspergillus Niger AS 3.1858. Fitoterapia. 2013;91:256–60.
Xie FF, Li SW, Cheng ZN, Liu XL, Zhang H, Li PJ, et al. Determination of 20(s)-protopanaxadiol in human plasma by HPLC-MS/MS: application to a pharmacokinetic study. Acta Pharm Sin B. 2013;3(6):385–91.
Chen C, Wang LS, Cao FR, Miao XQ, Chen TK, Chang Q, et al. Formulation of 20(s)-protopanaxadiol nanocrystals to improve oral bioavailability and brain delivery. Int J Pharm. 2016;497:239–47.
Chiu NTC, Guns EST, Adomat H, Jia W, Deb S. Identification of human cytochrome P450 enzymes involved in the hepatic and intestinal biotransformation of 20(S)-protopanaxadiol. Biopharm Drug Dispos. 2013;35:104–18. https://doi.org/10.1002/bdd.1873.
Zhang Q, Pu YQ, Wang B, Wang YQ, Dong T, Guo T, et al. Characterization, molecular docking, and In vitro dissolution studies of solid dispersions of 20(S)-protopanaxadiol. Molecules. 2017;22:274.
Kawabata Y, Wada K, Nakatani M, Yamada S, Onoue S. Formulation design for poorly water-soluble drugs based on biopharmaceutics classification system: basic approaches and practical applications. Int J Pharm. 2011;420:1–10.
Wang B, Pu YQ, Xu BL, Tao JS, Wang YQ, Zhang T, et al. Self-microemulsifying drug delivery system improved oral bioavailability of 20(S)-protopanxadiol: from preparation to evaluation. Chem Pharm Bull. 2015;63:688–93.
Zhu KX, Chen YX, Zhang T, Tao JS, Wang B. Study on preparation and release in vitro of 20(S)-protopanaxadiol complex microspheres. J Math Med. 2015;28:813–5 (in Chinese).
Wang B, Pu YQ, Tao JS, Xu BL, Zhang T. Determination and in vitro evaluation of 20(S)-PPD tablets. Chin J Exp Tradit Med Formulae. 2011;17:89–94 (in Chinese).
Pu YQ, Zhang XT, Zhang Q, Wang B, Chen YX, Zang CQ, et al. 20(S)-Protopanaxadiol phospholipid complex: process optimization, characterization, in vitro dissolution and molecular docking studies. Molecules. 2016;21:1396.
Wang B, Zang CQ, Chen YX, Tao JS, Zhang T. Preparation of 20(S)-protopanaxadiol-phospholipid complex HAP assemblies. J Chin Med Mater. 2014;37:2282–5 (in Chinese).
Raj SP, Angela LS. Stability of marketed albendazole suspensions and correlation between zeta potential and sedimentation. Int Res J Pharm. 2011;2(3):154–6.
Arshady R. Suspension, emulsion, and dispersion polymerization: a methodological survey. Colloid Polym Sci. 1992;170:717–32.
Patil SK, Wagh KS, Parik VB, Akarte AM, Baviskar DT. Strategies for solubility enhancement of poorly soluble drugs. Int J Pharm Sci Rev Res. 2011;8(2):74–80.
Patel HM, Patel BB, Shah CN. Nanosuspension: a novel approach to enhance solubility of poorly water soluble drugs – a review. Int J Adv Pharm. 2016;5(2):21–9.
Naeini YS, Aminzare M, Golestani-Fard F, Khorasanizadeh F, Salahi E. Suspensions stability of Titania nanoparticles studied by UV-VIS spectroscopy method. Iran J Mater Sci. 2012;9(1):62–8.
Tharwat T. Polymeric surfactants in disperse systems. Adv Colloid Interf Sci. 2009;147–148:281–99.
Wang L, Sun YH, Kuang C, Zhang XR. Preparation and evaluation of taste masked oral suspension of arbidol hydrochloride. A J Pharm Sci. 2015;10:73–9.
Goibier L, Lecomte S, Leal-Calderon F, Faure C. The effect of surfactant crystallization on partial coalescence in O/W emulsions. J Colloid Interface Sci. 2017;500:304–14.
Pongsamart K, Kleinebudde P, Puttipipatkhachorn S. Preparation of fenofibrate dry emulsion and dry suspension using octenyl succinic anhydride starch as emulsifying agent and solid carrier. Int J Pharm. 2016;498:347–54.
Hauss DJ, Fogal SE, Ficorilli JV, Price CA, Roy T, Jayaraj AA, et al. Lipid-based delivery systems for improving the bioavailability and lymphatic transport of a poorly water-soluble LTB4 inhibitor. J Pharm Sci. 1998;87(2):164–9.
Cui F, Wang YS, Wang JM, Feng LL, Ning KJ. Preparation of redispersible dry emulsion using Eudragit E 100 as both solid carrier and unique emulsifier. Colloids Surf A Physicochem Eng Asp. 2007;307:137–41.
Pohlen M, Pirker L, Lustrik M, Dreu R. A redispersible dry emulsion system with simvastatin prepared via fluid bed layering as a means of dissolution enhancement of a lipophilic drug. Int J Pharm. 2018;549:325–34.
Zhang JJ, Gao Y, Qian S, Liu X, Zu H. Physicochemical and pharmacokinetic characterization of a spray-dried malotilate emulsion. Int J Pharm. 2011;414:186–92.
Mehanna MM, Alwattar JK, Elmaradny HA. Optimization, physicochemical characterization and in vivo assessment of spray dried emulsion: a step toward bioavailability augmentation and gastric toxicity minimization. Int J Pharm. 2015;496:766–79.
Mao JJ, Zhang T, Wang B, Tao JS. Prescription of 20(S)-protopanaxadiol for suspension. Chin Tradit Pat Med. 2012;34(9):1680–4 (in Chinese).
Du YQ, Zhai YL, Zhang JH, Wu CN, Luo C, Sun J, et al. Development and evaluation of taste-masked dry suspension of cefuroxime axetil for enhancement of oral bioavailability. Asian J Pharm Sci. 2013;8:287–94.
Rao M, Aghav S. Spray-dried redispersible emulsion to improve oral bioavailability of Itraconazole. J Surfactant Deterg. 2014;17:807–17.
Yin YM, Cui FD, Kim JS, Choi MK, Choi BC, Chung SJ, et al. Preparation, characterization and in vitro intestinal absorption of a dry emulsion formulation containing atorvastatin calcium. Drug Deliv. 2009;16(1):30–6.
Moghimipour E, Salimi A, Leis F. Preparation and evaluation of tretinoin microemulsion based on pseudo-ternary phase diagram. Adv Pharm Bull. 2012;2(2):141–7.
Garg A, Bhalala K, Tomar DS, Wahajuddin. In-situ single pass intestinal perfusion and pharmacokinetic study of developed Lumefantrine loaded solid lipid nanoparticles. Int J Pharm. 2017;516:120–30.
Niczinger NA, Kallai-Szabo B, Lengyel M, Gordon P, Klebovich I, Antal I. Physicochemical analysis in the evaluation of reconstituted dry emulsion tablets. J Pharm Biomed Anal. 2017;134:86–93.
Kommuru TR, Gurley B, Khan MA, Reddy IK. Self-emulsifying drug delivery systems (SEDDS) of coenzyme Q10: formulation development and bioavailability assessment. Int J Pharm. 2001;212:233–46.
Lnaggar YS, El-Massik MA, Abdallah OY. Self-nanoemulsifying drug delivery systems of tamoxifen citrate:design and optimization. Int J Pharm. 2009;380:133–41.
Balakumar K, Raghavan CV, Selvan NT, Prasad RH, Abdu S. Self nanoemulsifying drug delivery system (SNEDDS) of rosuvastatin calcium: design, formulation, bioavailability and pharmacokinetic evaluation. Colloids Surf B: Biointerfaces. 2013;112:337–43.
Xi J, Chang Q, Chan CK, Meng ZY, Wang GN, Sun JB, et al. Formulation development and bioavailability evaluation of a self nano-emulsified drug delivery system of oleanolic acid. AAPS PharmSciTech. 2009;10:172–82.
Nor S, Woi P, Ng S. Characterisation of ionic liquids nanoemulsion loaded with piroxicam for drug delivery system. J Mol Liq. 2017;234:30–9.
Shakeel F, Haqa N, Alanazi FK, Alsarra IA. Polymeric solid self-nanoemulsifying drug delivery system of glibenclamide using coffee husk as a low cost biosorbent. Powder Technol. 2014;256:352–60.
Badran MM, Taha EI, Tayel MM, Al-Suwayeh SA. Ultra-fine self- nanoemulsifying drug delivery system for transdermal delivery of meloxicam: dependency on the type of surfactants. J Mol Liq. 2014;190:16–22.
Agrawal AG, Kumar A, Gide PS. Formulation of solid self-nanoemulsifying drug delivery systems using N-methyl pyrrolidone as cosolvent. Drug Dev Ind Pharm. 2015;41:594–604.
Bergstrom CAS, Holm R, Jorgensen SA, Andersson SBE, Artursson P, Beato S. Early pharmaceutical profiling to predict oral drug absorption: current status and unmet needs. Eur J Pharm Sci. 2014;57:173–99.
Ogaji IJ, Hoag SW. Effect of grewia gum as a suspending agent on ibuprofen pediatric formulation. AAPS PharmSciTech. 2011;12(2):507–13.
Fedele L, Colla L, Bobbo S. Viscosity and thermal conductivity measurements of water-based nanofluids containing titanium oxide nanoparticles. Int J Refrig. 2012;35:1359–66.
Xia HJ, Zhang ZH, Jin X, Hu Q, Chen XY, Jia XB. A novel drug-phospholipid complex enriched with micelles: preparation and evaluation in vitro and in vivo. Int J Nanomedicine. 2013;8:545–54.
Han MH, Chen J, Chen SL, Wang XT. Development of a UPLC-ESI-MS/MS assay for 20(S)-Protopanaxadiol and pharmacokinetic application of its two formulations in rats. Anal Sci. 2010;26:749–53.
Kumar RS, Yagnesh NS. Pharmaceutical suspensions: patient compliance oral dosage forms. World J Pharm Sci. 2016;5(12):1471–537.
Beg S, Swain S, Singh HP, Patra Ch N, Rao ME. Development, optimization, and characterization of solid self-nanoemulsifying drug delivery systems of valsartan using porous carriers. AAPS PharmSciTech. 2012;13:1416–27.
Christensen KL, Pedersen GP, Kristensen HG. Preparation of redispersible dry emulsions by spray drying. Int J Pharm. 2001;212:187–94.
Yang Y, Fan CM, He X, Ren K, Zhang JK, He YJ, et al. Study on biopharmaceutics classification and oral bioavailability of a novel multikinase inhibitor NCE for cancer therapy. Int J Mol Sci. 2014;15:7199–212.
Ge Z, Zhang XX, Gan L, Gan Y. Redispersible, dry emulsion of lovastatin protects against intestinal metabolism and improves bioavailability. Acta Pharmacol Sin. 2008;29(8):990–7.
Barve PJR, KH. Intestinal perfusion of lamivudine using single pass intestinal perfusion. Indian J Pharm Sci. 2012;74(5):478–81.
Ali W, Williams AC, Rawlinson CF. Stochiometrically governed molecular interactions in drug: poloxamer solid dispersions. Int J Pharm. 2010;391(31):162–8.
Pu XH, Sun J, Li M, He ZG. Formulation of nanosuspensions as a new approach for the delivery of poorly soluble drugs. Curr Nanosci. 2009;5:417–27.
Keservani RK, Kesharwani R, Gujarathi NA, Sharma AK. Formulation and characterization of Piroxicam reconstituted suspension. IJPI J Pharm Cosmet. 2011;3(1):69–76.
Marina G, Elena M, Carlotti ME, Michele T, Philippe P. Formulation of dry emulsion for topical applications. J Dispers Sci Technol. 2009;30(6):823–33.
Amidon GE, Higuchi WI, Ho NFH. Theoretical and experi- mental studies of transport of micelle-solubilized solutes. J Pharm Sci. 1982;71(1):77–84.
Miller JM, Beig A, Krieg BJ, Carr RA, Borchardt TB, Amidon GE, et al. The solubility–permeability interplay: mechanistic modeling and predictive application of the impact of micellar solubilization on intestinal permeation. Mol Pharm. 2011;8(5):1848–56.
Poelma FGJ, Breäs R, Tukker JJ. Intestinal absorption of drugs. III. The influence of taurocholate on the disappearance kinetics of hydrophilic and lipophilic drugs from the small intestine of the rat. Pharm Res. 1990;7(4):392–7.
Poelma FGJ, Breäs R, Tukker JJ, Crommelin DJA. Intestinal absorption of drugs. The influence of mixed micelles on the disappearance kinetics of drugs from the small intestine of the rat. J Pharm Pharmacol. 1991;43(5):317–24.
Yano K, Masaoka Y, Kataoka M, Sakuma S, Yamashita S. Mechanisms of membrane transport of poorly soluble drugs: role of micelles in oral absorption processes. J Pharm Sci. 2010;99(3):1336–45.
Miller JM, Beig A, Carr RA, Spence JK, Dahan A. A win-win solution in oral delivery of lipophilic drugs: supersaturation via amorphous solid dispersions increases apparent solubility without sacrifice of intestinal membrane permeability. Mol Pharm. 2012;9:2009–16.
Dahan A, Beig A, Ioffe-Dahan V, Agbaria R, Miller J. The twofold advantage of the amorphous form as an oral drug delivery practice for lipophilic compounds: increased apparent solubility and drug flux through the intestinal membrane. AAPS J. 2013;15:347–53.
Acknowledgments
This work was supported by grants from Program of Shanghai Committee of Science and Technology (Grant No. 18401931400), Program of Shanghai Academic/Technology Research Leader (Grant No. 18XD1403700), National Scientific and Technological Major Special Project of China (Grant No. 2018ZX09201008-002), and Scientific Research Program of Shanghai University of Traditional Chinese Medicine (Grant No. 18TS087).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(PDF 141 kb)
Rights and permissions
About this article
Cite this article
Zhang, L., Xu, C., Mao, J. et al. Formulation and Characterization of Novel Dry Suspension and Dry Emulsion of 20(S)-Protopanaxadiol. AAPS PharmSciTech 20, 275 (2019). https://doi.org/10.1208/s12249-019-1487-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-019-1487-8