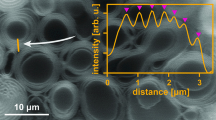
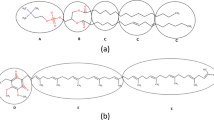
Abstract
Elastic liposoxy1mes (ELs) are biocompatible bilayer vesicular systems commonly used in the transdermal delivery of drugs. Compared with conventional liposomes (CLs), the strong deformation ability conferred by edge activators (EAs) is one of the most critical properties of ELs. However, due to limited research methods, little is known about the effect of EAs on the deformation abilities of vesicles. In this study, taking sodium cholate as an example, a multiscale study was carried to study the effect of EAs on the deformability of ELs, including in vitro diffusion experiment at macroscale, “vesicle-pore” model experiment at the microscale and flat patch model experiment at the molecular scale. As a result, it was found that sodium cholate could decrease the kc of DPPC bilayer, which enabled it to remain morphologically intact during a strong deformation process. Such kind of differences on deformation ability made pogostone ELs (contain sodium cholate) present a better permeation effect compared with that of pogostone CLs. All of these provide a multiscale and thorough understanding of the effect of sodium cholate on the deformation ability of ELs.







Similar content being viewed by others
References
Benson HA. Elastic liposomes for topical and transdermal drug delivery. Curr Drug Delivery. 2009;6(3):217–26.
Hussain A, Singh S, Sharma D, Webster TJ, Shafaat K, Faruk A. Elastic liposomes as novel carriers: recent advances in drug delivery. Int J Nanomedicine. 2017;12:5087–08.
Duangjit S, Opanasopit P, Rojanarata T, Ngawhirunpat T. Evaluation of meloxicam-loaded cationic transfersomes as transdermal drug delivery carriers. AAPS PharmSciTech. 2013;14(1):133–40.
Garg V, Singh H, Bhatia A, Raza K, Singh SK, Singh B, et al. Systematic development of transethosomal gel system of piroxicam: formulation optimization, in vitro evaluation, and ex vivo assessment. AAPS PharmSciTech. 2017;18(1):58–71.
Utreja P, Jain S, Tiwary AK. Evaluation of biosafety and intracellular uptake of Cremophor EL free paclitaxel elastic liposomal formulation. Drug Deliv. 2012;19(1):11–20.
Ahad A, Al-Saleh AA, Al-Mohizea AM, Al-Jenoobi FI, Raish M, Yassin AEB, et al. Formulation and characterization of novel soft nanovesicles for enhanced transdermal delivery of eprosartan mesylate. Saudi Pharm J. 2017;25(7):1040–6.
Ahad A, Aqil M, Kohli K, Sultana Y, Mujeeb M, Ali A. Formulation and optimization of nanotransfersomes using experimental design technique for accentuated transdermal delivery of valsartan. Nanomedicine. 2012;8(2):237–49.
Cevc G, Blume G. Lipid vesicles penetrate into intact skin owing to the transdermal osmotic gradients and hydration force. Biochim Biophys Acta Biomembr. 1992;1104(1):226–32.
Cevc G, Gebauer D, Stieber J, Schätzlein A, Blume G. Ultraflexible vesicles, transfersomes, have an extremely low pore penetration resistance and transport therapeutic amounts of insulin across the intact mammalian skin. Biochim Biophys Acta Biomembr. 1998;1368(2):201–15.
Chen J, Lu WL, Gu W, Lu SS, Chen ZP, Cai BC. Skin permeation behavior of elastic liposomes: role of formulation ingredients. Expert Opin Drug Deliv. 2013;10(6):845–56.
Benson HA. Transfersomes for transdermal drug delivery. Expert Opin Drug Deliv. 2006;3(6):727–37.
Al Shuwaili AH, Rasool BK, Abdulrasool AA. Optimization of elastic transfersomes formulations for transdermal delivery of pentoxifylline. Eur J Pharm Biopharm. 2016;102:101–14.
Subongkot T, Pamornpathomkul B, Rojanarata T, Opanasopit P, Ngawhirunpat T. Investigation of the mechanism of enhanced skin penetration by ultradeformable liposomes. Int J Nanomedicine. 2014;9:3539–50.
Zhang YT, Xu YM, Zhang SJ, Zhao JH, Wang Z, Xu DQ, et al. In vivo microdialysis for the evaluation of transfersomes as a novel transdermal delivery vehicle for cinnamic acid. Drug Dev Ind Pharm. 2014;40(3):301–7.
Ntimenou V, Fahr A, Antimisiaris SG. Elastic vesicles for transdermal drug delivery of hydrophilic drugs: a comparison of important physicochemical characteristics of different vesicle types. J Biomed Nanotechnol. 2012;8(4):613–23.
Alvi IA, Madan J, Kaushik D, Sardana S, Pandey RS, Ali A. Comparative study of transfersomes, liposomes, and niosomes for topical delivery of 5-fluorouracil to skin cancer cells: preparation, characterization, in-vitro release, and cytotoxicity analysis. Anti-Cancer Drugs. 2011;22(8):774–82.
Duangjit S, Obata Y, Sano H, Onuki Y, Opanasopit P, Ngawhirunpat T, et al. Comparative study of novel ultradeformable liposomes: menthosomes, transfersomes and liposomes for enhancing skin permeation of meloxicam. Biol Pharm Bull. 2014;37(2):239–47.
El Zaafarany GM, Awad GA, Holayel SM, Mortada ND. Role of edge activators and surface charge in developing ultradeformable vesicles with enhanced skin delivery. Int J Pharm. 2010;397(1–2):164–72.
Jain SK, Gupta Y, Jain A, Rai K. Enhanced transdermal delivery of acyclovir sodium via elastic liposomes. Drug Deliv. 2008;15(3):141–7.
Uchino T, Lefeber F, Gooris G, Bouwstra J. Characterization and skin permeation of ketoprofen-loaded vesicular systems. Eur J Pharm Biopharm. 2014;86(2):156–66.
Gonzalez-Rodriguez ML, Arroyo CM, Cozar-Bernal MJ, Gonzalez RP, Leon JM, Calle M, et al. Deformability properties of timolol-loaded transfersomes based on the extrusion mechanism. Statistical optimization of the process. Drug Dev Ind Pharm. 2016;42(10):1683–94.
Marrink SJ, Corradi V, Souza PCT, Ingolfsson HI, Tieleman DP, Sansom MSP. Computational modeling of realistic cell membranes. Chem Rev. 2019;119:6184–226.
Hossain MS, Berg S, Bergstrom CAS, Larsson P. Aggregation behavior of medium chain fatty acids studied by coarse-grained molecular dynamics simulation. AAPS PharmSciTech. 2019;20(2):61.
Watson MC, Morriss-Andrews A, Welch PM, Brown FL. Thermal fluctuations in shape, thickness, and molecular orientation in lipid bilayers. II. Finite surface tensions. J Chem Phys. 2013;139(8):084706.
Watson MC, Brandt EG, Welch PM, Brown FL. Determining biomembrane bending rigidities from simulations of modest size. Phys Rev Lett. 2012;109(2):028102.
Levine ZA, Venable RM, Watson MC, Lerner MG, Shea JE, Pastor RW, et al. Determination of biomembrane bending moduli in fully atomistic simulations. J Am Chem Soc. 2014;136(39):13582–5.
Leonard AN, Wang E, Monje-Galvan V, Klauda JB. Developing and testing of lipid force fields with applications to modeling cellular membranes. Chem Rev. 2019;119:6227–69.
Brannigan G, Brown FL. A consistent model for thermal fluctuations and protein-induced deformations in lipid bilayers. Biophys J. 2006;90(5):1501–20.
Arnarez C, Uusitalo JJ, Masman MF, Ingolfsson HI, de Jong DH, Melo MN, et al. Dry martini, a coarse-grained force field for lipid membrane simulations with implicit solvent. J Chem Theory Comput. 2015;11(1):260–75.
Marrink SJ, de Vries AH, Mark AE. Coarse grained model for semiquantitative lipid simulations. J Phys Chem B. 2004;108(2):750–60.
Ma L, Wu Z, Yang C, Guo S, Chen L, Qiao Y, et al. Preparation and quaility evaluation of pogostone transfersomes. China Pharmacy. 2019;30(01):50–4.
Chen H, Li Y, Wu X, Li C, Li Q, Qin Z, et al. LC-MS/MS determination of pogostone in rat plasma and its application in pharmacokinetic studies. Biomed Chromatogr. 2013;27(9):1092–9.
Wang X, Zou Y, Wang Y. GC-MS assay for the determination of pogostone in essential oil of herba pogostemis. Chinese Journal of Pharmaceutical Analysis. 2005;30(5):546–9.
Cheng X, Jo S, Lee HS, Klauda JB, Im W. CHARMM-GUI micelle builder for pure/mixed micelle and protein/micelle complex systems. Journal of Chemical Information & Modeling. 2013;53(8):2171–80.
Martínez L, Andrade R, Birgin EG, Martínez JM. PACKMOL: a package for building initial configurations for molecular dynamics simulations. J Comput Chem. 2010;30(13):2157–64.
David VDS, Lindahl E, Hess B, Groenhof G, Mark AE, Berendsen HJC. GROMACS: fast, flexible, and free. J Comput Chem. 2010;26(16):1701–18.
Harmandaris VA, Deserno M. A novel method for measuring the bending rigidity of model lipid membranes by simulating tethers. J Chem Phys. 2006;125(20):204905.
Khelashvili G, Kollmitzer B, Heftberger P, Pabst G, Harries D. Calculating the bending modulus for multicomponent lipid membranes in different thermodynamic phases. J Chem Theory Comput. 2013;9(9):3866–71.
Garcia RS, Bezlyepkina N, Knorr RL, Lipowsky R, Dimova R. Effect of cholesterol on the rigidity of saturated and unsaturated membranes: fluctuation and electrodeformation analysis of giant vesicles. Soft Matter. 2010;6(7):1472–82.
Chen Z, Rand RP. The influence of cholesterol on phospholipid membrane curvature and bending elasticity. Biophys J. 1997;73(1):267–76.
Rawicz W, Olbrich KC, McIntosh T, Needham D, Evans E. Effect of chain length and unsaturation on elasticity of lipid bilayers. Biophys J. 2000;79(1):328–39.
Stephanie TN, Nagle JF. HIV-1 fusion peptide decreases bending energy and promotes curved fusion intermediates. Biophys J. 2007;93(6):2048–55.
Klauda JB, Venable RM, Freites JA, O'Connor JW, Tobias DJ, Mondragon-Ramirez C, et al. Update of the CHARMM all-atom additive force field for lipids: validation on six lipid types. J Phys Chem B. 2010;114(23):7830–43.
Braun AR, Sachs JN, Nagle JF. Comparing simulations of lipid bilayers to scattering data: the GROMOS 43A1-S3 force field. J Phys Chem B. 2013;117(17):5065–72.
Melo MN, Ingólfsson HI, Marrink SJ. Parameters for martini sterols and hopanoids based on a virtual-site description. J Chem Phys. 2015;143(24):243152.
Acknowledgments
The content is solely the responsibility of the authors and does not necessarily represent the official views of the Beijing Natural Science Foundation. All simulations were performed at the National Supercomputer Center in Guangzhou.
Funding
This work was financially supported by the Beijing Natural Science Foundation (7162122).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, Z., Yang, C., Chen, L. et al. A Multiscale Study on the Effect of Sodium Cholate on the Deformation Ability of Elastic Liposomes. AAPS PharmSciTech 20, 311 (2019). https://doi.org/10.1208/s12249-019-1485-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-019-1485-x