Abstract
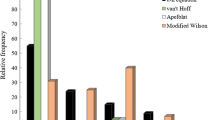
This study aimed to provide a rational experimental design to collect a minimum number of experimental data points for a drug dissolved in a given binary solvent mixture at various temperatures, and to describe a computational procedure to predict the solubility of the drugs in any solvent composition and temperature of interest. We gathered available solubility data sets from papers published from 2012 to 2016 (56 data sets, 3488 data points totally). The mean percentage deviations (MPD) used to check the accuracy of predictions was calculated by Eq. 10. Fifty-six datasets were analyzed using 8 training data points which the overall MPD was calculated to be 15.5% ± 15.1%, and for 52 datasets after excluding 5 outlier sets was 12.1% ± 8.9%. The paired t test was conducted to compare the MPD values obtained from the models trained by 7 and 8 training data points and the reduction in prediction overall MPD (from 17.7% to 15.5%) was statistically significant (p < 0.04). To further reduction in MPD values, the computations were also conducted using 9 training data points, which did not reveal any significant difference comparing to the predictions using 8 training data points (p > 0.88). This observation revealed that the model adequately trained using 8 data points and could be used as a practical strategy for predicting the solubility of drugs in binary solvent mixtures at various temperatures with acceptable prediction error and using minimum experimental efforts. These sorts of predictions are highly in demand in the pharmaceutical industry.


Similar content being viewed by others
References
Williams HD, Trevaskis NL, Charman SA, Shanker RM, Charman WN, Pouton CW, et al. Strategies to address low drug solubility in discovery and development. Pharmacol Rev. 2013;65:315–499.
Jouyban A, Fakhree MAA, Shayanfar A. Solubility prediction methods for drug/drug like molecules. Recent Patents Chem Eng. 2008;1:220–31.
Acree WE Jr. Mathematical representation of thermodynamic properties: Part 2 Derivation of the combined nearly ideal binary solvent (NIBS)/Redlich-Kister mathematical representation from a two-body and three-body interactional mixing model. Thermochim Acta. 1992;198:71–9.
Jouyban A, Acree WE Jr. Mathematical derivation of the Jouyban-Acree model to represent solute solubility data in mixed solvents at various temperatures. J Mol Liq. 2018;256:541–7.
Williams NA, Amidon GL. Excess free energy approach to the estimation of solubility in mixed solvent systems II: ethanol-water mixtures. J Pharm Sci. 1984;73:14–8.
Barzegar-Jalali M, Jouyban A. A general model from theoretical cosolvency models. Int J Pharm. 1997;152:247–50.
Jouyban A. The modified Wilson model and predicting drug solubility in water-cosolvent mixtures. Chem Pharm Bull. 1998;46:1058–61.
Jouyban A, Chan HK, Romero S, Khoubnasabjafari M, Bustamante P. Solubility prediction in water-ethanol mixtures based on the excess free energy approach using a minimum number of experimental data. Pharmazie. 2004;59:117–20.
Barzegar-Jalali M, Hanaee J. Model for solubility estimation in mixed solvent systems. Int J Pharm. 1994;109:291–5.
Barzegar-Jalali M, Jouyban A. Models for calculating solubility in binary solvent systems. Int J Pharm. 1996;140:237–46.
Yalkowsky SH, Roseman TJ. Solubilization of drugs by co-solvents. In: Yalkowsky SH, editor. Techniques of solubilization of drugs. New York: Marcel Dekker Inc; 1981. p. 91–134.
Ruckenstein E, Shulgin I. Solubility of drugs in aqueous solutions - part 2: binary nonideal mixed solvent. Int J Pharm. 2003;260:283–91.
Jouyban A, Valaee L, Barzegar-Jalali M, Clark BJ, Acree WE Jr. Comparison of various cosolvency models for calculating solute solubility in water-cosolvent mixtures. Int J Pharm. 1999;177:93–101.
Jouyban A, Acree WE Jr. In silico prediction of drug solubility in water-ethanol mixtures using Jouyban-Acree model. J Pharm Pharm Sci. 2006;9:262–9.
Jouyban A, Chew NY, Chan HK, Sabour M, Acree WE Jr. A unified cosolvency model for calculating solute solubility in mixed solvents. Chem Pharm Bull. 2005;53:634–7.
Jouyban A, Shayanfar A, Panahi-Azar V, Soleymani J, Yousefi BH, Acree WE Jr, et al. Solubility prediction of drugs in mixed solvents using partial solubility parameters. J Pharm Sci. 2011;100:4368–82.
Li H, Yang W, Hao J, Fan S, Yang S, Guo Q. Experimental measurement and thermodynamic models for solid-liquid equilibrium of 3-amino-1-adamantanol in different pure solvents and in (H2O + ethanol) binary solvent mixtures. J Mol Liq. 2016;215:127–34.
Jiang L, Li S, Jiang J, Qiu J, Wang P. Solid-liquid equilibrium of l-arabinose in water + ethanol solvent system from 283.15 to 308.15 K. J Mol Liq. 2015;211:406–10.
Yang C, Li Q, Wu Y. Thermodynamic models for determination of solid-liquid equilibrium of the 4-methylbenzoic acid in pure and binary organic solvents. J Mol Liq. 2017;238:432–9.
Xu J, Wang Y, Yuan F, Huang Q, Hao H. Solid-liquid equilibrium of l-alanyl-l-glutamine form II in methanol + water and isopropanol + water systems. J Mol Liq. 2016;219:930–6.
Jouyban A, Dastmalchi S, Chan HK, Hanaee J, Javanmard A, Barzegar-Jalali M. Solubility prediction for furosemide in water-cosolvent mixtures using the minimum number of experiments. Drug Dev Ind Pharm. 2001;27:577–83.
Jouyban A, Khoubnasabjafari M, Chan HK, Acree WE Jr. Mathematical representation of solubility of amino acids in binary aqueous-organic solvent mixtures at various temperatures using the Jouyban-Acree model. Pharmazie. 2006;61:789–92.
Jouyban A. Solubility prediction of drugs in water–polyethylene glycol 400 mixtures using Jouyban–Acree model. Chem Pharm Bull. 2006;54:1561–6.
Jouyban A. In silico prediction of drug solubility in water-dioxane mixtures using the Jouyban-Acree model. Pharmazie. 2007;62:46–50.
Jouyban A. Prediction of drug solubility in water-propylene glycol mixtures using Jouyban-Acree model. Pharmazie. 2007;62:365–7.
Jouyban A, Acree WE Jr. Prediction of drug solubility in ethanol-ethyl acetate mixtures at various temperatures using the Jouyban-Acree model. J Drug Del Sci Technol. 2007;17:159–60.
Jouyban A, Fakhree MAA, Acree WE Jr. Comment on “measurement and correlation of solubilities of (z)-2-(2-aminothiazol-4-yl)-2-methoxyiminoacetic acid in different pure solvents and binary mixtures of water + (ethanol, methanol, or glycol)”. J Chem Eng Data. 2012;57:1344–6.
Jouyban A, Martinez F, Acree WE Jr. Further calculations on solubility of 3-amino-1-adamantanol in ethanol+water binary solvent mixtures at various temperatures. J Mol Liq. 2016;219:211–5.
Jouyban A, Acree WE Jr. Calculation of solubility of N-ethylcarbazole in ethanol+petroleum ether mixtures at various temperatures. Korean J Chem Eng. 2016;33:1698–705.
Jouyban A, Martinez F, Acree WE Jr. Correct derivation of a combined version of the Jouyban–Acree and van’t Hoff model and some comments on ‘Determination and correlation of the solubility of myricetin in ethanol and water mixtures from 288.15 to 323.15 K’. Phys Chem Liq. 2017;55:131–40.
Fan JP, Zheng B, Liao DD, Yu JX, Cao YH, Zhang XH, et al. Determination and modeling of the solubility of (limonin in methanol or acetone+water) binary solvent mixtures at T=283.2K to 318.2K. J Chem Thermodyn. 2016;98:353–60.
Zhou Z, Qu Y, Wang J, Wang S, Liu J, Wu M. Measurement and correlation of solubilities of (Z)-2-(2-aminothiazol-4-yl)- 2-methoxyiminoacetic acid in different pure solvents and binary mixtures of water + (ethanol, methanol, or glycol). J Chem Eng Data. 2011;56:1622–8.
Jouyban A, York P, Hanna M, Clark BJ. Solubility prediction of salmeterol xinafoate in water-dioxane mixtures. Int J Pharm. 2001;216:33–41.
Jouyban A, Chew NY, Chan HK, Khoubnasabjafari M, Acree WE Jr. Solubility prediction of salicylic acid in water ethanol propylen glycol mixtures using the Jouyban Acree model. Pharmazie. 2006;4:318–21.
Wang P, Jiang J, Jia X, Jiang L, Li S. Solubility of trehalose in water + ethanol solvent system from (288.15 to 318.15) K. J Chem Eng Data. 2014;59:1872–6.
Yang GD, Li C, Zeng AG, Qu QH, Yang X, Bian XL. Solubility of osthole in a binary system of ethanol and water. Fluid Phase Equilib. 2012;325:41–4.
Ahumada EA, Delgado DR, Martínez F. Solution thermodynamics of acetaminophen in some PEG 400+water mixtures. Fluid Phase Equilib. 2012;332:120–7.
Roy S, Mahali K, Dolui BK. Thermodynamic solvation of a series of homologous α-amino acids in aqueous mixtures of 1,2-dimethoxyethane. J Solut Chem. 2013;42:1472–87.
Mahali K, Roy S, Dolui BK. Solvation thermodynamics of a series of homologous α-amino acids in non-aqueous binary mixtures of protic ethylene glycol and dipolar aprotic acetonitrile. J Solut Chem. 2013;42:1096–110.
Wei D, Li H, Li YN, Zhu J. Effect of temperature on the solubility of 3-aminopyridine in binary ethanol+toluene solvent mixtures. Fluid Phase Equilib. 2012;316:132–4.
Qing XY, Fu HL, Shu G, Liu MJ, Zhou JY, Wu WB, et al. Solubility and solution thermodynamics of diphenoxylate in different pure solvents. J Chem Eng Data. 2015;60:1629–33.
Sun D, Ren R, Dun W, Zhang H, Zhao L, Zhang L, et al. Measurement and correlation of the solubility of l-carnitine in different pure solvents and ethanol-acetone solvent mixture. J Chem Eng Data. 2014;59:1984–90.
Wang G, Wang Y, Hu X, Ma Y, Hao H. Determination and correlation of cefoperazone solubility in different pure solvents and binary mixture. Fluid Phase Equilib. 2014;361:223–8.
Goga ST, Mchedlov-Petrossyan NO, Glazkova EN, Lebed AV. Thermodynamics of solubility and solvation of N-cetylpyridinium perchlorate and related compounds in water-propanol-2 system. J Mol Liq. 2013;177:237–42.
Bhat MA, Haq N, Shakeel F. Solubility of N-(4-chlorophenyl)-2-(pyridin-4-ylcarbonyl)hydrazinecarbothioamide in PEG 400 + water co-solvent mixtures at 298.15K to 338.15K. Thermochim Acta. 2014;589:235–40.
Zhou L, Zhang P, Yang G, Lin R, Wang W, Liu T, et al. Solubility of chrysin in ethanol and water mixtures. J Chem Eng Data. 2014;59:2215–20.
Sun H, Li M, Jia J, Tang F, Duan E. Measurement and correlation of the solubility of 2,6-diaminohexanoic acid hydrochloride in aqueous methanol and aqueous ethanol mixtures. J Chem Eng Data. 2012;57:1463–7.
Shazly GA, Haq N, Shakeel F. Solution thermodynamics and solubilization behavior of diclofenac sodium in binary mixture of Transcutol-HP and water. Pharmazie. 2014;69:335–9.
Kai Y, Hu Y, Cao Z, Liu X, Liu Y, Yang W. Measurement and correlation solubility and mixing properties of l-malic acid in pure and mixed organic solvents. Fluid Phase Equilib. 2013;360:466–71.
Shakeel F, Bhat MA, Haq N, Fathi-Azarbayjani A, Jouyban A. Solubility and thermodynamic parameters of a novel anti-cancer drug (DHP-5) in polyethylene glycol 400+water mixtures. J Mol Liq. 2017;229:241–5.
Shazly GA, Haq N, Shakeel F. Solution thermodynamics and solubility prediction of glibenclamide in Transcutol + water co-solvent mixtures at 298.15-333.15 K. Arch Pharm Res. 2014;37:746–51.
Dun W, Wu S, Tang W, Wang X, Sun D, Du S, et al. Solubility of ibuprofen sodium dihydrate in acetone + water mixtures: experimental measurement and thermodynamic modeling. J Chem Eng Data. 2014;59:3415–21.
Cantillo EA, Delgado DR, Martinez F. Solution thermodynamics of indomethacin in ethanol + propylene glycol mixtures. J Mol Liq. 2013;181:62–7.
Shakeel F, Alanazi FK, Alsarra IA, Haq N. Solubility prediction of indomethacin in PEG 400 + water mixtures at various temperatures. J Mol Liq. 2013;188:28–32.
Yang Y, Zhang Q, Cao C, Cheng L, Shi Y, Yang W, et al. Solubility and solution thermodynamics of 2,5-thiophenedicarboxylic acid in (water + ethanol) binary solvent mixtures. Thermochim Acta. 2014;592:52–7.
Shakeel F, Haq N, Alanazi FK, Alsarra IA. Thermodynamics of solubility of isatin in (PEG 400 + water) mixed solvent systems at T = (298.15 to 338.15) K. J Chem Thermodyn. 2015;82:156–60.
Cheng Y, Huo L, Li X. Solubilities of isophthalic acid in acetic acid + water solvent mixtures. Chinese J Chem Eng. 2013;21:754–8.
Hong M, Xu L, Ren G, Chen J, Qi M. Solubility of lansoprazole in different solvents. Fluid Phase Equilib. 2012;331:18–25.
Hu Y, Wang L, Meng Z, Yang W. Measurement and correlation of the solubility of maleic acid in acetone-ethyl acetate mixtures. Thermochim Acta. 2012;538:75–8.
Mercado KC, Rodriguez GA, Delgado DR, Martinez F, Romdhani A. Solution thermodynamics of methorcarbamol in some ethonol + water mixtures. Quim Nov. 2012;35:1967–72.
Gandhi PJ, Murthy ZVP. Measurement of solubility of mitomycin C in ethanol-water solutions at different temperatures. Thermochim Acta. 2012;545:163–73.
Gomez JL, Rodriguez GA, Cristancho DM, Delgado DR, Martinez F. Solution thermodynamics of nimodipine in some PEG 400 + ethanol mixtures. Phys Chem Liq. 2013;51:651–62.
Li Y, Lü X. Solubilities of nizatidine in methanol + water, ethanol + water and i-propanol + water from 273.15 to 303.15 K. Chinese J Chem Eng. 2012;20:937–41.
Shakeel F, Alanazi FK, Alsarra IA, Haq N. Solubilization behavior of paracetamol in transcutol-water mixtures at (298.15 to 333.15) K. J Chem Eng Data. 2013;58:3551–6.
Soltanpour S, Bastami Z. Thermodynamic solubility of piroxicam in propylene glycol + water mixtures at 298.2-323.2 K - data report and modeling. J Serbian Chem Soc. 2015;80:509–15.
Sotomayor RG, Holguín AR, Romdhani A, Martínez F, Jouyban A. Solution thermodynamics of piroxicam in some ethanol + water mixtures and correlation with the jouyban-acree model. J Solut Chem. 2013;42:358–71.
Shakeel F, Alanazi FK, Alsarra IA, Haq N. Solubility of antipsychotic drug risperidone in Transcutol+water co-solvent mixtures at 298.15 to 333.15K. J Mol Liq. 2014;191:68–72.
Hu Y, Cao Z, Li J, Yang W, Kai Y, Zhi W. Solubilities of 4-Bromo-1,8-naphthalic anhydride in different pure solvents and binary solvent mixtures with the temperature range from (278.15 to 333.15) K. J Chem Eng Data. 2013;58:2913–8.
Wang K, Hu Y, Yang W, Shi Y, Li Y. Solubilities of succinimide in different pure solvents and binary methanol + ethyl acetate solvent mixtures. Thermochim Acta. 2012;538:79–85.
Delgado DR, Martínez F. Solubility and solution thermodynamics of some sulfonamides in 1-propanol + water mixtures. J Solut Chem. 2014;43:836–52.
Delgado DR, Martínez F. Preferential solvation of sulfadiazine, sulfamerazine and sulfamethazine in ethanol + water solvent mixtures according to the IKBI method. J Mol Liq. 2014;193:152–9.
Holguín AR, Delgado DR, Martínez F. Thermodynamic study of the solubility of triclocarban in ethanol + propylene glycol mixtures. Quim Nova. 2012;35:280–5.
Wang Z, Wang Q, Liu X, Fang W, Li Y, Xiao H. Measurement and correlation of solubility of xylitol in binary water+ethanol solvent mixtures between 278.00 K and 323.00K. Korean J Chem Eng. 2013;30:931–6.
Makridakis SG, Wheelwright SC, Hyndman RJ. Forecasting: methods and applications. Wiley series in management. 3rd ed. New York: Wiley; 1998.
Acknowledgements
This report is a part of the results of S. Dadmand’s Pharm D thesis submitted to the Faculty of Pharmacy, Tabriz University of Medical Sciences, Tabriz, Iran.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dadmand, S., Kamari, F., Acree, W.E. et al. Solubility Prediction of Drugs in Binary Solvent Mixtures at Various Temperatures Using a Minimum Number of Experimental Data Points. AAPS PharmSciTech 20, 10 (2019). https://doi.org/10.1208/s12249-018-1244-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-018-1244-4