Abstract
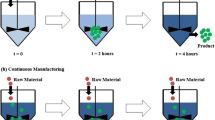
Continuous manufacturing (CM) of pharmaceuticals is a rapidly growing approach in the production of active pharmaceutical ingredients and finished products. The European Medicines Agency, the US Food and Drug Administration, and the Pharmaceuticals and Medical Devices Agency have independently stated their support for the introduction of CM and provided opportunities for early dialog between industries and regulatory agencies. This paper describes the current regulatory landscape and the regulatory harmonization.
Similar content being viewed by others
References
Allison G, Cain YT, Cooney C, Garcia T, Bizjak TG, Holte O, et al. Regulatory and quality considerations for continuous manufacturing May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):803–12.
Byrn S, Futran M, Thomas H, Jayjock E, Maron N, Meyer RF, et al. Achieving continuous manufacturing for final dosage formation: challenges and how to meet them May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):792–802.
Konstantinov KB, Cooney CL. White paper on continuous bioprocessing. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):813–20.
Page T, Dubina H, Fillipi G, Guidat R, Patnaik S, Poechlauer P, et al. Equipment and analytical companies meeting continuous challenges. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):821–31.
Myerson AS, Krumme M, Nasr M, Thomas H, Braatz RD. Control systems engineering in continuous pharmaceutical manufacturing. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 104(3):832–9.
Baxendale IR, Braatz RD, Hodnett BK, Jensen KF, Johnson MD, Sharratt P, et al. Achieving continuous manufacturing: technologies and approaches for synthesis, workup, and isolation of drug substance. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):781–91.
Srai JS, Badman C, Krumme M, Futran M, Johnston C. Future supply chains enabled by continuous processing-opportunities and challenges. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):840–9.
Nepveux K, Sherlock JP, Futran M, Thien M, Krumme M. How development and manufacturing will need to be structured-heads of development/manufacturing. May 20-21, 2014 Continuous Manufacturing Symposium. J Pharm Sci. 2015;104(3):850–64.
Lee SL, O’Connor TF, Yang X, Cruz CN, Chatterjee S, Madurawe RD, et al. Modernizing pharmaceutical manufacturing: from batch to continuous production. J Pharm Innov. 2015;10:191–9.
PMDA Innovative Manufacturing Technology WG. PMDA Views on Applying Continuous Manufacturing to Pharmaceutical Products for Industry (provisional draft). https://www.pmda.go.jp/files/000223712.pdf. Accessed 15 Jul 2018.
Nasr MM, Krumme M, Matsuda Y, Trout BL, Badman C, Mascia S, et al. Regulatory perspectives on continuous pharmaceutical manufacturing: moving from theory to practice: September 26-27, 2016, International Symposium on the Continuous Manufacturing of Pharmaceuticals. J Pharm Sci. 2017;106:3199–206.
Matsuda Y, Aoyama A, Ikematsu Y, Ishii R, Inoue K, Ohta T, et al. Points to Consider Regarding Continuous Manufacturing http://www.nihs.go.jp/drug/section3/AMED_CM_PtC.pdf. Accessed 15 Jul 2018.
Matsuda Y, Aoyama A, Aoyama E, Ikematsu Y, Inoue K, Ohta T, et al. State of Control in Continuous Pharmaceutical Manufacturing http://www.nihs.go.jp/drug/section3/AMED_CM_CONTROLST.pdf. Accessed 15 Jul 2018.
Regulatory Harmonization and Convergence. https://www.fda.gov/BiologicsBloodVaccines/InternationalActivities/ucm271079.htm . Accessed 17 Oct 2018.
Acknowledgements
I thank Sau L. Lee (US FDA) and Dolores Hernan (EMA) for sharing information about ETT and PAT team.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: James DiNunzio, Christine Moore, and Robert Meyer
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The views expressed in this manuscript by the author are personal and do not necessarily reflect the official position of the regulatory authority.
Rights and permissions
About this article
Cite this article
Matsuda, Y. Global Regulatory Landscape. AAPS PharmSciTech 20, 2 (2019). https://doi.org/10.1208/s12249-018-1230-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-018-1230-x