Abstract
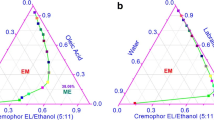
The purpose of this research was to develop microemulsions (ME) and microemulgels (MG) for enhancing transdermal delivery of Kaempferia parviflora (KP) extract. The methoxyflavones were used as markers. Various formulations of ME and MG containing 10% w/v KP extract were prepared, and the in vitro skin permeation and deposition were investigated. The potential ME system containing oleic acid (5% w/v), Tween 20 (20% w/v), PG (40% w/v), and water (35% w/v) was successfully formulated. ME with 10% w/v limonene (ME-L10%) showed higher methoxyflavones flux than ME-L5%, ME-L1%, ME without limonene, and KP extract in water, respectively. ME-L10% was selected for adding a gelling agent to form microemulgels (MG-L10%). However, the high viscosity of the gel formulation might control the diffusion of the compound from gel layer into the skin. Therefore, the liquid formulation provided potential ME droplets to deliver KP extract through the skin. Limonene also plays an effective role on the skin permeation, in which the histological image of the skin treated with ME-L10% exhibited larger space of each flattened keratinocyte layer in the stratum corneum compared to the skin treated with KP extract in water. Moreover, ME-L10% showed good stability. Therefore, ME-L10% was a potential formulation for improving transdermal delivery of KP extract.






Similar content being viewed by others
References
Tewtrakul S, Subhadhirasakul S. Effects of compounds from Kaempferia parviflora on nitric oxide, prostaglandin E2 and tumor necrosis factor-alpha productions in RAW264.7 macrophage cells. J Ethnopharmacol. 2008;120(1):81–4.
Chuthaputti A. Krachai Dam (Kaemferiaparviflora Wall. ex Baker): a champion herbal product. J Thai Trad Alter Med. 2013;11(1):4–16.
Sutthanut K, Sripanidkulchai B, Yenjai C, Jay M. Simultaneous identification and quantitation of 11 flavonoids constituents in Kaempferia parviflora by gas chromatography. J Chromatogr A. 2007;1143(1–2):227–33.
Sutthanut K, Lu X, Sripanidkulchai B, Yenjai C, Jay M. Solid liquid nanoparticles for transdermal delivery of Kaempferia parviflora extracts. J Biomed Nanotechnol. 2009;5(2):224–32.
Mekjaruskul C, Jay M, Sripanidkulchai B. Pharmacokinetics, tissue distribution, excretion, and metabolite identification of methoxyflavones in Kaempferia parviflora extract in rats. Drug Metab Dispos. 2012;40(12):2342–53.
Sutthanut K, Lu X, Jay M, Sripanidkulchai B. Solid lipid nanoparticles for topical administration of Kaempferia parviflora extracts. J Biomed Nanotechnol. 2009;5(2):224–32.
Mekjaruskul C, Yang YT, Leed MG, Sadgrove MP, Jay M, Sripanidkulchai B. Novel formulation strategies for enhancing oral delivery of methoxyflavones in Kaempferia parviflora by SMEDDS or complexation with 2-hydroxypropyl-beta-cyclodextrin. Int J Pharm. 2013;445(1–2):1–11.
Lawrence MJ, Rees GD. Microemulsion-based media as novel drug delivery systems. Adv Drug Deliv Rev. 2012;64(supplement):175–93.
Tavano L, Alfano P, Muzzalupo R, Cindio BD. Niosomes vs microemulsions: new carriers for topical delivery of capsaicin. Colloids Surf B Biointerfaces. 2011;87(2):333–9.
Liu C, Chang FY, Hung DK. Terpene microemulsions for transdermal curcumin delivery: effects of terpenes and cosurfactants. Colloids Surf B Biointerfaces. 2011;82(1):63–70.
Patel H, Barot BS, Parejiya PB, Shelat PK, Shukla A. Topical delivery of clobetasol propionate loaded microemulsion based gel for effective treatment of vitiligo: ex vivo permeation and skin irritation studies. Colloids Surf B Biointerfaces. 2013;102:86–94.
Fouad S, Basalious EB, EL-Nabarawi MA, Tayel SA. Microemulsion and poloxamer microemulsion-based gel for sustained transdermal delivery of diclofenac epolamine using in-skin drug depot: in vitro/in vivo evaluation. Int J Pharm. 2013;453:569–78.
Sutthanut K, Sripanidkulchai B, Yenjai C, Jay M. Simultaneous identification and quantitation of 11 flavonoid constituents in Kaempferia parviflora by gas chromatography. J Chromatogr A. 2007;1143(1):227–33.
Abu-Elayazid SK, Kassem AA, Samy AM, Gomaa ME. Evaluation of skin permeation and pharmacological effects of tenoxicam nanoemulsion in topical formulations. Asian J Pharm Health Sci. 2011;1(3):99–105.
Tuntiyasawasdikul S, Limpongsa E, Jaipakdee N, Sripanidkulchai B. Transdermal permeation of Kaempferia parviflora methoxyflavones from isopropylmyristate-based vehicles. AAPS PharmSciTech. 2014;15:947–55.
Kogan A, Garti N. Microemulsions as transdermal drug delivery vehicles. Adv Colloid Interf Sci. 2006;123-126:369–85.
Idrees MA, Rahman NU, Ahmad S, Ali MY, Ahmad I. Enhance transdermal delivery of flurbiprofen via microemulsions: effects of different types of surfactants and cosurfactants. DARU J Pharm Sci. 2011;19(6):433–9.
Subongkot T, Duangjit S, Rojanarata T, Opanasopit P, Ngawhirunpat T. Ultradeformable liposomes with terpenes for delivery of hydrophilic compound. J Liposome Res. 2012;22(3):254–62.
Ngawhirunpat T, Worachun N, Opanasopit P, Rojanarata T, Panomsuk S. Cremophor RH40-PEG 400 microemulsions as transdermal drug delivery carrier for ketoprofen. Pharm Dev Technol. 2013;18(4):798–803.
Okabe H, Takayama K, Nagai T, et al. Effect of d-limonene and related compounds on the percutaneous absorption of indomethacin. Drug Des Deliv. 1989;4:313–21.
Aqil M, Ahad A, Sultana Y, Ali A. Status of terpenes as skin penetration enhancers. Drug Discov Today. 2007;12:1061–7.
Femenía-Font A, Balaguer-Fernández C, Merino V, Rodilla V, López-Castellano A. Effect of chemical enhancers on the in vitro percutaneous absorption of sumatriptan succinate. Eur J Pharm Biopharm. 2005;61(1):50–5.
Mehta SK, Kaur G. Microemulsions: thermodynamic and dynamic properties. In: Tadashi M, editor. Thermodynamics: InTech; 2011. p. 382–409.
Hoffman A. Hydrogels for biomedical applications. Adv Drug Deliv Rev. 2012;64(1):18–23.
Williams AC, Barry BW. Penetration enhancers. Adv Drug Deliv Rev. 2004;56(5):603–18.
Lu WC, Chiang BH, Huang DW, Li PH. Skin permeation of D-limonene-based nanoemulsions as a transdermal carrier prepared by ultrasonic emulsification. Ultrason Sonochem. 2014;21(2):826–32.
Cornwell PA, Barry BW. Sesquiterpene components of volatile oils as skin penetration enhancers for the hydrophilic permeant 5-fluorouracil. J Pharm Pharmacol. 1994;46(4):261–9.
Gannu R, Palem CR, Yamsani VV, Yamsani SK, Yamsani MR. Enhanced bioavailability of lacidipine via microemulsion based transdermal gels: formulation optimization, ex vivo and in vivo characterization. Int J Pharm. 2011;388(1–2):231–41.
Hathout R, Woodman TJ, Mansour S, Mortada ND, Geneidi AS, Guy RH. Microemulsion formulations for the transdermal delivery of testosterone. Eur J Pharm Sci. 2010;40(3):188–96.
Acknowledgements
The authors would like to acknowledge the Faculty of Pharmacy, Silpakorn University, the Thailand Research Funds (IRN58W0004), and the National Natural Science Foundation of China (NSFC 81561148012) for the financial supports and Bangkok Lab & Cosmetic Co., Ltd., for Kaempferia parviflora (KP) extract and standard methoxyflavones.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
ESM 1
(DOCX 62 kb)
Rights and permissions
About this article
Cite this article
Rangsimawong, W., Wattanasri, P., Tonglairoum, P. et al. Development of Microemulsions and Microemulgels for Enhancing Transdermal Delivery of Kaempferia parviflora Extract. AAPS PharmSciTech 19, 2058–2067 (2018). https://doi.org/10.1208/s12249-018-1003-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-018-1003-6