Abstract
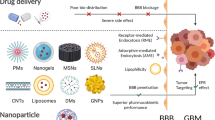
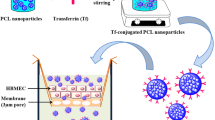
Brain tumor treatment employing methotrexate (MTX) is limited by the efflux mechanism of Pg-p on the blood–brain barrier. We aimed to investigate MTX-loaded chitosan or glycol chitosan (GCS) nanoparticles (NPs) in the presence and in the absence of a coating layer of Tween 80 for brain delivery of MTX. The effect of a low Tween 80 concentration was evaluated. MTX NPs were formulated following the ionic gelation technique and size and zeta potential measurements were acquired. Transport across MDCKII-MDR1 monolayer and cytotoxicity studies against C6 glioma cell line were also performed. Cell/particles interaction was visualized by confocal microscopy. The particles were shown to be cytotoxic against C6 cells line and able to overcome MDCKII-MDR1 cell barrier. GCS-based NPs were the most cytotoxic NPs. Confocal observations highlighted the internalization of Tween 80-coated fluorescent NPs more than Tween 80-uncoated NPs. The results suggest that even a low concentration of Tween 80 is sufficient for enhancing the transport of MTX from the NPs across MDCKII-MDR1 cells. The nanocarriers represent a promising strategy for the administration of MTX to brain tumors which merits further investigations under in vivo conditions.






Similar content being viewed by others
REFERENCES
Blakeley J. Drug delivery to brain tumors. Curr Neur Neurosci Rep. 2008;8:235–41.
Laquintana V, Trapani A, Denora N, Wang F, Gallo JM, Trapani G. New strategies to deliver anticancer drugs to the Central Nervous System. Expert Opin Drug Deliv. 2009;6:1017–32.
Provenzale JM, Mukundan S, Dewhirst M. The role of blood-brain barrier permeability in brain tumor imaging and therapeutics. Am J Roentgenol. 2005;185:763–7.
Tosi G, Costantino L, Ruozi B, Forni F, Vandelli MA. Polymeric nanoparticles for the drug delivery to the central nervous system. Exp Opin Drug Deliv. 2008;5:155–74.
Kreuter J, Ramge P, Petrov V, Hamm S, Gelperina SE, Engelhardt B, et al. Direct evidence that polysorbate-80-coated poly(butylcyanoacrilate) nanoparticles deliver drugs to the CNS via specific mechanisms requiring prior binding of drug to the nanoparticles. Pharm Res. 2003;20:409–16.
Gelperina S, Maksimenko O, Khalansky A, Vanchugova L, Shipulo E, Abbasova K, et al. Drug delivery to the brain using surfactant-coated poly(lactide-co-glycolide) nanoparticles: influence of the formulation parameters. Eur J Pharm Biopharm. 2010;74:157–63.
Zensi A, Begley D, Pontikis C, Legros C, Mihoreanu L, Wagner S, et al. Albumin nanoparticles targeted with Apo E enter the CNS by transcytosis and are delivered to neurones. J Control Release. 2009;137:78–86.
Morris PG, Abrey LE. Therapeutic challenges in primary CNS lymphoma. Lancet. 2009;8:581–92.
Duthie SJ. Folic acid-mediated inhibition of human colon cancer cell growth. Nutrition. 2001;17:736–7.
Kuznetsova N, Kandyba A, Vostrov I, Kadykov V, Gaenko G, Molotkovsky J, et al. Liposomes loaded with lipophilic prodrugs of methotrexate and melphalan as convenient drug delivery vehicles. J Drug Del Sci Tech. 2009;19:51–9.
Dhanikula RS, Argaw A, Bouchard JF, Hildgen P. Methotrexate loaded polyether-copolyester dendrimers for the treatment of gliomas: enhanced efficacy and intratumoral transport capability. Mol Pharm. 2008;5:105–16.
Denora N, Trapani A, Laquintana V, Lopedota A, Trapani G. Recent advances in medicinal chemistry and pharmaceutical technology-strategies for drug delivery to the brain. Curr Top Med Chem. 2009;9:182–96.
Csaba N, Garcia-Fuentes M, Alonso MJ. The performance of nanocarriers for transmucosal drug delivery. Expert Opin Drug Del. 2006;3:463–78.
Trapani A, Lopedota A, Franco M, Cioffi N, Ieva E, Garcia-Fuentes M, et al. A comparative study of chitosan and chitosan/cyclodextrin nanoparticles as potential carriers for the oral delivery of small peptides. Eur J Pharm Biopharm. 2010;75:26–32.
Ieva E, Trapani A, Cioffi N, Ditaranto N, Monopoli A, Sabbatini L. Analytical characterization of chitosan nanoparticles for peptide drug delivery applications. Anal Bioanal Chem. 2009;393:207–15.
Du Y, Ding Y, Sun M, Zhang L, Jiang X, Yang C. Hollow chitosan/poly(acrylic acid) nanospheres as drug carriers. Biomacromolecules. 2007;8:1069–76.
Son YJ, Jang J-S, Cho YW, Chung H, Park R-W, Kwon I-C, et al. Biodistribution and anti-tumor efficacy of doxorubicin loaded glycol-chitosan nanoaggregates by EPR effect. J Control Release. 2003;91:135–45.
Hwang HY, Kim I-S, Kwon IC, Kim YH. Tumor targetability and antitumor effect of docetaxel-loaded hydrophobically modified glycol chitosan nanoparticles. J Control Release. 2008;128:23–31.
Min KH, Park K, Kim Y-S, Bae SM, Lee S, Jo HG, et al. Hydrophobically modified glycol chitosan nanoparticles-encapsulated camptothecin enhance the drug stability and tumor targeting in cancer therapy. J Control Release. 2008;127:208–18.
Soni S, Babbar AK, Sharma RK, Banerjee T, Maitra A. Pharmacoscintigraphic evaluation of polysorbate80-coated chitosan nanoparticles for brain targeting. Am J Drug Del. 2005;3:205–12.
Mensch J, Oyarzabal J, Mackie C, Augustijns P. In vivo, in vitro and in silico methods for small molecule transfer across BBB. J Pharm Sci. 2009;98:4429–68.
Trapani A, Sitterberg J, Bakowsky U, Kissel T. The potential of glycol chitosan nanoparticles as carrier for low water soluble drugs. Int J Pharm. 2009;375:97–106.
Lopedota A, Trapani A, Cutrignelli A, Chiarantini L, Pantucci E, Curci R, et al. The use of Eudragit RS 100/cyclodextrin nanoparticles for the transmucosal administration of glutathione. Eur J Pharm Biopharm. 2009;72:509–20.
Gaillard PJ, de Boer AG. Relationship between permeability status of the blood-brain barrier and in vitro permeability coefficient of a drug. Eur J Pharm Sci. 2000;12:95–102.
Park J, Han TH, Lee KY, Han SS, Hwang JJ, Moon DH, et al. N-acetyl histidine-conjugated glycol chitosan self assembled nanoparticles for intracytoplasmic delivery of drugs: endocytosis, exocytosis and drug release. J Control Release. 2006;115:37–45.
de Campos A, Diebold Y, Carvalho ELS, Sanchez A, Alonso MJ. Chitosan nanoparticles as new ocular drug delivery systems: in vitro stability, in vivo fate, and cellular toxicity. Pharm Res. 2004;21:803–10.
Huang M, Ma Z, Khor E, Lim LY. Uptake of FITC-chitosan nanoparticles by A549 cells. Pharm Res. 2002;19:1488–94.
Behrens I, Vila Pena AI, Alonso MJ, Kissel T. Comparative uptake studies of bioadhesive and non-bioadhesive nanoparticles in human intestinal cell lines and rats: the effect of mucus on particle uptake adsorption and transport. Pharm Res. 2002;19:1185–93.
Mao S, Bakowsky U, Jintapattanakit A, Kissel T. Self assembled polyelectrolyte nanocomplexes between chitosan derivatives and insulin. J Pharm Sci. 2006;95:1035–48.
Loverre A, Ditonno P, Crovace A, Gesualdo L, Ranieri E, Pontrelli P, et al. Ischemia-reperfusion induces glomerular and tubular activation of proinflammatory and antiapoptotic pathways: differential modulation by rapamycin. J Am Soc Nephrol. 2004;15:2675–86.
Kamau SW, Kramer SD, Gunthert M, Wounderlj-Allenspach H. Effect of the modulation of the membrane lipid composition on the localization and function of P-glycoprotein in MDR1-MDCK cells. In vitro Cell. Dev Biol Anim. 2005;41:207–16.
Janes KA, Calvo P, Alonso M. Polysaccharide colloidal nanoparticles as delivery systems for macromolecules. Adv Drug Del Rev. 2001;47:83–97.
Gan Q, Wang T, Cochrane C, McCarron P. Modulation of surface charge, particle size and morphological properties of chitosan–TPP nanoparticles intended for gene delivery. Colloids Surf B Biointerfaces. 2005;44:65–73.
Wang Q, Rager JD, Weinstein K, Kardos PS, Dobson GL, Li J, et al. Evaluation of the MDR-MDCK cell line as permeability screen for the blood–brain barrier. Int J Pharm. 2005;288:349–59.
Hu Y, Ding Y, Ding D, Sun M, Zhang L, Jiang X, et al. Hollow chitosan/poly(acrylic acid) nanospheres as drug carriers. Biomacromolecules. 2007;8:1069–76.
Kohler N, Sun C, Wang J, Zhang M. Methotrexate-modified superparamagnetic nanoparticles and their intracellular uptake into human cancer cells. Langmuir. 2005;21:8558–64.
Friche E, Jensen PB, Sehested M, Demant EJ, Nissen NN. The solvents Cremophor EL and between 80 modulate daunorubicin resistance in the multidrug resistant Ehrlich ascites tumor. Cancer Commun. 1990;2:297–303.
Beduneau A, Saulnier P, Benoit J-P. Active targeting of brain tumors using nanocarriers. Biomaterials. 2007;28:4947–67.
Janes KA, Alonso MJ. Depolymerized chitosan nanoparticles for protein delivery: preparation and characterization. J Appl Polym Sci. 2003;88:2769–76.
Bonferoni MC, Sandri G, Rossi S, Ferrari F, Gibin S, Caramella C. Chitosan citrate as multifunctional polymer for vaginal delivery. Evaluation of penetration enhancement and peptidase inhibition properties. Eur J Pharm Sci. 2008;33:166–76.
ACKNOWLEDGMENTS
This project was financed by Università degli Studi di Bari (Progetti di Ateneo 2008). We thank Dr. Antonella Loverre (Dipartimento di Emergenza Trapianti di Organi, (D.E.T.O.) Bari Hospital, Bari, Italy) for interpretation of confocal microphotographs. We also thank Prof. Giuseppe Trapani (University of Bari) for helpful discussions.
Declaration of interest statement
The authors state no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trapani, A., Denora, N., Iacobellis, G. et al. Methotrexate-Loaded Chitosan- and Glycolchitosan-Based Nanoparticles: A Promising Strategy for the Administration of the Anticancer Drug to Brain Tumors. AAPS PharmSciTech 12, 1302–1311 (2011). https://doi.org/10.1208/s12249-011-9695-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-011-9695-x