Abstract
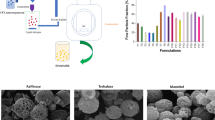
Rifampicin-loaded nanoparticles were prepared using two different molecular weights of poly-(ethylene oxide)-block-distearoyl phosphatidyl-ethanolamine (mPEG2000–DSPE and mPEG5000–DSPE) polymers. Particle sizes of all formulations studied were in the range of 162–395 nm. The entrapment efficiency (EE) was not affected by the copolymer’s molecular weight, and the highest EE (100%) was obtained with drug to copolymer ratio of 1:5. The differential scanning calorimetry (DSC) thermograms showed Tg of rifampicin-loaded PEG–DSPE nanoparticles that shifted to a lower value, indicating entrapment of rifampicin in polymer matrix. The Fourier transformed infrared spectra revealed no chemical interactions between the drug and both copolymers. The in vitro drug release from the formulations occurred over 3 days and followed first-order release kinetic and Higuchi diffusion model. The nebulization of rehydrated lyophilized rifampicin mPEG–DSPE formulations had mass median aerodynamic diameter of 2.6 µm and fine particle fraction of 42%. The aerodynamic characteristic of the preparations was not influenced by the molecular weight of the copolymers. Therefore, it is suggested that both mPEG–DSPE are promising candidates as rifampicin carrier for pulmonary delivery.







Similar content being viewed by others
References
World Health Organization. Tuberculosis facts 200. Geneva: WHO; 2007.
Burman WJ, Cohn DL, Rietmeijer CA, Judson FN, Sbarbaro JA, Reves RR. Noncompliance with directly observed therapy for tuberculosis: epidemiology and effect on the outcome of treatment. Chest. 1997;111:1168–73.
Fu J, Fiegel J, Krauland E, Hanes J. New polymeric carriers for controlled drug delivery following inhalation or injection. Biomaterials. 2002;23:4425–33.
Prabakaran D, Singh P, Jaganathan KS, Vyas SP. Osmotically regulated asymmetric capsular systems for simultaneous sustained delivery of anti-tubercular drugs. J Control Release. 2004;95:239–48.
Shishoo CJ, Shah SA, Rathod IS, Savale SS, Vora MJ. Impaired bioavailability of rifampicin in presence of isoniazid from fixed dose combination (FDC) formulation. Int J Pharm. 2001;228:53–67.
Singh S, Mariappan TT, Shankar R, Sarda N, Singh B. A critical review of the probable reasons for the poor/variable bioavailability of rifampicin from anti-tubercular fixed-dose combination (FDC) products, and the likely solutions to the problem. Int J Pharm. 2001;228:5–17.
Dutt M, Khuller GK. Liposomes and PLG microparticles as sustained release antitubercular drug carriers-an in vitro-in vivo study. Int J Antimicrob Agents. 2001;18:245–52.
O'Hara P, Hickey AJ. Respirable PLGA microspheres containing rifampicin for the treatment of tuberculosis: manufacture and characterization. Pharm Res. 2000;17:955–61.
Rao BS, Murthy KVR. Studies on rifampicin release from ethylcellulose coated nonpareil beads. Int J Pharm. 2002;231:97–106.
Suarez S, O'Hara P, Kazantseva M, Newcomer CE, Hopfer R, McMurray DN et al. Airways delivery of rifampicin microparticles for the treatment of tuberculosis. J Antimicrob Chemother. 2001;48:431–4.
Vyas SP, Kannan ME, Jain S, Mishra V, Singh P. Design of liposomal aerosols for improved delivery of rifampicin to alveolar macrophages. Int J Pharm. 2004;269:37–49.
Gaber NN, Darwis Y, Peh KK, Tan YTF. Characterization of polymeric micelles for pulmonary delivery of beclomethasone dipropionate. J Nanosci Nanotechnol. 2006;6:1–7.
Torchilin VP. Polymer-coated long-circulating microparticulate pharmaceuticals. J Microencapsul. 1998;15:1–19.
Zalipsky S. Long circulating, cationic liposomes containing amino-PEG-phosphatidylethanolamine. FEBS Lett. 1994;353:71–4.
Allen C, Maysinger D, Eisenberg A. Nano-engineering block copolymer aggregates for drug delivery. Colloids Surf B: Biointerfaces. 1999;16:3–27.
Jones MC, Leroux JC. Polymeric micelles—a new generation of colloidal drug carriers. Eur J Pharm Biopharm. 1999;48:101–11.
Lukyanov AN, Gao Z, Torchilin VP. Micelles from polyethylene glycol/phosphatidylethanolamine conjugates for tumor drug delivery. J Control Release. 2003;91:97–102.
Yamamoto H, Kuno Y, Sugimoto S, Takeuchi H, Kawashima Y. Surface-modified PLGA nanosphere with chitosan improved pulmonary delivery of calcitonin by mucoadhesion and opening of the intercellular tight junctions. J Control Release. 2005;102:373–81.
Azarmi S, Roa WH, Lolbenberg R. Targeted delivery of nanoparticles for the treatment of lung diseases. Adv Drug Deliv Rev. 2008;60:863–75.
Suarez S, Hickey AJ. Drug properties affecting aerosol behavior. Respir Care. 2000;45:652–66.
Joshi M, Misra A. Dry powder inhalation of liposomal Ketotifen fumarate: Formulation and characterization. Int J Pharma. 2001;223:15–27.
Sung JC, Pulliam BL, Edwards DA. Nanoparticles for drug delivery to the lungs. Trends Biotechnol. 2007;25:563–70.
Wiggins NA. The development of a mathematical approximation technique to determine the mass median aerodynamic diameter (MMAD) and geometric standard deviation (GSD) of drug particles in an inhalation aerosol spray. Drug Dev Ind Pharm. 1991;17:1971–86.
Hu Y, Jiang X, Ding Y, Zhang L, Yang C, Zhang J et al. Preparation and drug release behaviors of nimodipine-loaded poly(caprolactone)-poly(ethylene oxide)-polylactide amphiphilic copolymer nanoparticles. Biomaterials. 2003;24:2395–404.
Kreuter J. Nanoparticles—a historical perspective. Int J Pharm. 2007;331:1–10.
Chou TH, Chu IM. Thermodynamic characteristics of DSPC/DSPE-PEG2000 mixed monolayers on the water subphase at different temperatures. Colloids Surf B: Biointerfaces. 2003;27:333–44.
Zhang X, Jackson JK, Burt HM. Development of amphiphilic diblock copolymers as micellar carriers of taxol. Int J Pharm. 1996;132:195–206.
Darwis Y, Kellaway IW. Nebulisation of rehydrated freeze-dried beclomethasone dipropionate liposomes. Int J Pharm. 2001;215:113–21.
Agrawal S, Ashokraj Y, Bharatam PV, Pillai O, Panchagnula R. Solid-state characterization of rifampicin samples and its biopharmaceutic relevance. Eur J Pharm Sci. 2004;22:127–44.
Rastogi R, Sultana Y, Aqil M, Ali A, Kumar S, Chuttani K et al. Alginate microspheres of isoniazid for oral sustained drug delivery. Int J Pharm. 2007;334:71–7.
Kim SY, Shin ILG, Lee YM, Cho CS, Sung YK. Methoxy poly (ethylene glycol) and Є-caprolactone amphiphilic block copolymeric micelle containing indomethacin. II. Micelle formulation and drug release behaviours. J Control Release. 1998;51:13–22.
Lavasanifar A, Samuel J, Kwon GS. Poly(ethylene oxide)-block-poly(L-amino acid) micelles for drug delivery. Adv Drug Deliv Rev. 2002;54:169–90.
Zhang Y, Zhuo RX. Synthesis and in vitro drug release behavior of amphiphilic triblock copolymer nanoparticles based on poly (ethylene glycol) and polycaprolactone. Biomaterials. 2005;26:6736–42.
Vaghi A, Berg E, Liljedahl S, Svensson JO. In vitro comparison of nebulised budesonide (Pulmicort Respules®) and beclomethasone dipropionate (Clenil® per Aerosol). Pulm Pharmacol Ther. 2005;18:151–3.
Hickey AJ, Kuchel K, Masinde LE. Method of aerosol particle size characterization. In: Hickey AJ, editor. Pharmaceutical inhalation aerosol technology. New York: Marcel Dekker; 1992. p. 218–53.
Acknowledgment
The authors would like to thank Universiti Sains Malaysia, Penang, Malaysia, for providing the research grant to support this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdulla, J.M.A., Tan, Y.TF. & Darwis, Y. Rehydrated Lyophilized Rifampicin-Loaded mPEG–DSPE Formulations for Nebulization. AAPS PharmSciTech 11, 663–671 (2010). https://doi.org/10.1208/s12249-010-9428-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-010-9428-6