Abstract
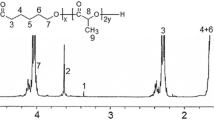
This study aimed to design methyprednisolone (MP)-loaded poly(d,l lactide-co-glycolide) (PLGA) microspheres (MS) intended for intra-articular administration. MP was encapsulated in four different types of PLGA by using an S/O/W technique. The effects of β-irradiation at the dose of 25 kGy were evaluated on the chemical and physicochemical properties of MS and the drug release profiles. The S/O/W technique with hydroxypropylmethylcellulose (HPMC) as surfactant allowed obtaining MS in the tolerability size (7–50 µm) for intra-articular administration. The MP encapsulation efficiency ranged 56–60%. HPMC traces were evidenced in the loaded and placebo MS by attenuated total reflectance Fourier transform infrared spectroscopy. MS made of the capped PLGA DL5050 2M (MS 2M) and uncapped PLGA DL5050 3A (MS 3A) prolonged the release of MP over a 2- to 3-month period with a triphasic (burst release–dormant period–second release pulse) and biphasic release pattern, respectively. The β-irradiation did not significantly alter the morphology, chemical, and physicochemical properties of MS. The only variation was evidenced in the drug release for MS 2M in term of shorting of the dormant period. The minimal variations in the properties of irradiated PLGA MS, which are in disagreement with literature data, may be attributed to a radioprotecting effect exerted by HPMC.





Similar content being viewed by others
References
W. Lavelle, E. D. Lavelle, and L. Lavelle. Intra-articular injections. Med. Clin. N. Am. 91:241–250 (2007).
N. Gerwin, C. Hops, and A. Lucke. Intra-articular drug delivery in osteoarthritis. Adv. Drug Del. Rev. 58:226–242 (2006).
H. Derendorf, H. Mollmann, A. Gruner, D. Haack, and G. Gyselby. Pharmacokinetics and pharmacodynamics of glucocorticoid suspensions after intra-articular administration. Clin. Pharmacol. Ther. 39:313–317 (1986).
J. H. Ratcliffe, I. M. Hunneyball, A. Smith, C. G. Wilson, and S. S. Davis. Preparation and evaluation of biodegradable polymeric systems for the intra-articular delivery of drugs. J. Pharm. Pharmacol. 36(7):431–434 (1984).
World Anti-Doping Agency Prohibited List, version January 2007. www.wada-ama.org.
E. Horisawa, K. Kubota, I. Tuboi, K. Sato, H. Yamamoto, H. Takeuchi, and Y. Kawashima. Size-dependency of DL-lactide/glycolide copolymer particulates for intra-articular delivery system on phagocytosis in rat synovium. Pharm. Res. 19:132–139 (2002).
A. G. Hausberg, R. A. Kenley, and P. P. DeLuca. Gamma irradiation effects on molecular weight degradation of poly(DL-lactide-co-glycolide) microparticles. Pharm. Res. 12:851–856 (1995).
L. Montanari, M. Costantini, E. C. Signorotti, L. Valvo, M. Cantucci, M. Bartolomei, P. Fattibene, S. Onori, A. Faucitano, B. Conte, and I. Genta. Gamma irradiation effects on poly (DL-lactide-co-glycolide) microspheres. J. Controll. Rel. 56:219–229 (1998).
Y. W. Jo, B. H. Woo, A. M. Hazrati, and P. P. DeLuca. Use of Pharmasep unit for processing microspheres. AAPS PharmSciTech. 2(1): article. http://www.aapspharmscitech.org (2001).
B. Bittner, K. Mäder, C. Kroll, H. H. Borchert, and T. Kissel. Tetracycline HCl-loaded poly(DL-lactide-co-glycolide) microspheres prepared by a spray-drying technique: influence of γ-irradiation on radical formation and polymer degradation. J. Controll. Rel. 59:23–32 (1999).
C. Martínez-Sancho, R. Herrero-Vanrell, and S. Negro. Study of gamma-irradiation effects on aciclovir. poly(DL-lactic-co-glycolic) acid microspheres for intravitreal administration. J. Controll. Rel. 99(21):41–52 (2004).
J. S. C. Loo, C. P. Ooi, and F. Y. C. Boey. Degradation of poly(lactide-co-glycolide) (PLGA) and poly(L-lactide) (PLLA) by electron beam radiation. Biomater. 26:1359–1376 (2005).
L. Montanari, F. Cilurzo, F. Selmin, B. Conti, I. Genta, G. Poletti, F. Orsini, and L. Valvo. Poly(lactide-co-glycolide) microspheres containing bupivacaine: comparison between gamma and beta irradiation effects. J. Controll. Rel. 90:281–190 (2003).
J. A. Bushell, M. Claybourn, H. E. Williams, and D. M. Murphy. EPR and ENDOR study of γ- and β-radiation sterilization in poly(lactide-co-glycolide) polymers and microspheres. J. Controll. Rel. 110:49–57 (2005).
W. Friess, and M. Schlapp. Sterilization of gentamicin containing collagen/PLGA microparticle composites. Eur. J. Pharm. Biopharm. 63:176–187 (2006).
J. Mauduit, N. Bukh, and M. Vert. Gentamycin/poly(lactic acid) blends aimed at sustained release local antibiotic therapy administered per-operatively. I. The case of gentamycin base and gentamycin sulfate in poly(D,L-lactic acid) oligomers. J. Controll. Rel. 23:209–220 (1993).
P. Sansdrap, and A. J. Moës. Influence of manufacturing parameters on the size characteristics and the release profiles of nifedipine from poly(DL-lactide-co-glycolide) microspheres. Int. J. Pharm. 98(1–3):157–174 (1993).
S. Giovagnoli, P. Blasi, A. Schoubben, C. Rossi, and M. Ricci. Preparation of large porous biodegradable microspheres by using a simple double-emulsion method for capreomycin sulfate pulmonary delivery. Int. J. Pharm. 333:103–111 (2007).
W. I. Higuchi, W. E. HamLin, and S. C. Metha. Infrared attenuated total reflectance (ATR) method for observing the water-mediated surface phase reversion of methylprednisolone II to I during dissolution. J. Pharm. Sci. 58(9):1145–1146 (1969).
M. P. Kane, and K. Tsuji. Radiolytic degradation scheme for cobalt-60-irradiated corticosteroids. J. Pharm. Sci. 72:30–35 (1983).
F. Cilurzo, F. Selmin, P. Minghetti, L. Montanari, C. Lenardi, F. Orsini, and G. Poletti, Comparison between gamma and beta irradiation effects on hydroxypropylmethylcellulose and gelatin hard capsules. AAPS PharmSciTech 6(4): article 73. http://www.aapspharmscitech.org (2005).
L. Maggi, L. Segale, E. Ochoa Maciste, A. Buttafava, A. Faucitano, and U. Conte. Chemical and physical stability of hydroxypropylmethylcellulose matrices containing diltiazem hydrochloride after gamma irradiation. J. Pharm. Sci. 92:131–140 (2003).
L. Montanari, F. Cilurzo, B. Conti, I. Genta, A. Groppo, L. Valvo, A. Faucitano, and A. Buttafava. Gamma irradiation effects and EPR investigation on poly(lactide-co-glycolide) microspheres containing bupivacaine. Il Farmaco. 57:427–433 (2002).
Acknowledgments
This research project was supported by the grant “Ricerca 2006 sui farmaci, sulle sostanze e sulle pratiche mediche utilizzabili a fini doping nelle attività sportive” from the Ministero della Salute (Rome, Italy). The authors would like to acknowledge Dr. Silvio Faragò of Centro Sperimentale della Seta (Milan, Italy) for the SEM investigations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cilurzo, F., Selmin, F., Minghetti, P. et al. Design of Methylprednisolone Biodegradable Microspheres Intended for Intra-articular Administration. AAPS PharmSciTech 9, 1136–1142 (2008). https://doi.org/10.1208/s12249-008-9158-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-008-9158-1