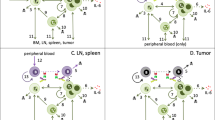
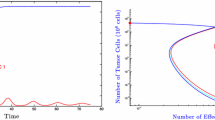
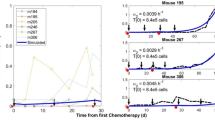
Abstract
Chimeric antigen receptor (CAR) T-cell therapy is an immunotherapy that has recently become highly instrumental in the fight against life-threatening diseases. A variety of modeling and computational simulation efforts have addressed different aspects of CAR T-cell therapy, including T-cell activation, T- and malignant cell population dynamics, therapeutic cost-effectiveness strategies, and patient survival. In this article, we present a systematic review of those efforts, including mathematical, statistical, and stochastic models employing a wide range of algorithms, from differential equations to machine learning. To the best of our knowledge, this is the first review of all such models studying CAR T-cell therapy. In this review, we provide a detailed summary of the strengths, limitations, methodology, data used, and data gap in currently published models. This information may help in designing and building better models for enhanced prediction and assessment of the benefit-risk balance associated with novel CAR T-cell therapies, as well as with the data need for building such models.




Similar content being viewed by others
References
Gross G, Waks T, Eshhar Z. Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci U S A. 1989;86(24):10024–8.
Novartis Pharmaceuticals Corporation. Kymriah™ (tisagenlecleucel) [Package Insert]. East Hanover, NJ. 2018.
Kite Pharma Inc. Yescarta® (axicabtagene ciloleucel) [Package Insert]. Santa Monica, CA. 2017.
Newick K, O'Brien S, Moon E, Albelda SM. CAR T cell therapy for solid tumors. Annu Rev Med. 2017;68:139–52.
Seif M, Einsele H, Loeffler J. CAR T Cells beyond cancer: hope for immunomodulatory therapy of infectious diseases. Front Immunol. 2019;10:2711.
Hernandez-Vargas EA. Modeling and control of infectious diseases in the Host. 1 ed. 1, 2019. 256 p.
Tomlin CJ, Axelrod JD. Biology by numbers: mathematical modelling in developmental biology. Nat Rev Genet. 2007;8(5):331–40.
Bansal A, Sullivan SD, Lin VW, Purdum AG, Navale L, Cheng P, et al. Estimating long-term survival for patients with relapsed or refractory large B-cell lymphoma treated with chimeric antigen receptor therapy: a comparison of standard and mixture cure models. Med Decis Mak. 2019;39(3):294–8.
Finney OC, Brakke HM, Rawlings-Rhea S, Hicks R, Doolittle D, Lopez M, et al. CD19 CAR T cell product and disease attributes predict leukemia remission durability. J Clin Invest. 2019;129(5):2123–32.
George JT, Levine H. Stochastic modeling of tumor progression and immune evasion. J Theor Biol. 2018;458:148–55.
Hanson S. Mathematical modelling of immuno-oncology and related immunology. Duke University; 2019.
Whittington MD, McQueen RB, Ollendorf DA, Kumar VM, Chapman RH, Tice JA, et al. Long-term survival and value of chimeric antigen receptor T-cell therapy for pediatric patients with relapsed or refractory leukemia. JAMA Pediatr. 2018;172(12):1161–8.
de Jesus Rodrigues B, Barros LRC, Almeida RC. Three-compartment model of CAR T-cell immunotherapy. bioRxiv. 2019;779793.
Hopkins B, Pan Y, Tucker M, Huang ZJ. A model-based investigation of cytokine dynamics in immunotherapies. Processes. 2019;7(1):12.
Kimmel GJ, Locke FL, Altrock PM. Evolutionary dynamics of CAR T cell therapy. bioRxiv. 2019;717074.
Mostolizadeh R, Afsharnezhad Z, Marciniak-Czochra A. Mathematical model of chimeric anti-gene receptor (CAR) T cell therapy with presence of cytokine. Numer Algebra Control Optim. 2018;8(1):63.
Talkington A, Dantoin C, Durrett R. Ordinary differential equation models for adoptive immunotherapy. Bull Math Biol. 2018;80(5):1059–83.
Hanson S, Grimes DR, Taylor-King JP, Bauer B, Warman PI, Frankenstein Z, et al. Toxicity management in CAR T cell therapy for B-ALL: mathematical modelling as a new avenue for improvement. BioRxiv. 2016:049908.
Sahoo P, Yang X, Abler D, Maestrini D, Adhikarla V, Frankhouser D, et al. Mathematical deconvolution of CAR T-cell proliferation and exhaustion from real-time killing assay data. J R Soc Interface. 2020;17(162).
Schacht C, Meade A, Banks HT, Enderling H, Abate-Daga D. Estimation of probability distributions of parameters using aggregate population data: analysis of a CAR T-cell cancer model. Math Biosci Eng. 2019;16(6):7299–326.
Teachey DT, Lacey SF, Shaw PA, Melenhorst JJ, Maude SL, Frey N, et al. Identification of predictive biomarkers for cytokine release syndrome after chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Cancer Discov. 2016;6(6):664–79.
Toor AA, Chesney A, Zweit J, Reed J, Hashmi SK. A dynamical systems perspective on chimeric antigen receptor T-cell dosing. Bone Marrow Transplant. 2019;54(3):485–9.
Madabushi R, Benjamin JM, Grewal R, Pacanowski MA, Strauss DG, Wang Y, et al. The US Food and Drug Administration’s model-informed drug development paired meeting pilot program: early experience and impact. Clin Pharmacol Ther. 2019;106(1):74–8.
Unni P, Seshaiyer P. Mathematical modeling, analysis, and simulation of tumor dynamics with drug interventions. Comput Math Methods Med. 2019;2019:4079298.
Makaryan SZ, Cess CG, Finley SD. Modeling immune cell behavior across scales in cancer. Wiley Interdisciplinary Reviews: Systems Biology and Medicine. 2020:e1484.
Rohrs JA, Wang P, Finley SD. Understanding the dynamics of T-cell activation in health and disease through the lens of computational modeling. JCO Clin Cancer Inform. 2019;3:1–8.
Chaudhury A, Zhu X, Chu L, Goliaei A, June CH, Kearns JD, et al. Chimeric antigen receptor T cell therapies: a review of cellular kinetic-pharmacodynamic modeling approaches. J Clin Pharmacol. 2020;60:S147–S59.
Zhao Z, Condomines M, van der Stegen SJC, Perna F, Kloss CC, Gunset G, et al. Structural design of engineered costimulation determines tumor rejection kinetics and persistence of CAR T cells. Cancer Cell. 2015;28(4):415–28.
Harris DT, Hager MV, Smith SN, Cai Q, Stone JD, Kruger P, et al. Comparison of T cell activities mediated by human TCRs and CARs that use the same recognition domains. J Immunol. 2018;200(3):1088–100.
Kersh EN, Shaw AS, Allen PM. Fidelity of T cell activation through multistep T cell receptor ζ phosphorylation. Science. 1998;281(5376):572–5.
Housden HR, Skipp PJ, Crump MP, Broadbridge RJ, Crabbe T, Perry MJ, et al. Investigation of the kinetics and order of tyrosine phosphorylation in the T-cell receptor ζ chain by the protein tyrosine kinase Lck. Eur J Biochem. 2003;270(11):2369–76.
Mukhopadhyay H, Cordoba S-P, Maini PK, van der Merwe PA, Dushek O. Systems model of T cell receptor proximal signaling reveals emergent ultrasensitivity. PLoS Comput Biol. 2013;9(3):e1003004.
Mukhopadhyay H, de Wet B, Clemens L, Maini PK, Allard J, van der Merwe PA, et al. Multisite phosphorylation modulates the T cell receptor ζ-chain potency but not the switchlike response. Biophys J. 2016;110(8):1896–906.
Rohrs JA, Zheng D, Graham NA, Wang P, Finley SD. Computational model of chimeric antigen receptors explains site-specific phosphorylation kinetics. Biophys J. 2018;115(6):1116–29.
Rohrs JA, Siegler EL, Wang P, Finley SD. ERK activation in CAR T cells is amplified by CD28-mediated increase in CD3ζ phosphorylation. iScience. 2020;23(4):101023.
Cess CG, Finley SD. Data-driven analysis of a mechanistic model of CAR T cell signaling predicts effects of cell-to-cell heterogeneity. J Theor Biol. 2020;489:110125.
Ali S, Kjeken R, Niederlaender C, Markey G, Saunders TS, Opsata M, et al. The European medicines agency review of Kymriah (Tisagenlecleucel) for the treatment of acute lymphoblastic leukemia and diffuse large B-cell lymphoma. Oncologist 2020;25(2):\.
Cao J-X, Gao W-J, You J, Wu L-H, Liu J-L, Wang Z-X. The efficacy of anti-CD19 chimeric antigen receptor T cells for B-cell malignancies. Cytotherapy. 2019;21(7):769–81.
Ninomiya S, Narala N, Huye L, Yagyu S, Savoldo B, Dotti G, et al. Tumor indoleamine 2, 3-dioxygenase (IDO) inhibits CD19-CAR T cells and is downregulated by lymphodepleting drugs. Blood J Am Soc Hematol. 2015;125(25):3905–16.
Ruella M, Klichinsky M, Kenderian SS, Shestova O, Ziober A, Kraft DO, et al. Overcoming the immunosuppressive tumor microenvironment of Hodgkin lymphoma using chimeric antigen receptor T cells. Cancer Discov. 2017;7(10):1154–67.
Singh AP, Zheng X, Lin-Schmidt X, Chen W, Carpenter TJ, Zong A, et al. Development of a quantitative relationship between CAR-affinity, antigen abundance, tumor cell depletion and CAR-T cell expansion using a multiscale systems PK-PD model. MAbs. 2020;12(1):1688616.
Awasthi R, Pacaud L, Waldron E, Tam CS, Jager U, Borchmann P, et al. Tisagenlecleucel cellular kinetics, dose, and immunogenicity in relation to clinical factors in relapsed/refractory DLBCL. Blood Adv. 2020;4(3):560–72.
Stein AM, Grupp SA, Levine JE, Laetsch TW, Pulsipher MA, Boyer MW, et al. Tisagenlecleucel model-based cellular kinetic analysis of chimeric antigen receptor-T cells. CPT Pharmacometrics Syst Pharmacol. 2019;8(5):285–95.
Hardiansyah D, Ng CM. Quantitative systems pharmacology model of chimeric antigen receptor T-cell therapy. Clin Transl Sci. 2019;12(4):343–9.
Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507–17.
Mueller KT, Maude SL, Porter DL, Frey N, Wood P, Han X, et al. Cellular kinetics of CTL019 in relapsed/refractory B-cell acute lymphoblastic leukemia and chronic lymphocytic leukemia. Blood. 2017;130(21):2317–25.
Mueller KT, Waldron E, Grupp SA, Levine JE, Laetsch TW, Pulsipher MA, et al. Clinical pharmacology of tisagenlecleucel in B-cell acute lymphoblastic leukemia. Clin Cancer Res. 2018;24(24):6175–84.
Liu C, Ayyar VS, Zheng X, Chen W, Zheng S, Mody H, et al. Model-based cellular kinetic analysis of chimeric antigen receptor-t cells in humans. Clin Pharmacol Ther. 2020.
De Boer RJ, Homann D, Perelson AS. Different dynamics of CD4+ and CD8+ T cell responses during and after acute lymphocytic choriomeningitis virus infection. J Immunol. 2003;171(8):3928–35.
Grant TS, Burns D, Kiff C, Lee D. A case study examining the usefulness of cure modelling for the prediction of survival based on data maturity. Pharmacoeconomics. 2019. https://doi.org/10.1007/s40273-019-00867-5.
Lin JK, Lerman BJ, Barnes JI, Boursiquot BC, Tan YJ, Robinson AQL, et al. Cost effectiveness of chimeric antigen receptor T-cell therapy in relapsed or refractory pediatric B-cell acute lymphoblastic leukemia. J Clin Oncol. 2018:JCO2018790642.
Furzer J, Gupta S, Nathan PC, Schechter T, Pole JD, Krueger J, et al. Cost-effectiveness of tisagenlecleucel vs standard care in high-risk relapsed pediatric acute lymphoblastic leukemia in Canada. JAMA Oncol. 2020:e195909.
Sarkar RR, Gloude NJ, Schiff D, Murphy JD. Cost-effectiveness of chimeric antigen receptor T-cell therapy in pediatric relapsed/refractory B-cell acute lymphoblastic leukemia. J Natl Cancer Inst. 2019;111(7):719–26.
Whittington MD, McQueen RB, Ollendorf DA, Kumar VM, Chapman RH, Tice JA, et al. Long-term survival and cost-effectiveness associated with axicabtagene ciloleucel vs chemotherapy for treatment of B-cell lymphoma. JAMA Netw Open. 2019;2(2):e190035.
Roth JA, Sullivan SD, Lin VW, Bansal A, Purdum AG, Navale L, et al. Cost-effectiveness of axicabtagene ciloleucel for adult patients with relapsed or refractory large B-cell lymphoma in the United States. J Med Econ. 2018;21(12):1238–45.
Banwarth-Kuhn M, Sindi S. How and why to build a mathematical model: a case study using prion aggregation. J Biol Chem. 2020;295(15):5022–35.
Chelliah V, Lazarou G, Bhatnagar S, Gibbs JP, Nijsen M, Ray A, et al. Quantitative systems pharmacology approaches for immuno-oncology: adding virtual patients to the development paradigm. Clin Pharmacol Ther. 2020.
Polak S, Tylutki Z, Holbrook M, Wiśniowska B. Better prediction of the local concentration–effect relationship: the role of physiologically based pharmacokinetics and quantitative systems pharmacology and toxicology in the evolution of model-informed drug discovery and development. Drug Discov Today. 2019;24(7):1344–54.
Marshall S, Burghaus R, Cosson V, Cheung S, Chenel M, DellaPasqua O, et al. Good practices in model-informed drug discovery and development: practice, application, and documentation. CPT Pharmacometrics Syst Pharmacol. 2016;5(3):93–122.
Kuemmel C, Yang Y, Zhang X, Florian J, Zhu H, Tegenge M, et al. Consideration of a credibility assessment framework in model-informed drug development: potential application to physiologically-based pharmacokinetic modeling and simulation. CPT Pharmacometrics Syst Pharmacol. 2020;9(1):21–8.
Turtle CJ, Hanafi LA, Berger C, Gooley TA, Cherian S, Hudecek M, et al. CD19 CAR-T cells of defined CD4+:CD8+ composition in adult B cell ALL patients. J Clin Invest. 2016;126(6):2123–38.
Turtle CJ, Hanafi L-A, Berger C, Hudecek M, Pender B, Robinson E, et al. Immunotherapy of non-Hodgkin’s lymphoma with a defined ratio of CD8+ and CD4+ CD19-specific chimeric antigen receptor–modified T cells. Sci Transl Med. 2016;8(355):355ra116.
Neelapu SS. CAR-T efficacy: is conditioning the key? Blood. J Am Soc Hematol. 2019;133(17):1799–800.
Hay KA, Hanafi LA, Li D, Gust J, Liles WC, Wurfel MM, et al. Kinetics and biomarkers of severe cytokine release syndrome after CD19 chimeric antigen receptor-modified T-cell therapy. Blood. 2017;130(21):2295–306.
Gorovits B, Koren E. Immunogenicity of Chimeric antigen receptor T-cell therapeutics. J BioDrugs. 2019:1–10.
Xia A-L, Wang X-C, Lu Y-J, Lu X-J, Sun B. Chimeric-antigen receptor T (CAR-T) cell therapy for solid tumors: challenges and opportunities. Oncotarget. 2017;8(52):90521–31.
Date V, Nair S. Emerging vistas in CAR T-cell therapy: challenges and opportunities in solid tumors. Expert Opin Biol Ther. 2020:1–16.
Ramakrishna S, Barsan V, Mackall C. Prospects and challenges for use of CAR T cell therapies in solid tumors. Expert Opin Biol Ther. 2020;20(5):503–16.
Bagley SJ, O’Rourke DM. Clinical investigation of CAR T cells for solid tumors: lessons learned and future directions. Pharmacol Ther. 2020;205:107419.
Castellarin M, Sands C, Da T, Scholler J, Graham K, Buza E, et al. A rational mouse model to detect on-target, off-tumor CAR T cell toxicity. JCI Insight. 2020;5(14).
Gardner RA, Ceppi F, Rivers J, Annesley C, Summers C, Taraseviciute A, et al. Preemptive mitigation of CD19 CAR T-cell cytokine release syndrome without attenuation of antileukemic efficacy. Blood. 2019;134(24):2149–58.
Kadauke S, Myers RM, Li Y, Aplenc R, Baniewicz D, Barrett DM, et al. Risk-adapted preemptive tocilizumab to prevent severe cytokine release syndrome after CTL019 for pediatric b-cell acute lymphoblastic leukemia: a prospective clinical trial. J Clin Oncol. JCO.20.02477.
Si S, Teachey DT. Spotlight on tocilizumab in the treatment of CAR-T-cell-induced cytokine release syndrome: clinical evidence to date. Ther Clin Risk Manag. 2020;16:705.
Majzner RG, Mackall CL. Clinical lessons learned from the first leg of the CAR T cell journey. Nat Med. 2019;25(9):1341–55.
Zhang J, Li J, Ma Q, Yang H, Signorovitch J, Wu E. A review of two regulatory approved anti-CD19 CAR T-cell therapies in diffuse large B-cell lymphoma: why are indirect treatment comparisons not feasible? Adv Ther. 2020;37(7):3040–58.
Locke FL, Rossi JM, Neelapu SS, Jacobson CA, Miklos DB, Ghobadi A, et al. Tumor burden, inflammation, and product attributes determine outcomes of axicabtagene ciloleucel in large B-cell lymphoma. Blood Adv. 2020;4(19):4898–911.
Milone MC, Bhoj VG. The pharmacology of T cell therapies. Mol Ther Methods Clin Dev. 2018;8:210–21.
Valentinuzzi D, Jeraj R. Computational modelling of modern cancer immunotherapy. Phys Med Biol. 2020;65:24TR01.
Acknowledgements
The authors thank Gwendolyn Halford, FDA Library, for curating the literature search queries. We would like to thank Joanne Berger and Daniel Sloper, FDA Library, for editing the manuscript.
Funding
This project was supported in part by an appointment to the Research Participation Program at OBE/CBER, U.S. Food and Drug Administration, administered by the Oak Ridge Institute for Science and Education through an interagency agreement between the U.S. Department of Energy and the FDA.
Author information
Authors and Affiliations
Contributions
Conceptualization: UN, MRM, ONY, HY
Data curation: UN, ONY
Formal analysis: UN, MRM, ONY, XW
Funding acquisition: HY
Investigation: UN, MRM, ONY, XW
Methodology: UN, ONY
Project administration: ONY, HY
Resources: HY
Supervision: HY
Visualization: UN, MRM
Writing—original draft: UN, MRM, ONY, XW, HY
Writing—review and editing: UN, MRM, ONY, XW, HY
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Disclaimer
This article reflects the views of the authors and should not be construed to represent the FDA’s views or policies. These comments do not bind or obligate FDA.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 111 kb)
Rights and permissions
About this article
Cite this article
Nukala, U., Rodriguez Messan, M., Yogurtcu, O.N. et al. A Systematic Review of the Efforts and Hindrances of Modeling and Simulation of CAR T-cell Therapy. AAPS J 23, 52 (2021). https://doi.org/10.1208/s12248-021-00579-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12248-021-00579-9