Abstract
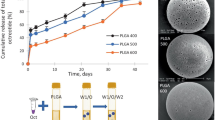
The 1-month Lupron Depot® (LD) encapsulating water-soluble leuprolide in poly(lactic-co-glycolic acid) (PLGA) microspheres is a benchmark product upon which modern long-acting release products are often compared. Despite expiration of patent coverage, no generic product for the LD has been approved in the USA, likely due to the complexity of components and manufacturing processes involved in the product. Here, we describe the reverse engineering of the LD composition and important product attributes. Specific attributes analyzed for microspheres were as follows: leuprolide content by three methods; gelatin content, type, and molecular weight distribution; PLGA content, lactic acid/glycolic acid ratio, and molecular weight distribution; mannitol content; in vitro drug release; residual solvent and moisture content; particle size distribution and morphology; and glass transition temperature. For the diluent, composition, viscosity, and specific gravity were analyzed. Analyzed contents of the formulation and the determined PLGA characteristics matched well with the official numbers stated in the package insert and those found in literature, respectively. The gelatin was identified as type B consistent with ~ 300 bloom. The 11-μm volume-median microspheres in the LD slowly released the drug in vitro in a zero-order manner after ~ 23% initial burst release. Very low content of residual moisture (< 0.5%) and methylene chloride (< 1 ppm) in the product indicates in-water drying is capable of removing solvents to extremely low levels during manufacturing. The rigorous approach of reverse engineering described here may be useful for development of generic leuprolide-PLGA microspheres as well as other new and generic PLGA microsphere formulations.










Similar content being viewed by others
References
Okada H. One- and three-month release injectable microspheres of the LH-RH superagonist leuprorelin acetate. Adv Drug Deliv Rev. 1997;28(1):43–70.
Plosker GL, Brogden RN. Leuprorelin. Drugs. 1994;48(6):930–67.
Annual reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934. United States Securities and Exchange Commission.
21 CFR 314.94(a)(9)(iii).
Wang Y, Wen Q, Choi SH. FDA’s regulatory science program for generic PLA/PLGA-based drug products. Am Pharm Rev. 2016;19(4):5–9.
Lupron Depot® package insert. [Internet]. AbbVie Inc.; Available from: http://www.rxabbvie.com/pdf/lupronuro_pi.pdf
Ogawa Y, Yamamoto M, Okada H, Yashiki T, Shimamoto T. A new technique to efficiently entrap leuprolide acetate into microcapsules of polylactic acid or copoly(lactic glycolic) acid. Chem Pharm Bull (Tokyo). 1988;36(3):1095–103.
Okada H, Inoue Y, Ogawa Y. Prolonged release microcapsules. US Patent No. 1997;5:643,607.
Gelatin handbook [Internet]. Vol. 2015. Gelatin Manufacturers Institute of America; 2012. Available from: http://www.gelatin-gmia.com/images/GMIA_Gelatin_Manual_2012.pdf
Ogawa Y, Yamamoto M, Takada S, Okada H, Shimamoto T. Controlled-release of leuprolide acetate from polylactic acid or copoly(lactic/glycolic) acid microcapsules: influence of molecular weight and copolymer ratio of polymer. Chem Pharm Bull. 1988;36(4):1502–7.
Sophocleous AM, Desai K-GHG, Mazzara JM, Tong L, Cheng JX, Olsen KF, et al. The nature of peptide interactions with acid end-group PLGAs and facile aqueous-based microencapsulation of therapeutic peptides. J Control Release. 2013;172(3):662–70.
Hirota K, Doty AC, Ackermann R, Zhou J, Olsen KF, Feng MR, et al. Characterizing release mechanisms of leuprolide acetate-loaded PLGA microspheres for IVIVC development I: In vitro evaluation. J Control Release. 2016;244(Pt B):302–13.
Rundlof T, Mathiasson M, Bekiroglu S, Hakkarainen B, Bowden T, Arvidsson T. Survey and qualification of internal standards for quantification by 1H NMR spectroscopy. J Pharm Biomed Anal. 2010;52(5):645–51.
Zhang Y, Sophocleous AM, Schwendeman SP. Inhibition of peptide acylation in PLGA microspheres with water-soluble divalent cationic salts. Pharm Res. 2009;26(8):1986–94.
Rolling-ball viscometer Lovis 2000 M/ME product website [internet]. Anton Paar; Available from: http://www.anton-paar.com/us-en/products/details/rolling-ball-viscometer-lovis-2000-mme/.
Zheng S, Smith P, Burton L, Adams ML. Sensitive fluorescence-based method for the rapid determination of polysorbate-80 content in therapeutic monoclonal antibody products. Pharm Dev Technol 2014;1–5.
Bailey BA, Desai KGH, Ochyl LJ, Ciotti SM, Moon JJ, Schwendeman SP. Self-encapsulating poly(lactic-co-glycolic acid) (PLGA) microspheres for intranasal vaccine delivery. Mol Pharm. 2017;14(9):3228–37.
Ding AG, Shenderova A, Schwendeman SP. Prediction of microclimate pH in poly(lactic-co-glycolic acid) films. J Am Chem Soc. 2006;128(16):5384–90.
Okada H, Inoue Y, Heya T, Ueno H, Ogawa Y, Toguchi H. Pharmacokinetics of once-a-month injectable microspheres of leuprolide acetate. Pharm Res. 1991;8(6):787–91.
Jain RA. The manufacturing techniques of various drug loaded biodegradable poly(lactide-co-glycolide) (PLGA) devices. Biomaterials. 2000;21(23):2475–90.
Schrier JA, DeLuca PP. Recombinant human bone morphogenetic protein-2 binding and incorporation in PLGA microsphere delivery systems. Pharm Dev Technol. 1999;4(4):611–21.
Dawson RMC, Elliott DC, Elliott WH, Jones KM. Data for biochemical research. New York: Oxford University Press; 1989. p. 592.
Harris ELV, Angal S. Protein purification methods: a practical approach (the practical approach series). New York: Oxford University Press; 1990. p. 179.
Okada H, Doken Y, Ogawa Y, Toguchi H. Preparation of three-month depot injectable microspheres of leuprorelin acetate using biodegradable polymers. Pharm Res. 1994;11(8):1143–7.
Ogawa Y, Okada H, Yamamoto M, Shimamoto T. In vivo release profiles of leuprolide acetate from microcapsules prepared with polylactic acids or copoly(lactic/glycolic) acids and in vivo degradation of these polymers. Chem Pharm Bull. 1988;36(7):2576–81.
Acknowledgements
We gratefully thank Jason Romberg from Anton Paar USA Inc. who helped with the determination of viscosity of the injection diluent.
Funding
This research was funded by FDA contract HHSF223201510170C A0001 BAA. This paper reflects the views of the authors and should not be construed to represent FDA’s views or policies.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
ESM 1
(DOCX 21 kb)
Rights and permissions
About this article
Cite this article
Zhou, J., Hirota, K., Ackermann, R. et al. Reverse Engineering the 1-Month Lupron Depot®. AAPS J 20, 105 (2018). https://doi.org/10.1208/s12248-018-0253-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12248-018-0253-2