ABSTRACT
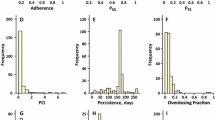
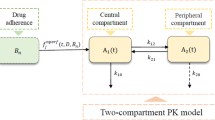
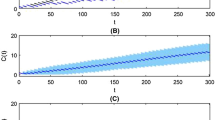
Lack of adherence is a frequent cause of hospitalizations, but its effects on dosing patterns have not been extensively investigated. The purpose of this work was to critically evaluate a novel pharmacometric model for deriving the relationships of adherence to dosing patterns and the dosing interval distribution. The hybrid, stochastic model combines a Markov chain process with the von Mises distribution. The model was challenged with electronic medication monitoring data from 207 hypertension patients and against 5-year persistence data. The model estimates distributions of dosing runs, drug holidays, and dosing intervals. Drug holidays, which can vary between individuals with the same adherence, were characterized by the patient cooperativity index parameter. The drug holiday and dosing run distributions deviate markedly from normality. The dosing interval distribution exhibits complex patterns of multimodality and can be long-tailed. Dosing patterns are an important but under recognized covariate for explaining within-individual variance in drug concentrations.






Similar content being viewed by others
REFERENCES
Paes AH, Bakker A, Soe-Agnie CJ. Impact of dosage frequency on patient compliance. Diabetes Care. 1997;20(10):1512–7.
Urquhart J, De Klerk E. Contending paradigms for the interpretation of data on patient compliance with therapeutic drug regimens. Stat Med. 1998;17(3):251–67. discussion 387–259.
Urquhart J. Ascertaining how much compliance is enough with outpatient antibiotic regimens. Postgrad Med J. 1992;68 Suppl 3:S49–58. discussion S59.
Catalan VS, LeLorier J. Predictors of long-term persistence on statins in a subsidized clinical population. Value Health J Int Soc Pharmacoecono Outcomes Res. 2000;3(6):417–26.
Larsen J, Andersen M, Kragstrup J, Gram LF. High persistence of statin use in a Danish population: compliance study 1993–1998. Br J Clin Pharmacol. 2002;53(4):375–8.
Hughes DA, Walley T. Predicting “real world” effectiveness by integrating adherence with pharmacodynamic modeling. Clin Pharmacol Ther. 2003;74(1):1–8.
Circular Statistics (version 0.4–7) (computer program). Venice, Italy, San Luis Obispo, CA, USA: Department of Environmental Sciences, Informatics and Statistics, Foscari University, Venice, Italy; 2013.
R: A language and environment for statistical computing (computer program). Vienna, Austria: R Foundation for Statistical Computing; 2014.
Rajarshi MB. Markov chains and their extensions. Statistical inference for discrete time stochastic processes. New York, Springer; 2012:19–38.
Feller W. An introduction to probability theory and its applications. 3rd ed. New York: Wiley; 1968.
Uspensky JV. Introduction to mathematical probability. 1st ed. New York: McGraw-Hill Book Company; 1937.
Fu JC, Koutras MV. Distribution theory of runs—a Markov-chain approach. J Am Stat Assoc. 1994;89(41):1050.
Koutras MV, Alexandrou VA. Runs, scans and urn model distributions: a unified Markov chain approach. Ann Inst Stat Math. 1995;47(19):743.
Johnson NL. Poisson distribution. In: Balding DJ, Fisher NI, Johnstone IM, Kadane JB, Molenberghs G, Ryan LM, Scott DW, Smith AFM, Teugels JL, editors. Univariate discrete distributions. 3rd ed. United States: Wiley; 2005. p. 156–207.
Levy G. A pharmacokinetic perspective on medicament noncompliance. Clin Pharmacol Ther. 1993;54(3):242–4.
Gallivan S, Pagel C, Utley M, Franklin BD, Taxis K, Barber N. A technical note concerning non-adherence to drug therapy: exact expressions for the mean and variance of drug concentration. Health Care Manag Sci. 2008;11(3):296–301.
Boissel JP, Nony P. Using pharmacokinetic–pharmacodynamic relationships to predict the effect of poor compliance. Clin Pharmacokinet. 2002;41(1):1–6.
Kiwuwa-Muyingo S, Oja H, Walker SA, Ilmonen P, Levin J, Todd J. Clustering based on adherence data. Epidemiol Perspect Innov EP+I. 2011;8:3.
Girard P, Sheiner LB, Kastrissios H, Blaschke TF. Do we need full compliance data for population pharmacokinetic analysis? J Pharmacokinet Biopharm. 1996;24(3):265–82.
Girard P, Blaschke TF, Kastrissios H, Sheiner LB. A Markov mixed effect regression model for drug compliance. Stat Med. 1998;17(20):2313–33.
Wong D, Modi R, Ramanathan M. Assessment of Markov-dependent stochastic models for drug administration compliance. Clin Pharmacokinet. 2003;42(2):193–204.
Urquhart J. Role of patient compliance in clinical pharmacokinetics. A review of recent research. Clin Pharmacokinet. 1994;27(3):202–15.
Urquhart J. Patient non-compliance with drug regimens: measurement, clinical correlates, economic impact. Eur Heart J. 1996;17(Suppl A(14)):8–15.
Olivieri NF, Matsui D, Hermann C, Koren G. Compliance assessed by the Medication Event Monitoring System. Arch Dis Child. 1991;66(12):1399–402.
Arnet I, Haefeli WE. Overconsumption detected by electronic drug monitoring requires subtle interpretation. Clin Pharmacol Ther. 2000;67(1):44–7.
ACKNOWLEDGMENTS
Support from the National Multiple Sclerosis Society (RG4836-A-5) to the Ramanathan laboratory is gratefully acknowledged.
Financial Conflicts
See disclosure statement.
Confidentiality
Use of the information in this manuscript for commercial, non-commercial, research or purposes other than peer review not permitted prior to publication without expressed written permission of the author.
Author Contributions
Kelly Fellows – Conducted experiments, data analysis, manuscript preparation.
Sheril Alexander, Alyssa Droopad, Jenna Covelli, Vivian Rodriguez-Cruz – Data acquisition.
Murali Ramanathan – Study concept and design, data analysis, manuscript preparation.
Disclosure
Dr. Murali Ramanathan received research funding from the National Multiple Sclerosis Society and the Department of Defense. He received compensation for serving as an Editor from the American Association of Pharmaceutical Scientists. These are unrelated to the research presented in this report.
Kelly Fellows, Sheril Alexander, Alyssa Droopad, Jenna Covelli and Vivian Rodriguez-Cruz have no conflicts to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
(GIF 54 kb)
High Resolution Image
(TIFF 1229 kb)
ESM 1
(DOCX 126 kb)
Rights and permissions
About this article
Cite this article
Fellows, K., Rodriguez-Cruz, V., Covelli, J. et al. A Hybrid Markov Chain–von Mises Density Model for the Drug-Dosing Interval and Drug Holiday Distributions. AAPS J 17, 427–437 (2015). https://doi.org/10.1208/s12248-014-9713-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-014-9713-5