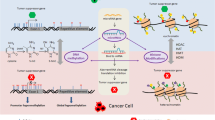
Abstract
In recent years, “nutri-epigenetics,” which focuses on the influence of dietary agents on epigenetic mechanism(s), has emerged as an exciting novel area in epigenetics research. Targeting of aberrant epigenetic modifications has gained considerable attention in cancer chemoprevention research because, unlike genetic changes, epigenetic alterations are reversible and occur during early carcinogenesis. Aberrant epigenetic mechanisms, such as promoter DNA methylation, histone modifications, and miRNA-mediated post-transcriptional alterations, can silence critical tumor suppressor genes, such as transcription factors, cell cycle regulators, nuclear receptors, signal transducers, and apoptosis-inducing and DNA repair gene products, and ultimately contribute to carcinogenesis. In an effort to identify and develop anticancer agents which cause minimal harm to normal cells while effectively killing cancer cells, a number of naturally occurring phytochemicals in food and medicinal plants have been investigated. This review highlights the potential role of plant-derived phytochemicals in targeting epigenetic alterations that occur during carcinogenesis, by modulating the activity or expression of DNA methyltransferases, histone modifying enzymes, and miRNAs. We present in detail the epigenetic mode of action of various phytochemicals and discuss their potential as safe and clinically useful chemopreventive strategies.

Similar content being viewed by others
Abbreviations
- Akt:
-
v-akt murine thymoma viral oncogene homolog 1
- AM:
-
Allyl mercaptan
- AP-1:
-
Activator Protein-1
- AR:
-
Androgen receptor
- Bax:
-
BCL2-associated X protein
- Bcl2:
-
B-cell CLL/lymphoma 2
- Bcl-xL:
-
B-cell lymphoma-extra large
- Bmi-1:
-
B-cell-specific Moloney murine leukemia virus integration site 1
- BRCA1:
-
Breast cancer 1, early onset
- CBP:
-
CREB-binding protein
- CCND2:
-
Cyclin D2
- Cdc25A:
-
Cell division cycle 25 homolog A
- Cdk:
-
Cyclin-dependent kinase
- c-Kit:
-
v-kit Hardy-Zuckerman 4 feline sarcoma viral oncogene homolog
- COMT:
-
Catechol-O-methyltransferase
- COX-2:
-
Cyclooxygenase-2
- CYLD:
-
Cylindromatosis (turban tumor syndrome)
- DADS:
-
Diallyl disulfide
- DAS:
-
Diallyl sulfide
- DATS:
-
Diallyl trisulfide
- DHFR:
-
Dihydrofolate reductase
- DMBA:
-
7,12-dimethylbenz(a)anthracene
- DNMT:
-
DNA methyltransferase
- DNMT-3L:
-
DNA (cytosine-5)-methyltransferase 3-like
- E2F:
-
E2F transcription factor
- EC:
-
[−]-epicatechin
- ECG:
-
[−]-epicatechin-3-gallate
- EGC:
-
[−]-epigallocatechin
- EGCG:
-
[−]-epigallocatechin-3-gallate
- EGFR:
-
Epidermal growth factor receptor
- ER:
-
Estrogen receptor
- ERβ:
-
Estrogen receptor beta
- ERBB2:
-
Human epidermal growth factor receptor 2
- ERα:
-
Estrogen receptor alpha
- EZH-2:
-
Enhancer of zeste homolog 2
- FOXO3a:
-
Forkhead box protein O3
- GCN5:
-
SAGA complex histone acetyltransferase catalytic subunit Gcn5
- GSTP1:
-
Glutathione-S-transferase pi 1
- HATs:
-
Histone acetyl transferases
- HDACs:
-
Histone deacetylases
- HER-2:
-
Human epidermal growth factor receptor 2
- HIF-1 α:
-
Hypoxia inducible factor 1, alpha subunit
- HKMTs:
-
Histone lysine methyltransferases
- hMLH1:
-
Human mutL homolog 1
- HOX family proteins:
-
Homeobox family proteins
- HSP90:
-
Heat shock protein 90
- hTERT:
-
Human telomerase reverse transcriptase
- IP-10:
-
TNF-induced interferon-gamma-inducible protein 10
- K:
-
Lysine
- LEF:
-
Lymphoid enhancer factor
- MBD:
-
Methylated DNA binding domain proteins
- MCL1:
-
Induced myeloid leukemia cell differentiation protein Mcl-1
- MCM-2:
-
Minichromosome maintenance gene
- MGMT-O(6):
-
Methylguanine-DNA methyltransferase
- MIP-2:
-
Macrophage inflammatory protein 2
- miRNA:
-
MicroRNA
- MTA-2:
-
Metastasis associated 1 family member 2
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- Notch1:
-
Notch homolog 1, translocation-associated (Drosophila)
- OSCs:
-
Organosulfur compounds
- p16INK4a:
-
Cyclin-dependent kinase 4 inhibitor A
- p21waf1/cip1:
-
Cyclin-dependent kinase inhibitor 1A
- p53:
-
Tumor protein 53
- PARP:
-
Poly ADP-ribose polymerase
- PCAF:
-
K(lysine) acetyltransferase 2B
- PcG:
-
Polycomb group proteins
- PDCD4:
-
Programmed cell death 4
- PEITC:
-
Phenethyl isothiocyanate
- PRPS1:
-
Phosphoribosyl pyrophosphate synthetase 1
- PTEN:
-
Phosphatase and tensin homolog deleted on chromosome 10
- RARβ2:
-
Retinoic acid receptor, beta 2
- R:
-
Arginine
- RAS:
-
Rat sarcoma transforming oncogene
- RASSF1A:
-
RAS association domain family 1A
- RXR alpha:
-
Retinoid X receptor, alpha
- SAH:
-
S-adenosyl-L-homocysteine
- SAM:
-
S-adenosyl methionine
- SAMC:
-
S-allylmercaptocysteine
- SIRT1:
-
Sirtuin (silent mating type information regulation 2 homolog) 1
- SLC16A1:
-
Solute carrier family 16, member 1
- SNX19:
-
Sorting nexin-19
- Sp1:
-
Transcription Factor Sp1
- TGFBR2:
-
Transforming growth factor, beta receptor II
- TGF-β:
-
Transforming growth factor, beta
- TTK:
-
Phosphotyrosine picked threonine-protein kinase
- VEGF:
-
Vascular endothelial cell growth factor
- ZBTB10:
-
Zinc finger and BTB domain containing 10
- ZEB1:
-
Zinc finger E-box binding homeobox 1
- ZNF513:
-
Zinc finger protein 513
REFERENCES
Henikoff S, Matzke MA. Exploring and explaining epigenetic effects. Trends Genet. 1997;13:293–5.
Dehan P, Kustermans G, Guenin S, Horion J, Boniver J, Delvenne P. DNA methylation and cancer diagnosis: new methods and applications. Expert Rev Mol Diagn. 2009;9:651–7.
Illingworth R, Kerr A, Desousa D, Jorgensen H, Ellis P, Stalker J, et al. A novel CpG island set identifies tissue-specific methylation at developmental gene loci. PLoS Biol. 2008;6:e22.
Suter MA, Aagaard-Tillery KM. Environmental influences on epigenetic profiles. Semin Reprod Med. 2009;27:380–90.
Jones PA, Baylin SB. The epigenomics of cancer. Cell. 2007;128:683–92.
Issa JP, Kantarjian HM. Targeting DNA methylation. Clin Cancer Res. 2009;15:3938–46.
Esteller M. Cancer epigenomics: DNA methylomes and histone-modification maps. Nat Rev Genet. 2007;8:286–98.
Kopelovich L, Crowell JA, Fay JR. The epigenome as a target for cancer chemoprevention. J Natl Cancer Inst. 2003;95:1747–57.
Gama-Sosa MA, Slagel VA, Trewyn RW, Oxenhandler R, Kuo KC, Gehrke CW, et al. The 5-methylcytosine content of DNA from human tumors. Nucleic Acids Res. 1983;11:6883–94.
Goelz SE, Vogelstein B, Hamilton SR, Feinberg AP. Hypomethylation of DNA from benign and malignant human colon neoplasms. Science. 1985;228:187–90.
Fullgrabe J, Kavanagh E, Joseph B. Histone onco-modifications. Oncogene. 2011;30:3391–403.
Mottet D, Castronovo V. Histone deacetylases: target enzymes for cancer therapy. Clin Exp Metastasis. 2008;25:183–9.
Sauve AA, Wolberger C, Schramm VL, Boeke JD. The biochemistry of sirtuins. Annu Rev Biochem. 2006;75:435–65.
Bannister AJ, Kouzarides T. Regulation of chromatin by histone modifications. Cell Res. 2011;21:381–95.
Spange S, Wagner T, Heinzel T, Kramer OH. Acetylation of non-histone proteins modulates cellular signalling at multiple levels. Int J Biochem Cell Biol. 2009;41:185–98.
Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6:857–66.
Winter J, Jung S, Keller S, Gregory RI, Diederichs S. Many roads to maturity: microRNA biogenesis pathways and their regulation. Nat Cell Biol. 2009;11:228–34.
Brait M, Sidransky D. Cancer epigenetics: above and beyond. Toxicol Mech Methods. 2011;21:275–88.
Hardy TM, Tollefsbol TO. Epigenetic diet: impact on the epigenome and cancer. Epigenomics. 2011;3:503–18.
Siddiqui IA, Adhami VM, Saleem M, Mukhtar H. Beneficial effects of tea and its polyphenols against prostate cancer. Mol Nutr Food Res. 2006;50:130–43.
Lee WJ, Shim JY, Zhu BT. Mechanisms for the inhibition of DNA methyltransferases by tea catechins and bioflavonoids. Mol Pharmacol. 2005;68:1018–30.
Fang MZ, Wang Y, Ai N, Hou Z, Sun Y, Lu H, et al. Tea polyphenol (-)-epigallocatechin-3- gallate inhibits DNA methyltransferase and reactivates methylation silenced genes in cancer cell lines. Cancer Res. 2003;63:7563–70.
Pandey M, Shukla S, Gupta S. Promoter demethylation and chromatin remodeling by green tea polyphenols leads to re-expression of GSTP1 in human prostate cancer cells. Int J Cancer. 2010;126:2520–33.
Nandakumar V, Vaid M, Katiyar SK. (-)-Epigallocatechin-3-gallate reactivates silenced tumor suppressor genes, Cip1/p21 and p16INK4a, by reducing DNA methylation and increasing histones acetylation in human skin cancer cells. Carcinogenesis. 2011;32:537–44.
Berletch JB, Liu C, Love WK, Andrews LG, Katiyar SK, Tollefsbol TO. Epigenetic and genetic mechanisms contribute to telomerase inhibition by EGCG. J Cell Biochem. 2008;103:509–19.
Meeran SM, Patel SN, Chan TH, Tollefsbol TO. A novel prodrug of epigallocatechin-3-gallate: differential epigenetic hTERT repression in human breast cancer cells. Cancer Prev Res (Phila). 2001;4:1243–54.
Volate SR, Muga SJ, Issa AY, Nitcheva D, Smith T, Wargovich MJ. Epigenetic modulation of the retinoid X receptor alpha by green tea in the azoxymethane-Apc Min/+ mouse model of intestinal cancer. Mol Carcinog. 2009;48:920–33.
Thakur VS, Gupta K, Gupta S. Green tea polyphenols causes cell cycle arrest and apoptosis in prostate cancer cells by suppressing class I histone deacetylases. Carcinogenesis. 2012;33:377–84.
Thakur VS, Gupta K, Gupta S. Green tea polyphenols increase p53 transcriptional activity and acetylation by suppressing class I histone deacetylases. Int J Oncol. 2012;41:353–61.
Balasubramanian S, Adhikary G, Eckert RL. The Bmi-1 polycomb protein antagonizes the (-)-epigallocatechin-3-gallate-dependent suppression of skin cancer cell survival. Carcinogenesis. 2010;31:496–503.
Tsang WP, Kwok TT. Epigallocatechin gallate up-regulation of miR-16 and induction of apoptosis in human cancer cells. J Nutr Biochem. 2010;21:140–6.
Fix LN, Shah M, Efferth T, Farwell MA, Zhang B. MicroRNA expression profile of MCF-7 human breast cancer cells and the effect of green tea polyphenon-60. Cancer Genomics Proteomics. 2010;7:261–77.
Kunnumakkara AB, Anand P, Aggarwal BB. Curcumin inhibits proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signaling proteins. Cancer Lett. 2008;269:199–225.
Liu Z, Xie Z, Jones W, Pavlovicz RE, Liu S, Yu J, et al. Curcumin is a potent DNA hypomethylation agent. Bioorg Med Chem Lett. 2009;19:706–9.
Khor TO, Huang Y, Wu TY, Shu L, Lee J, Kong AN. Pharmacodynamics of curcumin as DNA hypomethylation agent in restoring the expression of Nrf2 via promoter CpGs demethylation. Biochem Pharmacol. 2011;82:1073–8.
Shu L, Khor TO, Lee JH, Boyanapalli SS, Huang Y, Wu TY et al. Epigenetic CpG demethylation of the promoter and reactivation of the expression of neurog1 by curcumin in prostate LNCaP cells. AAPS J. 2011:606–14.
Jha AK, Nikbakht M, Parashar G, Shrivastava A, Capalash N, Kaur J. Reversal of hypermethylation and reactivation of the RARbeta2 gene by natural compounds in cervical cancer cell lines. Folia Biol (Praha). 2010;56:195–200.
Marcu MG, Jung YJ, Lee S, Chung EJ, Lee MJ, Trepel J, et al. Curcumin is an inhibitor of p300 histone acetylatransferase. Med Chem. 2006;2:169–74.
Balasubramanyam K, Varier RA, Altaf M, Swaminathan V, Siddappa NB, Ranga U, et al. Curcumin, a novel p300/CREB-binding protein-specific inhibitor of acetyltransferase, represses the acetylation of histone/nonhistone proteins and histone acetyltransferase dependent chromatin transcription. J Biol Chem. 2004;279:51163–71.
Kang J, Chen J, Shi Y, Jia J, Zhang Y. Curcumin-induced histone hypoacetylation: the role of reactive oxygen species. Biochem Pharmacol. 2005;69:1205–13.
Chen Y, Shu W, Chen W, Wu Q, Liu H, Cui G. Curcumin, both histone deacetylase and p300/CBP-specific inhibitor, represses the activity of nuclear factor kappa B and Notch-1 in Raji cells. Basic Clin Pharmacol Toxicol. 2007;101:427–33.
Liu HL, Chen Y, Cui GH, Zhou JF. Curcumin, a potent anti-tumor reagent, is a novel histone deacetylase inhibitor regulating B-NHL cell line Raji proliferation. Acta Pharmacol Sin. 2005;26(5):603–9.
Bora-Tatar G, Dayangac-Erden D, Demir AS, Dalkara S, Yelekci K, Erdem-Yurter H. Molecular modifications on carboxylic acid derivatives as potent histone deacetylase inhibitors: activity and docking studies. Bioorg Med Chem. 2009;17:5219–28.
Lee SJ, Krauthauser C, Maduskuie V, Fawcett PT, Olson JM, Rajasekaran SA. Curcumin-induced HDAC inhibition and attenuation of medulloblastoma growth in vitro and in vivo. BMC Cancer. 2011;11:144. doi:10.1186/1471-2407-11-144.
Hua WF, Fu YS, Liao YJ, Xia WJ, Chen YC, Zeng YX, et al. Curcumin induces down- regulation of EZH2 expression through the MAPK pathway in MDA-MB-435 human breast cancer cells. Eur J Pharmacol. 2010;637:16–21.
Sun M, Estrov Z, Ji Y, Coombes KR, Harris DH, Kurzrock R. Curcumin (diferuloylmethane) alters the expression profiles of microRNAs in human pancreatic cancer cells. Mol Cancer Ther. 2008;7:464–73.
Ali S, Ahmad A, Banerjee S, Padhye S, Dominiak K, Schaffert JM, et al. Gemcitabine sensitivity can be induced in pancreatic cancer cells through modulation of miR-200 and miR-21 expression by curcumin or its analogue CDF. Cancer Res. 2010;70:3606–17.
Zhang J, Zhang T, Ti X, Shi J, Wu C, Ren X, et al. Curcumin promotes apoptosis in A549/DDP multidrugresistant human lung adenocarcinoma cells through an miRNA signaling pathway. Biochem Biophys Res Commun. 2010;399:1–6.
Yang J, Cao Y, Sun J, Zhang Y. Curcumin reduces the expression of Bcl-2 by upregulating miR-15a and miR-16 in MCF-7 cells. Med Oncol. 2010;27:1114–8.
Mudduluru G, George-William JN, Muppala S, Asangani IA, Regalla K, Nelson LD, et al. Curcumin regulates miR-21 expression and inhibits invasion and metastasis in colorectal cancer. Biosci Rep. 2011;31:185–97.
Clarke JD, Dashwood RH, Ho E. Multi-targeted prevention of cancer by sulforaphane. Cancer Lett. 2008;269:291–304.
Traka M, Gasper AV, Smith JA, Hawkey CJ, Bao Y, Mithen RF. Transcriptome analysis of human colon caco-2 cells exposed to sulforaphane. J Nutr. 2005;135:1865–72.
Meeran SM, Patel SN, Tollefsbol TO. Sulforaphane causes epigenetic repression of hTERT expression in human breast cancer cell lines. PLoS One. 2010;5(7):e11457.
Myzak MC, Karplus PA, Chung FL, Dashwood RH. A novel mechanism of chemoprotection by sulforaphane: inhibition of histone deacetylase. Cancer Res. 2004;64:5767–74.
Myzak MC, Hardin K, Wang R, Dashwood RH, Ho E. Sulforaphane inhibits histone deacetylase activity in BPH-1, LnCaP and PC-3 prostate epithelial cells. Carcinogenesis. 2006;27:811–9.
Pledgie-Tracy A, Sobolewski MD, Davidson NE. Sulforaphane induces cell type-specific apoptosis in human breast cancer cell lines. Mol Cancer Ther. 2007;6:1013–21.
Myzak MC, Dashwood WM, Orner GA, Ho E, Dashwood RH. Sulforaphane inhibits histone deacetylase in vivo and suppresses tumorigenesis in Apc-minus mice. FASEB J. 2006;20:506–8.
Myzak MC, Tong P, Dashwood WM, Dashwood RH, Ho E. Sulforaphane retards the growth of human PC-3 xenografts and inhibits HDAC activity in human subjects. Exp Biol Med (Maywood). 2007;232:227–34.
Banerjee S, Kong D, Wang Z, Bao B, Hillman GG, Sarkar FH. Attenuation of multi-targeted proliferation-linked signaling by 3,30-diindolylmethane (DIM): from bench to clinic. Mutat Res. 2011;728:47–66.
Li Y, Li X, Guo B. Chemopreventive agent 3,30-diindolylmethane selectively induces proteasomal degradation of class I histone deacetylases. Cancer Res. 2010;70:646–54.
Li Y, Vandenboom 2nd TG, Kong D, Wang Z, Ali S, Philip PA, et al. Up-regulation of miR- 200 and let-7 by natural agents leads to the reversal of epithelial-to-mesenchymal transition in gemcitabine-resistant pancreatic cancer cells. Cancer Res. 2009;69:6704–12.
Li Y, Vandenboom TG, Wang Z, Ali S, Philip PA, Sarkar FH. miR-146a suppresses invasion of pancreatic cancer cells. Cancer Res. 2010;70:1486–95.
Jin Y, Zou X, Feng X. 3,30-Diindolylmethane negatively regulates Cdc25A and induces a G2/M arrest by modulation of microRNA 21 in human breast cancer cells. Anticancer Drugs. 2010;21:814–22.
Banerjee S, Li Y, Wang Z, Sarkar FH. Multi-targeted therapy of cancer by genistein. Cancer Lett. 2008;269:226–42.
Fang MZ, Chen D, Sun Y, Jin Z. Reversal of hypermethylation and reactivation of p16INK4a, RARbeta, and MGMT genes by genistein and other isoflavones from soy. Clin Cancer Res. 2005;11(19 Pt 1):7033–41.
King-Batoon A, Leszczynska JM, Klein CB. Modulation of gene methylation by genistein or lycopene in breast cancer cells. Environ Mol Mutagen. 2008;49:36–45.
Vardi A, Bosviel R, Rabiau N, Adjakly M, Satih S, Dechelotte P, et al. Soy phytoestrogens modify DNA methylation of GSTP1, RASSF1A, EPH2 and BRCA1 promoter in prostate cancer cells. In Vivo. 2010;24:393–400.
Adjakly M, Bosviel R, Rabiau N, Boiteux JP, Bignon YJ, Guy L, et al. DNA methylation and soy phytoestrogens: quantitative study in DU-145 and PC-3 human prostate cancer cell lines. Epigenomics. 2011;3:795–803.
Wang Z, Chen H. Genistein increases gene expression by demethylation of WNT5a promoter in colon cancer cell line SW1116. Anticancer Res. 2010;30:4537–45.
Majid S, Dar AA, Shahryari V, Hirata H, Ahmad A, Saini S, et al. Genistein reverses hypermethylation and induces active histone modifications in tumor suppressor gene B-cell translocation gene 3 in prostate cancer. Cancer. 2010;116:66–76.
Qin W, Zhu W, Shi H, Hewett JE, Ruhlen RL, Macdonald RS, et al. Soy isoflavones have an antiestrogenic effect and alter mammary promoter hypermethylation in healthy premenopausal women. Nutr Cancer. 2009;61:238–44.
Majid S, Kikuno N, Nelles J, Noonan E, Tanaka Y, Kawamoto K, et al. Genistein induces the p21WAF1/CIP1 and p16INK4a tumor suppressor genes in prostate cancer cells by epigenetic mechanisms involving active chromatin modification. Cancer Res. 2008;68:2736–44.
Hong T, Nakagawa T, Pan W, Kim MY, Kraus WL, Ikehara T, et al. Isoflavones stimulate estrogen receptor-mediated core histone acetylation. Biochem Biophys Res Commun. 2004;317:259–64.
Kikuno N, Shiina H, Urakami S, Kawamoto K, Hirata H, Tanaka Y, et al. Genistein mediated histone acetylation and demethylation activates tumor suppressor genes in prostate cancer cells. Int J Cancer. 2008;123:552–60.
Basak S, Pookot D, Noonan EJ, Dahiya R. Genistein down-regulates androgen receptor by modulating HDAC6-Hsp90 chaperone function. Mol Cancer Ther. 2008;7:3195–202.
Parker LP, Taylor DD, Kesterson J, Metzinger DS, Gercel-Taylor C. Modulation of microRNA associated with ovarian cancer cells by genistein. Eur J Gynaecol Oncol. 2009;30:616–21.
Majid S, Dar AA, Saini S, Chen Y, Shahryari V, Liu J, et al. Regulation of minichromosome maintenance gene family by microRNA-1296 and genistein in prostate cancer. Cancer Res. 2010;70:2809–18.
Sun Q, Cong R, Yan H, Gu H, Zeng Y, Liu N, et al. Genistein inhibits growth of human uveal melanoma cells and affects microRNA-27a and target gene expression. Oncol Rep. 2009;22:563–7.
Cheung KL, Kong AN. Molecular targets of dietary phenethyl isothiocyanate and sulforaphane for cancer chemoprevention. AAPS J. 2010;12:87–97.
Wang LG, Beklemisheva A, Liu XM, Ferrari AC, Feng J, Chiao JW. Dual action on promoter demethylation and chromatin by an isothiocyanate restored GSTP1 silenced in prostate cancer. Mol Carcinog. 2007;46:24–31.
Lea MA, Randolph VM, Lee JE, des Bordes C. Induction of histone acetylation in mouse erythroleukemia cells by some organosulfur compounds including allyl isothiocyanate. Int J Cancer. 2001;92:784–9.
Xiao L, Huang Y, Zhen R, Chiao JW, Liu D, Ma X. Deficient histone acetylation in acute leukemia and the correction by an isothiocyanate. Acta Haematol. 2010;123:71–6.
Huang YQ, Ma XD, Lai YD, Wang XZ, Chiao JW, Liu DL. Phenylhexyl isothiocyanate (PHI) regulates histone methylation and acetylation and induces apoptosis in SMMC-7721 cells. Zhonghua Gan Zang Bing Za Zhi. 2010;18:209–12.
Izzotti A, Calin GA, Steele VE, Cartiglia C, Longobardi M, Croce CM, et al. Chemoprevention of cigarette smoke-induced alterations of microRNA expression in rat lungs. Cancer Prev Res (Phila). 2010;3:62–72.
Izzotti A, Larghero P, Cartiglia C, Longobardi M, Pfeffer U, Steele VE, et al. Modulation of microRNA expression by budesonide, phenethyl isothiocyanate and cigarette smoke in mouse liver and lung. Carcinogenesis. 2010;31:894–901.
Savouret JF, Quesne M. Resveratrol and cancer: a review. Biomed Pharmacother. 2002;56:84–7.
Stefanska B, Rudnicka K, Bednarek A, Fabianowska-Majewska K. Hypomethylation and induction of retinoic acid receptor beta 2 by concurrent action of adenosine analogues and natural compounds in breast cancer cells. Eur J Pharmacol. 2010;638:47–53.
Tili E, Michaille JJ, Alder H, Volinia S, Delmas D, Latruffe N, et al. Resveratrol modulates the levels of microRNAs targeting genes encoding tumor-suppressors and effectors of TGF signaling pathway in SW480 cells. Biochem Pharmacol. 2010;80:2057–65.
Wang RH, Zheng Y, Kim HS, Xu X, Cao L, Luhasen T, et al. Interplay among BRCA1, SIRT1, and surviving during BRCA1-associated tumorigenesis. Mol Cell. 2008;32:11–20.
Papoutsis AJ, Lamore SD, Wondrak GT, Selmin OI, Romagnolo DF. Resveratrol prevents epigenetic silencing of BRCA-1 by the aromatic hydrocarbon receptor in human breast cancer cells. J Nutr. 2010;140:1607–14.
Ariga T, Seki T. Antithrombotic and anticancer effects of garlic-derived sulfur compounds: a review. Biofactors. 2006;26:93–103.
Lea MA, Randolph VM, Patel M. Increased acetylation of histones induced by diallyl disulfide and structurally related molecules. Int J Oncol. 1999;15:347–52.
Druesne N, Pagniez A, Mayeur C, Thomas M, Cherbuy C, Duee PH, et al. Diallyl disulfide (DADS) increases histone acetylation and p21(waf1/cip1) expression in human colon tumor cell lines. Carcinogenesis. 2004;25:1227–36.
Giovannucci E. Tomatoes, tomato-based products, lycopene, and cancer: review of the epidemiologic literature. J Natl Cancer Inst. 1999;91:317–31.
Gibellini L, Pinti M, Nasi M, Montagna JP, De Biasi S, Roat E, et al. Quercetin and cancer chemoprevention. Evid Based Complement Alternat Med. 2011;2011:591356.
Tan S, Wang C, Lu C, Zhao B, Cui Y, Shi X, et al. Quercetin is able to demethylate the p16INK4a gene promoter. Chemotherapy. 2009;55:6–10.
Ruiz PA, Braune A, Holzlwimmer G, Quintanilla-Fend L, Haller D. Quercetin inhibits TNF- induced NF-kappaB transcription factor recruitment to proinflammatory gene promoters in murine intestinal epithelial cells. J Nutr. 2007;137:1208–15.
Lee WJ, Chen YR, Tseng TH. Quercetin induces FasL-related apoptosis, in part, through promotion of histone H3 acetylation in human leukemia HL-60 cells. Oncol Rep. 2011;25:583–91.
Priyadarsini RV, Vinothini G, Murugan RS, Manikandan P, Nagini S, et al. The flavonoid quercetin modulates the hallmark capabilities of hamster buccal pouch tumors. Nutr Cancer. 2011;63:218–26.
Heber D. Multi-targeted therapy of cancer by ellagitannins. Cancer Lett. 2008;269:262–8.
Wen XY, Wu SY, Li ZQ, Liu ZQ, Zhang JJ, Wang GF, et al. Ellagitannin (BJA3121), an anti-proliferative natural polyphenol compound, can regulate the expression of miRNAs in HepG2 cancer cells. Phytother Res. 2009;23:778–84.
Pandey M, Kaur P, Shukla S, Abbas A, Fu P, Gupta S. Plant flavone apigenin inhibits HDAC and remodels chromatin to induce growth arrest and apoptosis in human prostate cancer cells: in vitro and in vivo study. Mol Carcinog. 2012;51:952–62.
ACKNOWLEDGMENTS
The original work from author’s laboratory outlined in this review was supported by United States Public Health Service Grants RO1CA108512, RO1CA115491 and RO1AT002709. We acknowledge Shyama Prasad Mukherjee (SPM) fellowship provided to GD by the Council of Scientific and Industrial Research (CSIR), India and Fulbright-Nehru Doctoral and Professional Research fellowship provided by United States—India Educational Foundation (USIEF) for her work in the United States. MAB is supported by NIH 5T32DK007316 Ruth L. Kirschstein Pre-Doctoral Fellowship through the Metabolism Training Program. We apologize to those investigators whose original work could not be cited owing to the space limitations.
Conflict of Interest
The authors have no competing interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: Ah-Ng Tony Kong and Chi Chen
Rights and permissions
About this article
Cite this article
Thakur, V.S., Deb, G., Babcook, M.A. et al. Plant Phytochemicals as Epigenetic Modulators: Role in Cancer Chemoprevention. AAPS J 16, 151–163 (2014). https://doi.org/10.1208/s12248-013-9548-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-013-9548-5