Abstract
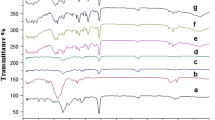
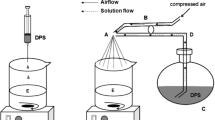
The purpose of this study was to examine the suitability of polystyrene-coated (PS-coated) microcapsules of drug-resin complex for achieving prolonged release of diltiazem-HCl, a highly water-soluble drug, in simulated gastric and intestinal fluid. The drug was bound to Indion 254, a cation-exchange resin, and the resulting resinate was microencapsulated with PS using an oil-in-water emulsion-solvent evaporation method. The effect of various formulation parameters on the characteristics of the microcapsules was studied. Mean diameter and encapsulation efficiency of the microcapsules rose with an increase in the concentration of emulsion stabilizer and the coat/core ratio, while the same characteristics tended to decrease with an increase in the volume of the organic disperse phase. The desorption of drug from the uncoated resinate was quite rapid and independent of the pH of the dissolution media. On the other hand, the drug release from the microcapsules was prolonged for different periods of time depending on the formulation parameters and was also found to be independent of the pH of the dissolution media. Both the encapsulation efficiency and the retardation of drug release were found to be dependent on the uniformity of coating, which in turn was influenced by the formulation parameters. Kinetic studies revealed that the desorption of drug from the resinate obeyed the typical particle diffusion process, whereas the drug release from the microencapsulated resinate followed the diffusion-controlled model in accordance with the Higuchi equation. PS appeared to be a suitable polymer to provide prolonged release of diltiazem independent of the pH of the dissolution media.
Similar content being viewed by others
References
Beckett AH. Alternative routes of drug administration and new drug delivery systems. In: Breimer DD, ed. Towards Better Safety of Drug and Pharmaceutical Products. Amsterdam, The Netherlands: Elsevier North Holland Biomedical Press; 1980:247–263.
Davis SS, Hardy JG, Taylor MJ, Whalley DR, Wilson CG. Comparative study of gastrointestinal transit of a pellet and tablet formulation. Int J Pharm. 1984;21:167–177.
Follonier N, Doelkar E. Biopharmaceutical comparison of oral multiple-unit and single-unit sustained release dosage forms. STP Pharm Sci. 1992;2:141–158.
Beck LR, Cowsar DR, Lewis DH, et al. A new long-acting injectable microcapsule system for the administration of progesterone. Fertil Steril. 1979;31:545–551.
Bodmeier R, McGinity JW. Polylactic acid microspheres containing quinidine base and quinidine sulphate prepared by solvent evaporation technique, II: some process parameters influencing the preparation and properties of microspheres. J Microencapsul. 1987;4:289–297.
Jones DS, Pearce KJ. An investigation of the effects of some process variables on the microencapsulation of propranolol hydrochloride by the solvent evaporation method. Int J Pharm. 1995;118:199–205.
Bodmeier R, McGinity JW. Polylactic acid microspheres containing quinidine base and quinidine sulphate prepared by solvent evaporation technique, I: methods and morphology. J Microencapsul. 1987;4:279–288.
Ogawa Y, Yamamoto M, Takada S, Okada H, Shimamoto T A new technique to efficiently entrap leuprolide acetate into microcapsules of PLA or co-poly(lactic/glycolic) acid microcapsules: influence of molecular weight and copolymer ratio of polymer. Chem Pharm Bull (Tokyo). 1988;36:1502–1505.
Alex R, Bodmeier R. Encapsulation of water soluble drugs by modified solvent evaporation method, I: effect of process and formulation variables on drug entrapment. J Microencapsul. 1990;7:347–355.
Cuna M, Alonso MJ, Torres D. Preparation and in vitro evaluation of mucoadhesive microparticles containing amoxycillin-resin complexes for drug delivery to the gastric mucosa. Eur J Pharm Biopharm. 2001; 51:199–205.
Sriwongjanya M, Bodmeier R. Entrapment of drug-loaded ion-exchange particles within polymeric microparticles. Int J Pharm. 1997;158:29–38.
Torres D, Boado L, Blanko D, Vila-Jato JL. Comparison between aqueous and non-aqueous solvent evaporation methods for micro-encapsulation of drug-resin complexes. Int J Pharm. 1998;173:171–182.
Raghunathan Y, Amsel L, Hinsvark O, Bryant W. Sustained release drug delivery system, I: coated ion-exchange resin system for phenyl-propanolamine and other drugs. J Pharm Sci. 1981;70:379–384.
Sprockel OL, Prapaitrakul W. Effect of eluant properties on drug release from cellulose acetate butyrate-coated drug resin complexes. Int J Pharm. 1988;48:217–222.
Sprockel OL, Price JC. Evaluation of sustained release aqueous suspensions containing microencapsulated drug-resin complexes. Drug Dev Ind Pharm. 1989;15:1275–1287.
Sprockel OL, Price JC. Development of an emulsion-solvent evaporation technique for microencapsulation of drug-resin complex. Drug Dev Ind Pharm. 1990;16:361–376.
Phagocytosis and Microparticles [Technical Data Sheet 430]. Warrington, PA: Polysciences, Inc. Available at: http://www.polysciences com/shop/assets/datasheets/430.pdf. Accessed: November 14, 2004.
Mac A, Negi D, Friend D. Preparation and characterization of poly(styrene) microcapsules containing corrosion inhibitors. J Microencapsul. 1989;6:361–367.
El-Gibaly I, Safwat SM, Ahmed MO. Microencapsulation of ketoprofen using w/o/w complex emulsion technique. J Microencapsul. 1996;13:67–87.
Park IK, Seo SJ, Akashi M, Akaike T, Cho CS. Controlled release of epidermal growth factor (EGF) from EGF-loaded polymeric nanoparticles composed of polystyrene as core and poly(methacrylic acid) as corona in vitro. Arch Pharm Res. 2003;26:649–652.
Mortada SM. Preparation of ethylcellulose microcapsules using the complex emulsification method. Pharmazie. 1982;37:427–429.
Atyabi F, Sharma HL, Mohammad HAH, Fell JT. Controlled drug release from coated floating ion exchange resin. J Control Release. 1996;42:25–28.
Khan KA, Rhodes CT. Effect of compaction pressure on the dissolution efficiency of some direct compression systems. Pharm Acta Helv. 1972;47:594–607.
Tamilvanan S, Sa B. Studies on the in vitro release characteristics of ibuprofen-loaded polystyrene microparticles. J Microencapsul. 2000;17:57–67.
Tamilvanan S, Sa B. Studies on the in vitro release behavior of indomethacin-loaded polystyrene microparticles. Int J Pharm. 2000;201:187–197.
Torres D, Seijo B, Garcia-Encina G, Alonso MeJ, Vila-Jato JL. Microencapsulation of ion-exchange resins by interfacial nylon polymerization. Int J Pharm. 1990;59:9–17.
Reichenberg D. Properties of ion-exchange resins in relation to their structure, III: kinetics of exchange. J Am Chem Soc. 1953;75:589–597.
Bhaskar R, Murthy RSR, Miglani BD, Viswanathan K. Novel method to evaluate diffusion controlled release of drug from resinate. Int J Pharm. 1986;28:59–66.
Motycka S, Nairn JG. Preparation and evaluation of microencapsulated ion-exchange resin beads. J Pharm Sci. 1979;68:211–215.
Ichikawa H, Fujioka K, Adeyeye MC, Fukumori Y. Use of ion-exchange resins to prepare 100 m-sized microcapsules with prolonged drug-release by the Wurster process. Int J Pharm. 2001;216:67–76.
Korsmeyer RW, Gurny R, Doelker EM, Buri P, Peppas NA. Mechanism of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15:25–35.
Schwartz JB, Simonelli AP, Higuchi WI. Drug release from wax matrices. I. Analysis of data with first-order kinetics and with the diffusion-controlled model. J Pharm Sci. 1968;57:274–277.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published: May 26, 2006
Rights and permissions
About this article
Cite this article
Halder, A., Sa, B. Preparation and in vitro evaluation of polystyrene-coated diltiazem-resin complex by oil-in-water emulsion solvent evaporation method. AAPS PharmSciTech 7, 46 (2006). https://doi.org/10.1208/pt070246
Received:
Accepted:
DOI: https://doi.org/10.1208/pt070246