Abstract
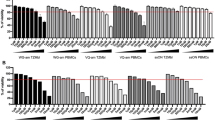
We have evaluated the specificity and the variation in activity against human immunodeficiency virus (HIV) infection of antisense oligodeoxynucleotides (ODNs) with regard to factors such as dose-response range, number and choice of experimental controls, backbone modifications of the ODNs, type of cell infection, length of assays, and delivery approach. The highest level of inhibition was achieved in our long-term assay with MOLT-3 cells acutely infected with HIV-1 (IIIB0 and treated with free phosphorothioate-modified ODNs (PS-ODNs). The highest level of specificity was observed in our short-term assay with MOLT-3 cells acutely infected with HIV-1 (IIIB) and treated with free PS-ODNs. the highest potency (IC50 level) was observed in our short-term chronic-infection model with (DLS)-delivered ODNs in which the DLS delivery improved the ODN activity up to 106 times compared to the activity of free ODNs. Thus, the near blocking of HIV replication obtained when using PS-ODNs appears because of the addition of extracellular and/or membranar effects. The higher efficacy of PS-ODNs compared to unmodified ODNs, when both are delivered with the DLS system, was demonstrated solely in our short-term assay with MOLT-3 cells. Important variations in the level of sequence specificity were observed and depended on the type of control used and the type of cell assay employed. It seems that all 3 groups of control-tested, random, sense sequence, and non-antisense T30177 ODNs might have distinct activity and, consequently, different modes of action in inhibiting HIV replication. Our data buttress the notion that the contribution of the sequence-specific mediated mode of action is minor compared to the other mechanisms involved in ODN antiviral activity.
Similar content being viewed by others
References
Stein CA, Tonkinson JL, Yakubov L. Phosphorothioate oligodeoxynucleotides-antisense inhibitors of gene expression? Pharmacol Ther. 1991;52:365–384.
Zamecnik PC, Stephenson M. Inhibition of Rous sarcoma virus replication and transformation by a specific oligodeoxynucleotide. Proc Natl Acad Sci U S A. 1978;75:280–284.
Zamecnik PC, Goodchild J, Taguchi Y, Sarin PS. Inhibition of replication and expression of human t-cell lymphotropic type III in cultured cells by exogenous synthetic oligonucleotides complementary to viral RNA. Proc Natl Acad Sci U S A. 1986;83:4143–4146.
Stein CA, Cheng Y-C. Antisense oligonucleotides as therapeutics agents: is the bullet really magical? Science. 1993;261:1004–1012.
Agrawal S, Temsamani J, Tang J-Y. Pharmacokinetics biodistribution, and stability of oligodeoxynucleotide phosphorothioates in mice. Proc Natl Acad Sci USA. 1991;88:7595–7599.
Bennett CF, Dean N, Ecker DJ, Monia BP. Pharmacology of antisense therapeutic agents: cancer and inflammation. In: Agrawal S, ed. Antisense Therapeutics. [city?], NJ: Humana Press; 1996:13–46.
Iversen PL, Mata J, Tracewell WG, Zon G. Pharmacokinetics of an antisense phosphorothioate oligodeoxynucleotide against rev from human immunodeficiency virus type 1 in adult male rat following single injections and continuous infusion. Antisense Res Dev. 1994;4:43–52.
Agrawal S, Temsamini J, Galbraith W, Tang J-Y. Pharmacokinetics of antisense oligonucleotides. Clin Pharmacokinet. 1995;28:7–16.
Roush W. Antisense aims for a renaissance. Science. 1997;276:1192–1193.
Wagner RW. The state of the art in antisense research. Nature Medecine. 1995;1:1116–1118.
Stein CA, Kreig AM. Problems in interpretation of data derived from in vitro and in vivo use of antisense oligodeoxynucleotides. Antisense Res Dev. 1994;4:67–69.
Wagner RW. Gene inhibition using antisense oligodeoxynucleotides. Nature. 1994;372:333–335.
Thierry AR, Tackle GB. Liposomes as a delivery system for antisense and rybozyme compounds. In: Akhtar S, ed Delivery Strategies for Antisense Oligonucleotide Therapeutics. Boca Raton, FL: CRC Press; 1995:199–221.
Thierry AR, Dritschilo A. Intracellular avallability of unmodified, phosphorothloated and liposomally encapsulated oligodeoxynucleotides for antisense activity. Nucleic Acids Res. 1992;21:5691–5698.
Lavigne C, Thierry AR. Enhanced antisense inhibition of human immunodeficiency virus type 1 in cell cultures by DLS delivery system. Biochem Biophys Res Commun. 1997;237:566–571.
Kim H-J, Lee K, O Rear JJ. A short sequence upstream of the 5 Major splice site is important for encapsidation of HIV-1 genomic RNA. Virology. 1994;198:336–340.
Muriaux D, Girard P-M, Bonnet-Mathonière B, Paoletti J. Dimerization of HIV-1Lai RNA at low ionic strength. J Biol Chem. 1995;270:8209–8216.
Agrawal S, Tang JY. GEM 91-an antisense oligonucleotide phosphorothioate as a therapeutic agent for AIDS. Antisense Res Dev. 1992;2:261–266.
Matsukura M, Zon G, Shinozuka K, et al. Regulation of viral expression of human immunodeficiency virus in vitro by an antisense phosphorothioate oligodeoxynucleotide against rev (art/trs) in chronically infected cells. Proc Natl Acad Sci U S A. 1989;86:4244–4248.
Ojwang JO, buckheit RW, Pommier Y, et al. T30177, an oligonucleotide stabilized by an intramolecular guanosine octet, is a potent inhibitor of laboratory strains and clinical isolates of human immunodeficiency virus type 1. Antimicrob Agents Chemother. 1995;39:2426–2435.
Popovic M, Read-Conole E, Gallo RC. T4 positive human neoplastic cell lines susceptible to and permissive for HTLV-III. Lancet. 1984;II:1472–1473.
Popovic M, Sarngadharan MG, Read E, Gallo RC. Detection, isolation, and continuous production of cytopathic retroviruses (HTLV-III) from patients with AIDS and pre-AIDS. Science. 1984;224:497–500.
Thierry AR, Lunardi-Iskandar Y, Bryant JL, Rabinovich P, Gallo RC, Mahan LC. Systemic gene therapy: biodistribution and long-term expression of a transgene in mice. Proc Natl Acad Sci USA. 1995;92:9742–9746.
Thierry AR, Rabinovich P, Peng B, Mahan LC, Bryant JL, Gallo RC. Characterization of liposome-mediated gene delivery: expression, stability and pharmacokinetics of plasmid DNA. Gene Ther. 1997;4:226–237.
Pauwels R, Balzarini J, Baba M, et al. Rapid and automated tetrazolium based colorimetric assay for the detection of anti-HIV compounds. J Virol Methods. 1988;20:309–321.
Gura T. Antisense has growing pains. Science. 1995;270:575–577.
Stein CA. Does antisense exist? Nature Medecine. 1995;1:1119–1121.
Stein CA. Phosphorothioate antisense oligodeoxynucleotides: questions of specificity. Trends Biotechnol. 1996;14:147–149.
Lisziewicz J, Sun D, Metelev V, Zamecnik P, Gallo RC, Agrawal S. Long-term treatment of human immunodeficiency virus-infected cells with antisense oligonucleotide phosphorothioates. Proc Natl Acad Sci U S A. 1993;90:3860–3864.
Agrawal S, Goodchild J, Civeira M, Sarin PS, Zamecnik PC. Nucleosides Nucleotides. 1989;8:819–823.
Kinchington D, Galpin S, Jaroszewski JW, Ghosh K, Subasinghe C, Cohen JS. A comparison of gag, pol and rev antisense oligodeoxynucleotides as inhibitors of HIV-1. Antivir Res. 1992;17:53–62.
Zelphati O, Imbach J-L, Signoret N, Zon G, Rayner B, Leserman L. Antisense oligonucleotides in solution or encapsulated in immunoliposomes inhibit replication of HIV-1 by several different mechanisms. Nucleic Acids Res. 1994;22:4307–4314.
Lavigne C, Thierry AR. Lipid-based delivery of combinations of antisense oligodeoxynucleotides for the in vitro inhibition of HIV-1 replication. AAPS Pharmsci. 2001;3:article 7. Available at: http://www.aapspharmaceutica.com/scientificjournals/pharmsci/journal/ 01_07.html.Accessed February 12, 2001.
Jendis J, Strack B, Volkmann S, Boni J, Molling K. Inhibition of replication of fresh HIV type 1 patient isolates by a polypurine tract-specific self-complementary oligodeoxynucleotide. AIDS Res Hum Retroviruses. 1996;12:1161–1168.
Tamamoto T, Yamamoto S, Kataoka T, Tokunaga T. Ability of oligonucleotides with certain palindromes to induce interferon production and augment natural killer cell activity is associated with their base length. Antisense Res Dev. 1994;4:119–122.
Krieg A, Yi, A-K, Matson, S, et al. CpG motifs in bacterial DNA trigger direct B-cell activation. Nature. 1995;374:546–549.
Agrawal S, Ikeuchi T, Sun D, et al. Inhibition of human immunodeficiency virus in early infected and chronically infected cells by antisense oligodeoxynucleotide and its phosphorothioate analogue. Proc Natl Acad Sci U S A. 1989;86:7790–7794.
Anazodo MI, Wainberg MA, Friesen AD, Wright JA. Sequence-specific inhibition of gene expression by a novel antisense oligodeoxynucleotide phosphorothioate directed against a nonregulatory region of the human immunodeficiency virus type 1 genome. J Virol. 1995;69:1794–1801.
Stein CA, Cleary AM, Yakubov L, Lederman S. Phosphorothioate oligodeoxynucleotides bind to the third variable loop domain (v3) of human immunodeficiency virus type 1 gp120. Antisense Res Dev. 1993;3:9–31.
Yakubov L, Khaled Z, Zhang L-M, Truneh A, Vlassov V, Stein CA. Mode of interaction of oligodeoxynucleotides with recombinant sCD4. J Biol Chem. 1993;268:18818–18823.
Yamaguchi K, Papp B, Zhang D, Ali AN, Agrawal S, Byrn RA. The multiple inhibitory mechanisms of GEM 91, a gag antisense phosphorothioate oligonucleotide, for human immunodeficiency virus type 1. AIDS Res Hum Retroviruses. 1997;13:545–554.
Majumdar C, Stein CA, Cohen JS, Broder S, Wilson SH. Stepwise mechanism of HIV reverse transcriptase: Primer function of phosphorothioate oligodeoxynucleotide. Biochemistry. 1989;28:1340–1346.
Lisziewicz J, Sun D, Weichold FF, et al. Antisense oligodeoxynucleotide phosphorothioate complementary to Gag mRNA blocks replication of human immunodeficiency virus type 1 in human peripheral blood cells. Proc Natl Acad Sci U S A. 1994;91:7942–7946.
Bordier B, Perala-Heale M, Degols G, et al. Sequence-specific inhibition of human immunodeficiency virus (HIV) reverse transcription by antisense oligonucleotides: Comparative study in cell-free assays and in HIV-infected cells. Proc Natl Acad Sci U S A. 1995;92:9383–9387.
White RR, Sullenger BA, Rusconi CP. Developing aptamers into therapeutics. J Clin Invest. 2000;106:929–934.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published: April 30, 2002
Rights and permissions
About this article
Cite this article
Lavigne, C., Yelle, J., Sauve, G. et al. Is antisense an appropriate nomenclature or design for oligodeoxynucleotides aimed at the inhibition of HIV-1 replication?. AAPS PharmSci 4, 9 (2002). https://doi.org/10.1208/ps040207
Received:
Published:
DOI: https://doi.org/10.1208/ps040207