Abstract
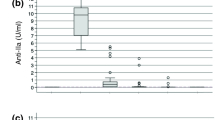
Heparin employed in cardiovascular surgeries often leads to a high incidence of bleeding complications. Protamine employed in heparin reversal, however, can cause severe adverse reactions. In an attempt to address this clinical problem, we developed low molecular weight protamine (LMWP) as a potentially effective and less toxic heparin antagonist. A homogeneous 1880-d peptide fragment, termed LMWP-TDSP5 and containing the amino acid sequence of VSRRRRRRGGRRRR was derived directly from protamine by enzymatic digestion of protamine with thermolysin. In vitro studies demonstrated that TDSP5 was capable of neutralizing various anticoagulant functions of both heparin and commercial low molecular weight heparin preparations. In addition, TDSP5 exhibited significantly reduced crossreactivity toward mouse sera containing antiprotamine antibodies. TDSP5 showed a decrease in its potential in activating the complement system. All of these findings suggested the possibility of markedly reduced protamine toxicity for TDSP5.
In this article, we conducted preliminary in vivo studies to further demonstrate the feasibility and utility of using LMWP as a nontoxic clinical protamine substitute. Dogs were chosen as test animals because they were known to magnify the typical human response to protamine. By using a full spectra of biological and clinical assays for heparin, including the anti-IIa and anti-Xa chromogenic assays and the activated partial, thromboplastin time and TCT clotting assays, TDSP5 showed that it could completely neutralize all these different anticoagulant functions of heparin in dogs. Although administration of protamine in dogs produced a significant reduction in mean arterial blood pressure (−14.9 mm Hg) and elevation in pulmonary artery systolic pressure (+5.0 mm Hg), the use of TDSP5 in dogs did not elicit any statistically significant change in any of the variables measured. Furthermore, the use of LMWP also significantly reduced the protamine-induced transient thrombocytopenic and granulocytopenic responses. The white blood cell counts and platelet counts decreased to 82.1% and 60.0% of baseline, respectively, in dogs given intravenous protamine compared to 97.8% and 88.6% of baseline in dogs receiving TDSP5. These preliminary findings indicated that LMWP could potentially provide an effective and safe means to control both heparin- and protamine-induced complications.
Similar content being viewed by others
References
Ando T, Yamasaki M, Suuzuki K. In: Kleinzeller A, ed. Protamine Molecular Biology, Biochemistry and Biophysics, Vol 12, New York: Springer-Verlag. 1973;1–109.
Metz S, Horrow JC. Protamine and newer heparin antagonists. In: Stoelting RK, eds Pharmacology and Physiology in Anesthetic Practice Vol 1 No 3. Fliadelphia, Pa: JB Lippincott, 1994;1–15.
Weiler JM, Gelhaus MA, Carter JG, et al. Serious adverse reactions to protamine sulfate: are alternatives needed?. J Allergy Clin Immunol. 1985;75:297–303.
Horrow JC. Protamine: a review of its toxicity. Anesth Analg. 1985;64:348–361.
Porsche R, Brenner ZR. Allergy to protamine sulfate. Heart Lung: J Acute Crit Care. 1999, 28:418–428.
Carr JA, Silverman N. The heparin-protamine interaction. J Cardiovasc Surg. 1999;40:659–666.
Click RL, Homburger HA, Bove AA. Complement activation from protamine sulfate administration after coronary angiography. Catheter Cardiovasc Diagn. 1989;16:221–225.
Conzen PF, Habazettl H, Gutmann R, et al. Thromboxane mediation of pulmonary hemodynamic responses after neutralization of heparin by protamine in pigs. Anesth Analg. 1989;68:25–31.
Rent R, Ertel N, Eisenstein R, Gewurz H. Complement activation by interaction of polyanions and polycatious. I. Heparin-protamine induced consumption of complement. J Immunol 1975;114:120–124.
Shanberge JN, Murato M, Quatrociocchi-Longe T, Neste LV. Heparin-protamine complexes in the production of heparin rebound and other complications of extracorporeal bypass procedures. Am J Clin Pathol. 1987;87:210–217.
Wakefield TW, Andrews PC, Wrobleski SK, et al. A [+18RGD] protamine-variant for nontoxic and effective reversal of heparin and low molecular weight heparin anticoagulation. J Surg Res. 1996;63:280–286.
DeLucia A, Wakefield TW, Andrews, PC, et al. Efficacy and toxicity of differently charged polycationic protamine-like peptides for heparin anticoagulation reversal. J Vasc Surg 1993;18:49–60.
Eika C. On the mechanism of platelet aggregation induced by heparin. protamine and polybrene. Scand J Haematol 1972;9:248–257.
Gobel-Geard RJ, Hassouna HI. Interaction of protamine sulfate with thrombin. Am J Hematol. 1983;14:227–233.
Chang LC, Liang J, Lee HF, Lee LM, Yang VC, Low molecular weight protamine (LMWP) as a nontoxic heparin/low molecular weight heparin antidote. I: preparation and characterization. AAPS Pharm Sci. 2001;3(2) Article 17. Available from: http//www.pharmsci.org/scientificjournals/pharmsci/journal/01_17html.
Chang LC, Lee LM, Liang J, Yang VC. Low molecular weight protamine (LMWP) as a nontoxic heparin low molecular weight heparin antidote. II: in vitro evaluation of efficacy and toxicity. AAPS Pharm Sci. 2001;3(2) Article 18: Available from http://www.pharmsci.org/scientificjournals/pharmsci/journal/01_18.html.
Yang VC, Port FK, Kim JS, Teng CL, Till GO, Wakefield TW. The use of immobilized protamine in removing heparin and preventing protamine-induced complications during extracorporeal blood circulation. Anesthesiology. 1991;75:288–297.
Jaques LB. Heparin-anionic polyelectrolyte drugs. Pharmacol Rev. 1979;33:99–166.
Conahan TJ, Andrews RW, MacVaugh H. Cardiovascular effects of protamine sulfate in man. Anesth Analg. 1981, 60:33–36.
Ellison N, Beatty CP, Blake DR, Wurzel HA, MacVaugh H. Heparin rebound studies in patients and volunteers. J Thorac Cardiovas Surg. 1974;67:723–729.
Fabian I, Aronson M. Mechanism of heparin rebound in vitro study. Thromb Res 1980;18:535–542.
Morel DR, Mo Costabella PM, Pitter J. Adverse cardiopulmonary effects and increased plasma thromboxane concentrations following the neutralization of heparin and protamine in awake sheep are infusion rate-dependent. Anesthesiology 1990;73:415–424.
Parkinson C, Gasso P. The use of the dog in toxicity tests on pharmaceutical compounds. Hum Experimental Toxicol 1993;12:99–109.
Wakefield TW, Whitehouse WM Jr. Stanley JC. Depressed cardiovascular function and altered platelet kinetics following protamine sulfate reversal of heparin activity. J Vasc Surg. 1986;1:346–355.
Jacob HS, Craddock PR, Hammersmidt DE, Moldow CF. Complement-induced granulocyte aggregation. N Engl J Med 1980;302:789–794.
Cavarocchi N, Schaff HV, Homburge HA, Schnell WA. Evidence for complement activation by protamine-heparin interaction after cardiopulmonary bypass. Surgery. 1985;98:525–531.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published: July 11, 2001
Rights and permissions
About this article
Cite this article
Chang, LC., Wrobleski, S., Wakefield, T.W. et al. Low molecular weight protamine as nontoxic heparin/low molecular weight heparin antidote (III): Preliminary in vivo evaluation of efficacy and toxicity using a canine model. AAPS PharmSci 3, 19 (2001). https://doi.org/10.1208/ps030319
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/ps030319