Conclusion
For quantitative bioanalytical method validation procedure and requirements, there was a relatively good agreement between chromatographic assays and ligand-binding assays. It was realized that the quantitative and qualitative aspects of bioanalytical method validation should be reviewed and applied appropriately.
-
1.
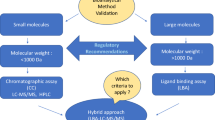
Some of the major concerns between the 2 methodologies related to the acceptable total error for precision and accuracy determination and acceptance criteria for an analytical run. The acceptable total error for precision and accuracy for both the methodologies is less than 30. The 4–6–15 rule for accepting an analytical run by a chromatographic method remained acceptable while a 4–6–20 rule was recommended for ligand-binding methodology.
-
2.
The 3rd AAPS/FDA Bioanalytical Workshop clarified the issues related to placement of QC samples, determination of matrix effect, stability considerations, use of internal standards, and system suitability tests.
-
3.
There was a major concern and issues raised with respect to stability and reproducibility of incurred samples. This should be addressed for all analytical methods employed. It was left to the investigators to use their scientific judgment to address the issue.
-
4.
In general, the 3rd AAPS/FDA Bioanalytical Workshop provided a forum to discuss and clarify regulatory concerns regarding bioanalytical method validation issues.
Similar content being viewed by others
References
Shah VP, Midha KK, Dighe S, et al. Analytical methods validation: bioavailability, bioequivalence and pharmacokinetic studies. Conference report.Eur J Drug Metab Pharmacokinet. 1991;16:249–255.
Shah VP, Midha KK, Findlay JW, et al. Bioanalytical method validation—a revisit with a decade of progress.Pharm Res. 2000;17:1551–1557.
Food and Drug Administration.Guidance for Industry: Bioanalytical Method Validation. Rockville, MD: US Department of Health and Human Services, FDA, Center for Drug Evaluation and Research; 2001.
DeSilva B, Smith W, Weiner R, et al. Recommendations for the bioanalytical method validation of ligand-binding assays to support pharmacokinetic assessments of macromolecules.Pharm Res. 2003;20:1885–1900.
Food and Drug Administration.Draft Guidance for Industry: Safety Testing of Drug Metabolites. Rockville, MD: US Department of Health and Human Services, FDA, Center for Drug Evaluation and Research; 2005.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published: February 9, 2007
Rights and permissions
About this article
Cite this article
Viswanathan, C.T., Bansal, S., Booth, B. et al. Workshop/conference report—Quantitative bioanalytical methods validation and implementation: Best practices for chromatographic and ligand binding assays. AAPS J 9, 4 (2007). https://doi.org/10.1208/aapsj0901004
Received:
Accepted:
DOI: https://doi.org/10.1208/aapsj0901004