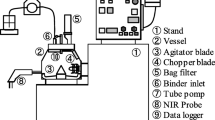
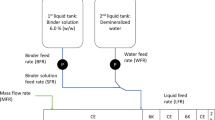
Abstract
Multi-resonance microwave sensors have recently been introduced for moisture monitoring of pharmaceutical particulates up to > 20% residual moisture. The extended measuring range compared to previous systems as well as the microwave moisture values independent of other physical attributes make them promising process analytical technology (PAT) tools for various pharmaceutical production processes. However, so far, research focused on measurements on raw materials or drug-free model granulates and has neither evaluated the applicability for materials with crystal water containing excipients nor for active ingredients. In this study, possible influence of crystal water was evaluated using lactose monohydrate and donepezil hydrochloride, an active pharmaceutical ingredient (API) against dementia. The study clearly showed that the contained hydrate does not cause interferences and is not monitored by the applied frequencies. Material-related limits measuring lactose monohydrate were only observed above typical granulation moistures and could be explained using raw resonance curves. Furthermore, the inclusion of donepezil hydrochloride into the monitored formulations and varied process parameters demonstrated the versatility of the microwave resonance sensor system. Inlet air temperature, spraying rate, and air flow were varied according to a 23 full factorial experimental design. A predictive model (R2 = 0.9699, RMSEC = 0.33%) could be established using samples produced with different process parameter settings adjusted according to the corner points of the full factorial design and validated on the center point granulation processes (RMSEV = 0.38%). Thereby, performance on actual formulations and conditions faced during process development could be thoroughly assessed, and hence, another key requirement for applicability in formulation development could be met.






Similar content being viewed by others
References
Zografi G. States of water associated with solids. Drug Dev Ind Pharm. 1988;14(14):1905–26. https://doi.org/10.3109/03639048809151997.
United States Pharmacopeial Convention, Inc, U.S. Pharmacopoeia-National Formulary [USP 41 NF 36]. Rockville: Water content; 2018. p. 6687–6691.
European Directorate for the Quality of Medicines & HealthCare, European Pharmacopoeia. 9th ed.. Strasbourg: Water: micro determination; 2017. p. 172
European Directorate for the Quality of Medicines & HealthCare, European Pharmacopoeia. 9th ed. Strasbourg: Loss on drying; 2017. p. 53
Watano S, Terashita K, Miyanami K. Development and application of infrared moisture sensor to complex granulation. Bull Univ Osaka Prefect Ser A. 1991;39(2):187–97.
Frake P, Greenhalgh D, Grierson SM, Hempenstall JM, Rudd DR. Process control and end-point determination of a fluid bed granulation by application of near infra-red spectroscopy. Int J Pharm. 1997;151(1):75–80. https://doi.org/10.1016/S0378-5173(97)04894-1.
Rantanen J, Lehtola S, Rämet P, Mannermaa J-P, Yliruusi J. On-line monitoring of moisture content in an instrumented fluidized bed granulator with a multi-channel NIR moisture sensor. Powder Technol. 1998;99(2):163–70. https://doi.org/10.1016/S0032-5910(98)00100-4.
Rantanen J, Antikainen O, Mannermaa J-P, Yliruusi J. Use of the near-infrared reflectance method for measurement of moisture content during granulation. Pharm Dev Technol. 2000;5(2):209–17. https://doi.org/10.1081/PDT-100100536.
Burggraeve A, Monteyne T, Vervaet C, Remon JP, de Beer T. Process analytical tools for monitoring, understanding, and control of pharmaceutical fluidized bed granulation: a review. Eur J Pharm Biopharm. 2013;83(1):2–15. https://doi.org/10.1016/j.ejpb.2012.09.008.
da Silva CAM, Butzge JJ, Nitz M, Taranto OP. Monitoring and control of coating and granulation processes in fluidized beds—a review. Adv Powder Technol. 2014;25(1):195–210. https://doi.org/10.1016/j.apt.2013.04.008.
Rantanen J, Jørgensen A, Räsänen E, Luukkonen P, Airaksinen S, Raiman J, et al. Process analysis of fluidized bed granulation. AAPS PharmSciTech. 2001;2(4):13–20. https://doi.org/10.1007/BF02830561.
Jørgensen A, Rantanen J, Karjalainen M, Khriachtchev L, Räsänen E, Yliruusi J. Hydrate formation during wet granulation studied by spectroscopic methods and multivariate analysis. Pharm Res. 2002;19(9):1285–91. https://doi.org/10.1023/A:1020621906855.
Halstensen M, de Bakker P, Esbensen KH. Acoustic chemometric monitoring of an industrial granulation production process—a PAT feasibility study. Chemom Intell Lab Syst. 2006;84(1–2):88–97. https://doi.org/10.1016/j.chemolab.2006.05.012.
Tsujimoto H, Yokoyama T, Huang CC, Sekiguchi I. Monitoring particle fluidization in a fluidized bed granulator with an acoustic emission sensor. Powder Technol. 2000;113(1):88–96. https://doi.org/10.1016/S0032-5910(00)00205-9.
Wang HG, Senior PR, Mann R, Yang WQ. Online measurement and control of solids moisture in fluidised bed dryers. Chem Eng Sci. 2009;64(12):2893–902. https://doi.org/10.1016/j.ces.2009.03.014.
Buschmüller C, Wiedey W, Doescher C, Plitzko M, Breitkreutz J. In-line monitoring of granule moisture and temperature throughout the entire fluidized-bed granulation process using microwave resonance technology: part I. Pharm Ind. 2009;71(8):1403–8.
Buschmüller C, Wiedey W, Doescher C, Plitzko M, Breitkreutz J. In-line monitoring of granule moisture and temperature throughout the entire fluidized-bed granulation process using microwave resonance technology: part II. Pharm Ind. 2009;71(9):6–12.
Buschmüller C, Wiedey W, Döscher C, Dressler J, Breitkreutz J. In-line monitoring of granule moisture in fluidized-bed dryers using microwave resonance technology. Eur J Pharm Biopharm. 2008;69(1):380–7. https://doi.org/10.1016/j.ejpb.2007.09.014.
Peters J, Taute W, Bartscher K, Döscher C, Höft M, Knöchel R, et al. Design, development and method validation of a novel multi-resonance microwave sensor for moisture measurement. Anal Chim Acta. 2017;961:119–27. https://doi.org/10.1016/j.aca.2017.01.021.
Peters J, Bartscher K, Döscher C, Taute W, Höft M, Knöchel R, et al. In-line moisture monitoring in fluidized bed granulation using a novel multi-resonance microwave sensor. Talanta. 2017;170:369–76. https://doi.org/10.1016/j.talanta.2017.03.105.
Roßteuscher-Carl K, Fricke S, Hacker MC, Schulz-Siegmund M. Influence of in line monitored fluid bed granulation process parameters on the stability of ethinylestradiol. Int J Pharm. 2015;496(2):751–8. https://doi.org/10.1016/j.ijpharm.2015.10.074.
Buschmüller C., In-line monitoring of granule moisture in fluidized bed granulators using microwave resonance technology as novel PAT tool, Dissertation, Düsseldorf, Heinrich-Heine-Universität, 2009.
Lourenço V, Herdling T, Reich G, Menezes JC, Lochmann D. Combining microwave resonance technology to multivariate data analysis as a novel PAT tool to improve process understanding in fluid bed granulation. Eur J Pharm Biopharm. 2011;78(3):513–21. https://doi.org/10.1016/j.ejpb.2011.02.008.
Corredor CC, Bu D, Both D. Comparison of near infrared and microwave resonance sensors for at-line moisture determination in powders and tablets. Anal Chim Acta. 2011;696(1–2):84–93. https://doi.org/10.1016/j.aca.2011.03.048.
Austin J, Gupta A, McDonnell R, Reklaitis GV, Harris MT. A novel microwave sensor to determine particulate blend composition on-line. Anal Chim Acta. 2014;819:82–93. https://doi.org/10.1016/j.aca.2014.02.016.
Magee TRA, McMinn WAM, Farrell G, Topley L, Al-Degs YS, Walker GM, et al. Moisture and temperature dependence of the dielectric properties of pharmaceutical powders. J Thermal Anal Calorim. 2013;111(3):2157–64. https://doi.org/10.1007/s10973-012-2739-y.
McLoughlin CM, McMinn WAM, Magee TRA. Physical and dielectric properties of pharmaceutical powders. Powder Technol. 2003;134(1–2):40–51. https://doi.org/10.1016/S0032-5910(03)00133-5.
European Directorate for the Quality of Medicines & HealthCare, European Pharmacopoeia. 9th ed. Strasbourg: Bulk and tapped density of powders; 2017. p. 359–62
Funding
This project was funded by the German Federal Environmental Foundation (Deutsche Bundesstiftung Umwelt, DBU, Project No. 30816).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Peters, J., Taute, W., Döscher, C. et al. Moisture Monitoring in Fluid-Bed Granulation by Multi-Resonance Microwave Sensor: Applicability on Crystal-Water Containing Donepezil Granules. AAPS PharmSciTech 20, 6 (2019). https://doi.org/10.1208/s12249-018-1209-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-018-1209-7