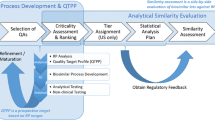
Abstract
Protein therapeutics have unique critical quality attributes (CQAs) that define their purity, potency, and safety. The analytical methods used to assess CQAs must be able to distinguish clinically meaningful differences in comparator products, and the most important CQAs should be evaluated with the most statistical rigor. High-risk CQA measurements assess the most important attributes that directly impact the clinical mechanism of action or have known implications for safety, while the moderate- to low-risk characteristics may have a lower direct impact and thereby may have a broader range to establish similarity. Statistical equivalence testing is applied for high-risk CQA measurements to establish the degree of similarity (e.g., highly similar fingerprint, highly similar, or similar) of selected attributes. Notably, some high-risk CQAs (e.g., primary sequence or disulfide bonding) are qualitative (e.g., the same as the originator or not the same) and therefore not amenable to equivalence testing. For biosimilars, an important step is the acquisition of a sufficient number of unique originator drug product lots to measure the variability in the originator drug manufacturing process and provide sufficient statistical power for the analytical data comparisons. Together, these analytical evaluations, along with PK/PD and safety data (immunogenicity), provide the data necessary to determine if the totality of the evidence warrants a designation of biosimilarity and subsequent licensure for marketing in the USA. In this paper, a case study approach is used to provide examples of analytical similarity exercises and the appropriateness of statistical approaches for the example data.




Similar content being viewed by others
References
FDA. Guidance for industry: quality considerations in demonstrating biosimilarity to a reference protein product. Office of Communications US FDA UCM291134, pp 1–20; 2015.
FDA. Guidance for industry: scientific considerations in demonstrating biosimilarity to a reference product. Office of Communications US FDA UCM291128, pp 1–25; 2015.
FDA Biosimilars: additional questions and answers regarding implementation of the Biologics Price Competition and Innovation Act of 2009. Office of Communications US FDA UCM273001, pp 1–14; 2015.
FDA. FDA briefing document oncologic drugs advisory committee meeting: BLA 125553 EP2006, a proposed biosimilar to Neupogen® (filgrastim). Office of Communications US FDA UCM428780, pp 1–62; 2015.
Kozlowski S. US FDA perspectives on biosimilar biological products, In 2014 Biotechnology Technology Summit. Rockville: IBBR University of Maryland; 2014.
Chatfield MJ, Borman PJ, Damjanov I. Evaluating change during pharmaceutical product development and manufacturing—comparability and equivalence. Qual Reliab Eng Int. 2011;27:629–40.
Chow SC. On assessment of analytical similarity in biosimilar studies. Drug Des. 2014;3:4.
Tsong Y. Equivalence margin determination for analytical biosimilar assessment. In: 2nd statistical and data management approaches for biotechnology drug development IABS. Rockville: USP Headquarters; 2015.
Tsong Y. Development of statistical approaches for analytical biosimilarity evaluation. In: DIA/FDA statistics forum 2015. North Bethesda: Bethesda North Marriott Hotel and Conference Center; 2015.
Dong X, Shen M, Tsong Y. EP2006 statistical equivalence testing for bioactivity and content, FDA, 2015. http://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/OncologicDrugsAdvisoryCommittee/UCM431118.pdf.
Kelley K. Confidence intervals for standardized effect sizes: theory, application and implementation. J Stat Softw. 2007;20:1–22.
Burdick RK, Borror CM, Montgomery D. Design and analysis of gauge R&R studies: making decisions with confidence intervals in random and mixed ANOVA models. Philadelphia: SIAM; 2005.
Apostol I, Brooks PD, Mathews AJ. Application of high-precision isotope ratio monitoring mass spectrometry to identify the biosynthetic origins of proteins. Protein Sci. 2001;10:1466–9.
Acknowledgments
We would also like to acknowledge the AAPS organization and the Biosimilars Focus Group for their support throughout this initiative.
Author information
Authors and Affiliations
Corresponding author
Additional information
See related article, doi: 10.1208/s12248-016-9987-x
Glossary
- 2D-NMR
-
Two-dimensional nuclear magnetic resonance spectroscopy
- SEC-MALS
-
Size exclusion chromatography-multiangle light scattering
- FFF
-
Field flow fractionation
- RP-HPLC
-
Reverse phase high-performance liquid chromatography
- SEC
-
Size exclusion chromatography
- CEX
-
Cation exchange chromatography
- IEF
-
Isoelectric focusing
- LC-MS
-
Liquid chromatography-mass spectrometry
- SPR
-
Surface plasmon resonance
- ELISA
-
Enzyme-linked immunosorbent assay
- HILIC
-
Hydrophilic interaction chromatography
- PAGE
-
Polyacrylamide gel electrophoresis
- FPLC
-
Fast protein liquid chromatography
- DSC
-
Differential scanning calorimetry
- AUC
-
Analytical ultracentrifugation
- PCR
-
Polymerase chain reaction
Rights and permissions
About this article
Cite this article
Burdick, R., Coffey, T., Gutka, H. et al. Statistical Approaches to Assess Biosimilarity from Analytical Data. AAPS J 19, 4–14 (2017). https://doi.org/10.1208/s12248-016-9968-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-016-9968-0