Abstract
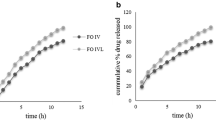
The objective of the present study was to develop a hydrodynamically balanced system for celecoxib as single-unit floating capsules. Various grades of low-density polymers were used for formulation of these capsules. The capsules were prepared by physical blending of celecoxib and the polymer in varying ratios. The formulation was optimized on the basis of in vitro buoyancy and in vitro release in citrate phosphate buffer pH 3.0 (with 1% sodium lauryl sulfate). Capsules prepared with polyethylene oxide 60K and Eudragit RL100 gave the best in vitro percentage release and were used as the optimized formulation. By fitting the data into zero-order, first-order, and Higuchi models, we concluded that the release followed zero-order kinetics, as the correlation coefficient (R value) was higher for zero-order release. For gamma scintigraphy studies, celecoxib was radiolabeled with technetium-99m by the stannous reduction method. To achieve the maximum labeling efficiency the process was optimized by studying the reaction at various pH conditions and stannous concentration levels. The radiolabeled complex was added to the optimized capsule, and dissolution studies were performed to ensure that there was no leaching of radioactivity from the capsules. Gamma imaging was performed in rabbits to assess the buoyancy of the optimized formulation. The optimized formulation remained buoyant during 5 hours of gamma scintigraphic studies in rabbits.
Similar content being viewed by others
References
Arora S, Ali J, Ahuja A, Khar RK, Baboota S. Floating drug delivery systems: a review.AAPS PharmSciTech. 2005;6:E372-E390.
Sheth PR, Tossounian JL, inventors. Hoffman-La Roche, assignee. Sustained release pharmaceutical capsules. US patent 4 126 672. November 21, 1978.
Ponchel G, Irache JM. Specific and non-specific bioadhesive particulate system for oral delivery to the GI tract.Adv Drug Del Rev. 1998;34:191–219.
Lenaerts VM, Gurny R.Bioadhesive Drug Delivery Systems. Boca Raton, FL: CRC Press; 1990.
Deshpande AA, Shah NH, Rhodes CT, Malick W. Development of a novel controlled-release system for gastric retention.Pharm Res. 1997;14:815–819.
Rednick AB, Tucker SJ, inventors. Sustained release bolus for animal husbandry. US patent 3 507 952. April 22, 1970.
Davis SS, Stockwell AF, Taylor MJ, et al. The effect of density on the gastric emptying of single and multiple unit dosage forms.Pharm Res. 1986;3:208–213.
Urguhart J, Theeuwes F, inventors. Alza Corporation, assignee. Drug delivery system comprising a reservoir containing a plurality of tiny pills. US patent 4 434 153. February 28, 1994.
Mamajek RC, Moyer ES, inventors. McNeilab, Inc, assignee. Drug dispensing device and method. US patent 4 207 890. June 17, 1980.
Fix JA, Cargill R, Engle K. Controlled gastric emptying, III: GRT of a non-disintegrating geometric shape in human volunteers.Pharm Res. 1993;10:1087–1089.
Kedzierewicz F, Thouvenot P, Lemut J, Etinine A, Hoffonan M, Maincene P. Evaluation of peroral silicone dosage forms in humans by gamma-scintigraphy.J Control Release. 1999;58:195–205.
Bechgaard H, Ladefoged K. Distribution of pellets in gastrointestinal tract. The influence on transit time exerted by the density or diameter of pellets.J Pharm Pharmacol. 1978;30:690–692.
Paulson SK, Zhang JY, Alan PB, et al. Pharmacokinetics, tissue distribution, metabolism, and excretion of celecoxib in rats.Drug Metabolism Dis. 2000;28:514–521.
Chen GL, Hao WH.In vitro performance of floating sustained release capsule of verapamil.Drug Dev Ind Pharm. 1998;24:1067–1072.
Shah MV, De Gennaro MD, Suryakarma H. An evaluation of albumin microcapsules prepared using a multiple emulsion technique.J Microencapsul. 1987;4:223–238.
Higuchi T. Rate of release of medicaments from ointment bases containing drugs in suspension.J Pharm Sci. 1961;50:874–875.
Hixson AW, Crowell JH. Dependence of reaction velocity upon surface and agitation: theoretical considerations.Ind Eng Chem. 1931;23:923–931.
Li S, Lin S, Chien TW, Daggy BP, Mirchandani HL. Statistical optimisation of gastric floating system for oral controlled delivery of calcium.AAPS PharmSciTech [serial online]. 2001;2:article 1.
Colombo P, Bettini R, Santi P, De Ascentis A, Peppas NA. Analysis of the swelling and release mechanisms from drug delivery systems with emphasis on drug solubility and water transport.J Control Release. 1996;39:231–237.
Ritger PL, Peppas NA. A simple equation for description of solute release, I: Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinder or discs.J Control Release. 1987;5:23–26.
Korsmeyer RW, Deolkar GEP, Peppas NA. Mechanisms of potassium chloride from compressed, hydrophilic, polymeric matrices: effect of entrapped air.J Pharm Sci. 1983;72:1189–1191.
Desai KGH. Preparation and characteristics of high-amylose com starch/pectin blend microparticles: a technical note.AAPS PharmSciTech. 2005;6:E202-E208.
Babbar AK, Singh AK, Goel HC, Chauhan UPS, Sharma RK. Evaluation of99mTc-labeled photosan-3, as a hematoporphyrin derivative, as a potential radiopharmaceutical for tumor scintigraphy.Nucl Med Biol. 2000;27:419–426.
Babbar AK, Singh T, Chauhan UPS. A critical evaluation of radiolabeling of glucaric acid with Tc-99m as a potential myocardial infarct imaging radiopharmaceutical.Ind J Nuclear Med. 1996;11:98–103.
Maggi L, Bruni R, Conte U. High molecular weight polyethylene oxides (PEOs) as an alternative to HPMC in controlled release dosage forms.Int J Pharm. 2000;195:229–238.
Pillay V, Fassihi R. Probing the dynamics of matrix hydration in the presence of electrolytes.Drug Deliv. 2001;8:87–92.
Costa FO, Sousa JJS, Pais AACC. Comparison of dissolution profiles of ibuprofen pellets.J Control Release. 2003;89:199–212.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published: December 28, 2007
Rights and permissions
About this article
Cite this article
Ali, J., Arora, S., Ahuja, A. et al. Formulation and development of floating capsules of celecoxib: In vitro and in vivo evaluation. AAPS PharmSciTech 8, 119 (2007). https://doi.org/10.1208/pt0804119
Received:
Revised:
Accepted:
DOI: https://doi.org/10.1208/pt0804119