Abstract
Background
The term low-grade oncocytic tumour of kidney is an emerging entity describing CD117-negative and cytokeratin 7-positive indolent tumors with overlapping morphological features between oncocytic tumors.
Case presentation
We present herein the case of a 77-year-old female with a 3.2-cm nodule in mid pole of the left kidney. The tumor was uniformly oncocytic with solid, compact nested and trabecular growth patterns. There was common areas of transition to central zones of stromal edema with marked tumor hypocellularity and growth in cords. Some of these areas had adjacent fresh hemorrhage. Immunohistochemistry showed strong and diffuse expression of cytokeratin 7 and negativity for cKIT/CD117.
Conclusion
The proper use of this new diagnostic category avoids labeling such tumors as unclassified renal cell carcinoma – a broad category with different morphologic features and heterogenous prognosis.
Similar content being viewed by others
Background
In 2019, Trpkov and colleagues described an emerging entity among unclassified renal cell neoplasms. They distinguished a new category of oncocytic tumors that do not fit in existing diagnostic entities (Trpkov et al. 2019; Trpkov and Hes 2019). The term low-grade oncocytic tumour of kidney was suggested to describe CD117-negative (in contrast to expected expression in oncocytomas and chromophobe carcinomas) and cytokeratin 7-positive tumors with overlapping morphological features between oncocytic tumors (Trpkov et al. 2019; Trpkov and Hes 2019). In the original series of 28 patients, all remained alive with no evidence of progression (median 21 months of follow up) after surgery (Trpkov et al. 2019).
Case presentation
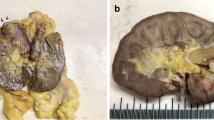
We present herein the case of a 77-year-old female who sought medical assistance due back pain for about 12 months. Abdominal magnetic resonance imaging showed a circumscribed solid tumour in the mid pole of the left kidney (Fig. 1a-b). It was a partially exophytic mass with central area suggestive of necrosis / hypovascularity. A radical nephrectomy was performed.
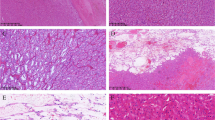
At gross examination, a 3.2-cm brown nodule was observed in mid pole. Cut surface showed central fresh hemorrhage. Tumor was grossly and microscopically confined to the kidney. The tumor cells were uniformly oncocytic with solid, compact nested and trabecular growth patterns. Oncocytic cells shared abundant eosinophilic cytoplasm and nuclei with occasional nucleoli (ISUP grades 1–2) without irregular contours such as “raisinoid”-shaped nuclei of chromophobe carcinomas. These solid areas showed common areas of transition to central zones of stromal edema with marked tumor hypocellularity and growth in cords (Fig. 2a-b). Some of these areas had adjacent fresh hemorrhage (Fig. 2c-d). Necrosis was not observed. Immunohistochemistry showed strong and diffuse expression of cytokeratin 7 (Fig. 2e) and negativity for cKIT/CD117 (Fig. 2f) in different blocks. Additional reactions showed positivity for PAX8 and absent expression of alpha-methylacyl-CoA racemase.
Low-grade oncocytic tumour of kidney. HE stain shows the transition between a solid oncocytic area with cords of tumor cells in edematous stromal (a, 40x; and b; 100x). HE stain also shows zones of fresh hemorrhage (c, 40x; and d, 100x). Stromal edematous areas show frequent hemosiderin-laden macrophages. Immunohistochemistry shows strong and diffuse expression of cytokeratin 7 (e, 40x) and absence of c-KIT expression (f, 100x). Again, hemosiderin-laden macrophages show brown pigment in edematous stroma
Discussion
The case presented herein fits in the description of this new entity. In the original series of 28 patients by Trpkov and colleagues, median age at diagnosis was 66 years and there was a slight female predominance 1.8: 1. Median tumor size was 3.0 cm and 68% of the tumors measured < 4.0 cm and were staged as pT1a (Trpkov et al. 2019). In addition to the proper immunoprofile, the case presented herein showed the described typical findings of coexistent fresh hemorrhage with transition to edematous stromal and growth pattern in cords.
The main differential diagnoses in this context are other oncocytic tumors of the kidney. Oncocytoma may have a broad spectrum of morphologic features (Trpkov et al. 2010). Oncocytomas are, however, typically CD117-positive and show a particular pattern of cytokeratin 7 expression – either entirely negative or predominantly negative with strong staining of isolated tumor cells or small tumor cell clusters. The feature of fresh hemorrhage and growth in cords intermixed in a edematous stroma (characteristic of low grade oncocytic tumour of kidney) is different from the insular growth of oncocytic cells in nests embedded in a fibrous or myxoid stroma (prototypical morphology of renal oncocytoma). High grade oncocytic tumor of the kidney is also an emerging and recently described entity in kidney neoplasia. The morphology and immunophenotype are similar to oncocytoma. In contrast, both oncocytoma and low grade oncocytic tumour of kidney lack the conspicuous finding of high grade ISUP 3 nuclear features (nucleolar prominence) (He et al. 2018). Renal cell carcinoma of chromophobe type could be ruled out in this case due to absence of CD117 expression and characteristic nuclear irregularities (“raisinoid nuclei”). Hybrid oncocytic chromophobe tumor is currently defined as a variant of chromophobe carcinoma. It may occur in sporadic form or related to oncocytomatosis and Birt-Hogg-Dubé syndrome. CD117 is invariably positive in these tumors and - even if raisinoid nuclei may not be detected (or only focally) -, oncocytic cells may show binucleation and perinuclear halos (Srigley et al. 2013).
Conclusion
It is important to recognize additional cases of this emerging entity. The proper use of this new diagnostic category avoids labeling such tumors as unclassified renal cell carcinoma – a broad category with different morphologic features and heterogenous prognosis. At this time, low grade oncocytic tumour of kidney is recognized as an indolent neoplasm with no adverse clinical outcomes reported.
Availability of data and materials
Not applicable.
Abbreviations
- HE:
-
Hematoxylin and eosin stain
- ISUP:
-
International Society of Urological Pathology
- PAX8:
-
Paired box gene 8
References
He H, Trpkov K, Martinek P, Isikci OT, Maggi-Galuzzi C, Alaghehbandan R et al (2018) "high-grade oncocytic renal tumor": morphologic, immunohistochemical, and molecular genetic study of 14 cases. Virchows Arch 473(6):725–738
Srigley JR, Delahunt B, Eble JN, Egevad L, Epstein JI, Grignon D et al (2013) The International Society of Urological Pathology (ISUP) Vancouver classification of renal Neoplasia. Am J Surg Pathol 37(10):1469–1489
Trpkov K, Hes O (2019) New and emerging renal entities: a perspective post-WHO 2016 classification. Histopathology 74(1):31–59
Trpkov K, Williamson SR, Gao Y, Martinek P, Cheng L, Sangoi AR et al (2019) Low-grade oncocytic tumour of kidney (CD117-negative, cytokeratin 7-positive): a distinct entity? Histopathology 75(2):174–184
Trpkov K, Yilmaz A, Uzer D, Dishongh KM, Quick CM, Bismar TA et al (2010) Renal oncocytoma revisited: a clinicopathological study of 109 cases with emphasis on problematic diagnostic features. Histopathology 57(6):893–906
Acknowledgements
Not applicable.
Funding
This study had no funding resources.
Author information
Authors and Affiliations
Contributions
DAA conceived the idea. DAA was the major contributor to the writing of the manuscript. DAA and ISP diagnosed the case. ISP, CGRAM and MVNX were major contributors for critically revising the manuscript for important intellectual content. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Written informed consent was obtained from the patient for participation in the study.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
de Andrade Melo, C.G.R., Xavier, M.V.N., Pimenta, I.S. et al. Low-grade oncocytic tumour of kidney (CD117-negative, cytokeratin 7-positive). Surg Exp Pathol 3, 22 (2020). https://doi.org/10.1186/s42047-020-00074-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s42047-020-00074-z