Abstract
Patient-derived organoids (PDOs) established from tissues from various tumor types gave the foundation of ex vivo models to screen and/or validate the activity of many cancer drug candidates. Due to their phenotypic and genotypic similarity to the tumor of which they were derived, PDOs offer results that effectively complement those obtained from more complex models. Yet, their potential for predicting sensitivity to combination therapy remains underexplored. In this review, we discuss the use of PDOs in both validation and optimization of multi-drug combinations for personalized treatment strategies in CRC. Moreover, we present recent advancements in enriching PDOs with diverse cell types, enhancing their ability to mimic the complexity of in vivo environments. Finally, we debate how such sophisticated models are narrowing the gap in personalized medicine, particularly through immunotherapy strategies and discuss the challenges and future direction in this promising field.
Similar content being viewed by others
Introduction
The widespread discussion on the ethics of animal experimentation emphasizes the necessity for the development of alternative in vitro models to advocate for the “3Rs initiative” that represent reduction, refinement, and replacement of laboratory animals [1]. Legislation signed in late December 2022, indicates a significant shift in drug safety regulation, as it eliminates the requirement for animal testing to obtain U.S. Food and Drug Administration approval for new medicines [2]. This marks a radical change from over 80 years of reliance on animal experimentation in the realm of drug safety. While drug discovery cannot rely solely on in vitro models, the establishment of solid pre-clinical ex vivo platforms using patient-derived material, can yield valuable outcomes that, in turn, contribute to minimizing the need for additional in vivo experiments and facilitating clinical transition.
For many years, two-dimensional (2D) cell cultures and in vivo tumor models have been the standard for drug discovery and cancer research [3]. Multiple research groups have elegantly reviewed the limitations of 2D models in current drug development while introducing new state of the art in vitro and ex vivo models [3,4,5]. In 2011, Sato et al. established for the first-time colorectal cancer organoids from freshly isolated patient-derived tissue [6]. Since then, the establishment of organoids as pre-clinical model with patient-specific signatures allowed bridging the gap between 2D and more complex models of colorectal cancer (CRC) [7]. Organoids are self-organising 3D tissues, which mimic essential functional, structural, and biological characteristics, as well as histopathological and genomic features of the tissue they derive from [7]. They can be cultivated from tissue-derived cells, induced pluripotent stem cells or from surgically removed patient samples [8,9,10]. The cultivation of PDOs requires an interdisciplinary network between fundamental researchers and clinicians, including surgeons, oncologists and clinical pathologists. Close collaboration between these groups within the team is essential to secure optimal conditions from tissue resection to model establishment [4]. In experimental precision oncology, such models serve for various applications, including drug screening, optimization of immunotherapies, validation of combination treatment, design of personalized drug regimen, mechanistical evaluation, and biomarker identification. Consequently, organoids have emerged as relatively inexpensive and representative platforms for modelling cancer heterogeneity and interactions with the tumor microenvironment (TME) in vitro [11].
CRC is a heterogenous tumor commonly defined by four Consensus Molecular Subtypes (CMS). Each subtype harbors a different immune landscape and tumor-microenvironment [12]. For example, CMS1 is characterized by MSI high tumor phenotype and is associated with a high T cell infiltration and activation. This CMS1 group is the most likely to respond to immunotherapy. On the other hand, the CMS2 and CMS3 are poorly immunogenic, characterized with a low immune cell infiltration. Finally, the CMS4 is strongly enriched with immunosuppressive cell populations (such as regulatory T cells (Treg) and M2-like macrophages) and stromal cells. The latter three CMS subtypes represent microsatellite stable (MSS) tumor phenotype and are less likely to respond to immunotherapy, thus a better understanding of those CMS subtypes will guide future drug selection and combination [12,13,14,15].
It was demonstrated that tumor organoids have a broad conservation of the phenotype of tumor cells [8, 10, 16, 17]. In addition, the cytokeratin 20 positive and cytokeratin 7 negative (CK20+/CK7−) immunophenotype, highly characteristic of CRC tumors, was observed in the majority of PDOs [8]. Furthermore, the expression of Ki67, CDX2, a homeobox gene that marks intestinal differentiation,
β-catenin, cytokeratins (CK), and especially CK20, all potential markers for the clinical diagnosis of CRC, was retained in organoids compared to the primary tumor Sect. [16]. Additionally, expression levels of mismatch repair (MMR)-related proteins, including MLH1, MSH6, MSH2, and PMS2, were detected in the PDOs [16]. With whole exome sequencing, researchers demonstrated a similarity in 96% mutations in key driver genes comparing PDOs and primary tumors [10, 17]. However, certain samples exhibited new somatic variants that were not detected in the primary tumor [8]. Overall, in most of the cases the PDOs generated resembled the patients’ primary tumor. On average, there is 76% accuracy in organoids predicting patient response, with a sensitivity of 0.79 and a specificity of 0.75 [18], making PDOs suitable to guide the selection of an effective therapeutic approach, personalized to each patient [9, 10, 17, 19,20,21]. Similarly, in case of organoids isolated from the liver, where CRC metastases are often located (mCRC), PDOs exhibited comparable responses to patient outcomes, underlining their clinical relevance [22]. Additionally, using PDOs cultivated from refractory mCRC patients, who underwent two standard of care treatment protocols, next-generation sequencing and subsequent drug screen analyses, yielded clinical results for four patients. Within three weeks, these patients received their recommended therapies, resulting in a minimum of 5 months stable disease [23]. However, not all significant responses in PDOs can be translated in effective clinical outcomes, indicating the need for further refinement in PDO-guided therapies [24]. Thus, while PDOs hold potential in personalized CRC treatment selection, continued research is essential to optimize their clinical utility and overcome existing limitations.
It is currently well acknowledged in the field that combinatory treatment holds better potential of cure over monotherapy interventions [25]. For cancer treatment, a multifactorial disease, such single target agents used at high doses have shown limited efficacy due to induction of resistance and relatively strong side effects [25, 26]. This paved the way for poly-pharmacology as a more potent therapeutic tool [27, 28], notably for complex diseases that fail to respond to monotherapy treatment strategies [29, 30]. Combinatory pharmacological treatment is defined as the use of pharmaceutical agents acting on multiple molecular targets or multiple biochemical pathways. This can be achieved by combining drugs, targeting different signaling pathways or by designing a drug that acts on multiple targets with various affinities. Nowadays, it is clinically achieved by the use of chemotherapy based combination, FOLFOXIRI (FOL: folinic acid; F: 5-FU; OX: oxaliplatin; IRI: irinotecan), as main or adjuvant treatment modality in different stages of CRC [31].
This attractive avenue as a novel cancer treatment strategy is still in its infancy, specifically for CRC, as our knowledge on the complex drug-drug interactions in the cancer environment is still not fully elucidated. Many challenges remain on how to (i) select the drug candidates and their dose, (ii) which parameter to evaluate the efficacy and toxicity of such combinations, (iii) which relevant pre-clinical cellular model to use for a facilitated clinical translation or how to (iv) evaluate drug-drug interactions [32]. Current advances in ‘omics’ technologies, together with in silico modelling and whole genome sequencing, have greatly improved the workflow and have contributed to drug (combination) development discovery, yet the validation in organoid models remains rather rare.
In this review, we focus upon PDOs application for the validation and optimization of multi-drug combinations as part of personalized treatment strategies for CRC. We discuss recent efforts to enrich organoid models, contributing to mimic the complexity of the in vivo environment. We address how such advanced models are utilized to bridge the gap for better tailored clinical translation using immunotherapeutic strategies, along with consequent challenges and potential future directions.
Combination of targeted therapies in CRC treatment-naïve and treatment-resistant organoids
Despite the strong potential attributed to PDOs as a pre-clinical model in translational oncology and therapy development against cancer, dedicated drug-combination screens for personalized treatment strategies for CRC using PDOs are scarcely reported. When it comes to combinations of targeted therapies, organoids have been employed in different modalities. Either to validate already established drug combinations, or to directly screen for new combinatorial treatments.
Treatment-naïve tissues
Multiple studies have highlighted the strong therapeutic potential of the combination of MEK and PI3KCA inhibitors [33, 34], using CRC 3D models [30, 35], however, very few validated this combination strategy directly in CRC PDOs. Atanasova et al. elegantly presented the difference of response to combination therapy in CRC according to a model used [36]. In their study, no difference in efficacy was reported between the triple-drug combination consisting of Torin 1 (mTOR1/2 inhibitor), MK2206 (AKT inhibitor) and selumetinib (MEK1/2 inhibitor), when compared to the efficacy of investigational Gedatolisib (pan-class I isoform PI3K and mTOR inhibitor) combined with selumetinib, in 2D and homogeneous 3D spheroids of DLD1 colon cancer cells. However, difference in activity was seen only in PDOs derived from both primary tumor and liver metastases CRC patients, harboring mutations in PI3K and/or RAS-RAF-MAPK pathways.
Another main strategy in leveraging the full potential of PDOs, consists in directly using the organoids technology in optimizing combinatory treatment strategies for CRC patients. In a recent study conducted by our research team, Ramzy et al. established an innovative platform for the rapid optimization of drug combination (ODC) using individually grown PDOs tailored specifically to individual patients [19]. To do so, the authors had to preliminarily optimize a culture seeding method that allows the organoids technology to be adapted to a high throughput drug screen (Fig. 1A). Their organoid culture procedure allowed to reproducibly culture single organoids per well at a standardized size of 350–450 μm. This proprietary phenotypically-driven platform called Therapeutically-Guided Multidrug Optimization (TGMO) [19, 35, 37], was applied to systematically screen combinations of tyrosine kinase inhibitors (TKIs) on PDOs obtained from individuals with primary and metastatic CRC, in a clinically relevant timeframe. Specifically, the ODC comprising of four drugs (Fig. 1B), i.e. regorafenib, vemurafenib, palbociclib, and lapatinib applied at low doses showcased a substantial inhibition of cell viability, achieving up to 88% reduction, in PDOs derived from a patient with CRC liver metastases (stage IV), characterized as CMS4/ CRC intrinsic subtype A (CRIS-A). In addition, the activity of the ODC showed a significant therapeutic window when evaluated on non-cancerous colon model. Moreover, the efficacy of the patient-specific ODCs was found to significantly surpass that of FOLFOXIRI, at clinically used doses. These findings underscored the potential of the TGMO platform in tailoring cancer treatment strategies by leveraging ex vivo patient material, in a clinically relevant time frame of 2–3 weeks [19].
Using as similar design of experiment as described previously in the study of Ramzy et al., Thng et al. applied a phenotypic approach, i.e. Quadratic Phenotypic Optimization Platform, to screen for synergistic two-drug combinations. It was done using PDOs from primary and metastasis lesions [38]. Interestingly, for two of the three pairs of matched PDOs, the authors found similar sensitivities in both primary and corresponding metastasis-based PDOs to the patient-specific synergistic two-drug combinations (Fig. 1B), i.e. regorafenib + SN38 and regorafenib + vorinostat (HDAC inhibitor [39]), respectively. However, for the third patient, different drug combinations were optimized showing different efficacy between the metastases and the primary PDO. The authors limited their screen on drug pairs, highlighting the difficulty for more than 2 drugs to be administered systemically.
Mertens et al. introduced a phenotypically-driven approach using a microscopy-based strategy, to screen for a triple-drug combination that allows to stabilize the cytotoxic effect in advanced stage of CRC [40]. Their drug-repurposing screen yielded 34 positive hits out of 414 clinically approved drug candidates tested, in monotherapies or drug-pairs, on a panel of 36 KRAS-mutant CRC PDOs. By complementing their metabolic inhibition assay with microscopy, they were able to identify the microtubule-targeting agents, more specifically Vinorelbine®, as the most potent class of compounds to result in a switch from a drug-induced cytostatic phenotype, upon treatment with lapatinib (a dual HER2/EGFR inhibitor [41]), trametinib (MEK1/2 inhibitor [42]) at clinically used doses adapted to a patient clinical maximum plasma concentration (Cmax), to a drug-induced cytotoxic phenotype. The authors tested multiple combinations of Pan-HER/MEK used in trials (i.e. afatinib + selumetinib) and observed a similar effect. Even though synergy was higher for the triple-drug combination of lapatininb + trametinib + Vinorelbine®, the observed anti-tumor effects remained unchanged when the dual TKIs were replaced by other mechanistically-identical drugs (afatinib + selumetinib), provided that downstream MAPK pathway signaling was effectively inhibited (Fig. 1B). Their findings were further validated in vivo, by reproducing the clinical exposure in tumor mouse models, where 62% reduction in tumor growth was observed for the combination of lapatininb + trametinib + Vinorelbine® compared to the sham-treated mice [40].
Chemo-resistant tissues
Described above dual inhibition using a combination of pan-HER2 and MEK1/2 inhibitors in addition to a third agent, has been also shown to be effective in CRC PDOs resistant to chemotherapy. Even though current treatment modalities for CRC, including chemo-/radio-therapy, targeted agents and immunotherapy have highly improved CRC patient survival, some patients have inherent genetic mutations that result in resistance to treatment, or lose sensitivity to treatment over time. These intrinsic or acquired resistance are mainly due to changes in tumor-drug metabolism, cellular transportation or target, as reviewed by others [43,44,45]. In a previous study, we have generated in vitro and ex vivo CRC cell models resistant to clinically used first line chemotherapy, FOLFOXIRI, concomitantly administered to the cells [19, 46, 47]. We highlighted the strong potential of ODCs in overcoming chemotherapy resistance in CRC, where different combinations of four TKIs inhibited up to 90% of viability in chemotherapy-resistant cells. However, this was shown in FOLFOXIRI-resistant cells cultured in 2D, and very limited number of studies have been carried out to optimize drug combinations on chemo-resistant organoids.
Boos et al. exposed KRAS and BRAF wild-type PDOs from three patients (one primary CRC metastatic tumor and two CRC liver metastases) to a chronic combination treatment of FOLFIRI + cetuximab, administered concomitantly, to establish ex vivo chemoresistance within a variable patient-dependent period from 4 to 6 months [48]. Interestingly, no mutational changes in KRAS were observed in all three PDOs after long-term exposure to the combination treatment. Therefore, the authors engineered a KRASG12D mutation using CRISPR/Cas9. This reduced the effect of cetuximab or a dual inhibition using afatinib + selumetinib in all PDOs tested. To overcome such resistance, the authors shed a light on Aurora A kinase as a potential target. The triple-therapy with afatinib + selumetinib + alisertib (Aurora A inhibitor) [49], illustrated in Fig. 1B, showed a significant reduction in cell viability of all PDOs that were EGFR-therapy resistant, through a concomitant cell cycle arrest in G2/M phase and the dual inhibition of EGFR pathway.
Usui et al. introduced a new strategy to overcome resistance to conventional chemotherapy in CRC. The authors underlined the strong potential of stem cell signal inhibitors in overcoming such resistance. They used their air-liquid interface (ALI) model [50], where patient-derived tissue, embedded in collagen layer submerged in intestinal stem cell media, formed ALI organoids over time recapitulating the intestinal epithelial and mesenchymal structure as the primary tumors. Their results highlighted that the combination of a Hedgehog signal inhibitors (AY9944 or GANT61) with 5-FU, irinotecan or oxaliplatin (Fig. 1B) significantly decreased the tumor organoids cell viability when compared to the activity of each chemotherapy alone. They further confirmed their results through protein levels expression, where the treatment of ALI organoids with AY9944 or GANT61 resulted in a downregulation of stem cell markers (c-MYC, Nanog and CD44) expression through downregulation of GLI-1 protein, known to be overexpressed in CRC chemo-resistant cell lines [50].
Even though the above-mentioned studies introduce novel approaches to optimize multi-drug combinations for CRC patients, the PDOs are still far from being ideal models as the immune, vascular and stromal compartments are not present. To leverage the full potential of drug combinations, more complex models need to be developed. Below we refer to studies where attempts to enrich the microenvironment of organoids are discussed.
Organoids formation and signaling pathways of optimized drug combinations (ODCs). (A) Schematic representation of different steps in an in vitro drug screening process. (B) Different combinations of targeted therapies optimized using CRC PDOs and their known targeted signaling pathways. Created with BioRender
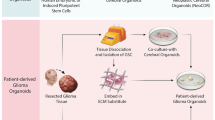
Microenvironment enrichments of PDOs
While PDOs maintain the tumor characteristics of a patient and can be predictive of their response to different treatments, as a preclinical model they have multiple limitations. This is mainly translated by the lack of certain actors present in TME, e.g. immune cells, endothelial cells and fibroblasts. While some PDOs from different cancer types preserved integrated components of the immune system [44], the majority of the PDOs do not contain immune cells [51].
Enrichment with immune cells
Therefore, different studies reported complex co-culture models of CRC PDOs with the addition of different types of immune cells as schematically represented in Fig. 2. This, in turn, provides an opportunity to explore response and resistance mechanisms to both immune- and targeted- combination therapies [52]. For instance, with the addition of IL-2, integrated immune components in PDOs can be retained for up to 7 days [38]. However, since only a limited number of immune cells are preserved, an additional number of immune cells of different subtypes need to be present to more accurately represent the TME. Fang et al. cultured CRC PDOs with patient-derived CD8+ T cells together with macrophages isolated from the peripheral blood mononuclear cells (PBMCs) (Fig. 2). The latter was activated by the addition of macrophage colony stimulating factor (M-CSF) for 7 days before co-culturing them with the CRC organoids [53]. The levels of SIRT1 in CRC was shown to influence the TME and cancer progression. CRC samples with high expression of the protein, had decreased levels of CD8 + T cells, while the percentage of tumor-associated macrophages (TAM) was increased, inhibiting tumor proliferation and the anti-tumor activity of CD8 + T cells [53]. This study using organoids co-culture reflects the crosstalk complexity that can be observed in patient samples with the advantage of offering a real-time follow-up setting.
Other research groups reported the addition of dendritic cells (DCs) to the PDOs culture. DC are required for the detection, processing, and presentation of tumor antigens as well as the activation of antigen-specific T cells to orchestrate an effective antitumor response. Subtil et al. successfully incorporated monocyte-derived dendritic cells (MoDCs) differentiated with granulocyte-macrophage colony-stimulating factor (GM-CSF) and IL-4 for 5 days, from healthy donors into a 3D collagen matrix PDOs co-culture system. In the presence of PDOs, reduced expression of CD86 was observed, hinting towards a tumor-induced immunosuppressive effect. Immature DCs (iDCs) were activated with IL-6, IL-1b, TNF-a and PGE2, for 24 h before harvesting. In co-cultures of PDOs with iDCs or mature DCs (mDCs), iDCs were being significantly closer to the tumor and demonstrated higher infiltration compared to the mDCs [54]. Due to the fact that the iDCs were not matured, they had ability to induce T cells differentiation into regulatory T cells, which was associated with pro-tumorigenic effects [55]. Furthermore, the co-culture of conventional DC type 2 (cDC2) with PDOs (Fig. 2) resulted in an infiltration of the cells into the PDOs and the activation of T cells in the co-culture. The cytokine production and secretions by the PDOs (like PGE2 and IL-6) was shown to influence the conversion of cDC2 to DC3-like cells which has been linked to impairments in T cell proliferation and activation thus inducing an immunosuppressive environment [56]. In this context, assessing the immune cells phenotypes in co-culture is also important to understand what drive the pro or anti-tumoral environment that probably impact as well the drug response.
Moreover, Natural Killer (NK) cells gained significant attention recently [57, 58], specifically in CRC, where tumor-infiltrating NK cells and their cooperation with T cells [59, 60] has been linked to a good prognosis [61, 62], highlighting their important role in controlling tumor growth [63] and overcoming resistance to chemo- and T cell-based immune-therapy [64]. Lanuza et al. developed a 3D co-cultured CRC model with allogenic activated NK cells (Fig. 2), enriched with CD56 antibody magnetic beads. It was shown that the NK cells concentration is a critical parameter during the optimization, as only at high concentration NK cells were able to promote tumor cell death before infiltration [65]. To improve the model relevance, Schnalzger et al. optimized a co-culture of tumor colon PDOs with NK cells. During the co-culture optimization they found that unlike T cells, NK cells killing and infiltration was dependent of extracellular matrix (ECM) density used in organoid co-culture [52]. Recently, an ALI culture method described by Neal et al., using PDO from primary and metastatic tumors was shown to retain stromal myofibroblast as well as immune cells like T, B, NK cells and macrophages. With this method, the authors showed the feasibility and the potential application of ALI-cultured organoids for testing the efficacy of immune checkpoint blockade treatments comprising a large proportion of TME components derived from patient samples [66].
Enrichment with stromal cells
Aside from immune cells, the TME consists of a variety of stromal cells. The stroma is defined as the surrounding ECM and the mesenchymal cells within it including endothelial cells (ECs) and cancer-associated fibroblasts (CAFs). Crosstalk between stromal, immune and cancer cells play a pivotal role in tumorigenesis and in treatment resistance in CRC [67].
ECs are the main components of the tumor vasculature, with angiogenesis being an important progression hallmark of cancer [68, 69]. Angiogenesis and TECs play critical role in CRC metastasis promoted by the development of neo-vascularization regulated by various pro-angiogenic factors (such as VEGF, PDGF, FGF, and angiopoietin) [70]. The angiogenic tumor vasculature, derived from activated ECs or stem cells, also expresses immunosuppressive molecules [71], that create a barrier for immune cells. The barrier function of the tumor vasculature is also caused by the lack of tumor endothelial cell adhesion molecules such as intercellular adhesion molecules (ICAMs), vascular cell adhesion molecules (VCAMs) and selectins - a phenomenon that is called endothelial cell anergy [72, 73]. This tumor endothelial cell anergy has been presented as a vascular immune checkpoint [74], that the tumor has hijacked from an embryonic regulatory mechanism [75].
Co-culturing organoids with ECs enhances the physiological relevance of the model by mimicking the tumor microenvironment more accurately. This approach enables the study of tumor-vascular interactions, angiogenesis, and drug responses in a more realistic setting. In a recent review, Strobel et al. presented various vascularization strategies within organoids, see Fig. 2 [76]. Important points raised were the use of tissue-specific endothelial cells, the culture conditions with the use of ECM and specific growth factors to support 3D structure and cell differentiation [76,77,78]. In this context, the addition of a perfusion system help in the development a vascular network within the organoid compared to static culture [79,80,81,82]. Investigating how CRC cells interact with endothelial cells can uncover new targets for anti-angiogenic therapies and combination therapies perspectives. Truelsen et al. introduced “The Cancer Angiogenesis Co-Culture assay” as an in vitro functional assay to study the treatment response to anti-angiogenic agents in organoids [83]. In their model, endothelial cells (HUVECs) were cultured in co-culture with fibroblasts, where HUVECs formed tube-like structures within the layers of fibroblasts and ECM. Two days later the PDOs were added to the co-culture. The authors highlighted the strong potential of regorafenib and bevacizumab, inhibitors of VEGFR and VEGF, respectively to inhibit endothelial tube formation in experimental conditions with PDOs compared to the control, with no tumoroids and no treatment.
Moreover, CAFs are responsible for the secretion of various chemo- and cytokines to promote the progression of CRC and might alter the immune cell population in the TME [84]. The CAFs are representing fibroblast population located within or surrounding the tumor [85]. In CRC the CAFs are often associated with poor prognosis and therapy resistance [86, 87]. The ‘mesenchymal-like’ CMS4 characterized by a high content of CAFs [88], represent an important number of cases in CRC (23% of the cases) [12, 89]. Therefore enriching organoid environment with CAFs was conducted to investigate their role in the TME remodeling, promoting tumor growth, invasion, and resistance to treatment. Strating et al., showed that such co-culture resulted in elevated expression of ECM components, increased glycolysis, or hypoxia [90]. Additionally, CAFs induced a partial epithelial-to-mesenchymal transition (EMT) in a cancer cell population, promoting immunosuppression and ECM stiffening. The co-culture medium composition revealed an elevated level of the immunosuppressive factors, such as TGF-b1, VEGF-A and lactate, responsible of the T cell proliferation inhibition. In the study of Luo et al., the addition of patient-derived CAFs to the organoids in a 2:1 ratio (Fig. 2), resulted in an enhanced tumor growth in CRC PDOs compared to culturing PDOs without CAFs, suggesting that the secretion of paracrine growth factors stimulated tumor progression. Transcriptional analyses revealed potential restoration of crucial survival pathways and cancer-CAF interactions, also observed in patient tumor tissues. Furthermore, drug efficacy testing in CRC PDOs and co-cultures with CAFs demonstrated increased resistance levels, emphasizing the importance of incorporating CAFs when assessing drug responses in PDOs [91].
Human colorectal organoid on-chip models are an emerging innovative methods that integrate microfluidic technology to create a more physiologically relevant system by ensuring a proper irrigation, cyclic strain and fluid shear stress mimicking in vivo conditions for the living cells (Fig. 2) [92]. The dynamic environment facilitates longer-term experiment and the investigation of tumor progression, metastasis, and the efficacy and safety of combination therapies [93, 94]. For example, Strelez et al., managed to optimize a chip model with CRC tumors, including both CAFs and endothelial cells, to study the influence of the TME on early metastatic spread [93]. This model can also be used to recapitulate the multicomponent structure of human organs to study dynamic interaction of the TME with treatment. Using a microfluidic chip, Jenkins et al., were able to evaluate the response and resistance to PD-1 blockade, by profiling the secreted cytokines in multi-cellular PDOs [95]. The engineering behind organoid on-chip models is challenging and still need improvement for standardization of the method for robust reproducibility but it unlocks new avenues of research by using models that are closer to in vivo [96].
Collectively, these co-culture models represent a valuable tool for the identification of promising new therapeutics and underscore the importance of multicellular interactions in this process. Alongside, future studies should be performed to standardize co-culture settings to harmonized protocols and to better replicate the treatments strategies.
Enriched organoid model to develop combined immunotherapeutic strategies
Organoids isolated from CRC have emerged as powerful prognostic models for immunotherapy screening in the past few years. In this field, the immune context of cancer has a significant impact on patient response to immunotherapy.
The CMS2 and CMS3 are poorly immunogenic with low immune cell infiltration, whereas the CMS4 is strongly enriched with immunosuppressive cell populations and stromal cells. Those CMS represent MSS tumor phenotype and are less likely to respond to immunotherapy. It was demonstrated that an immune infiltration in CRC by CD4 T cell, CD8 T cell, B cell, NK cell, tertiary lymphoid structure and macrophages represent a good prognostic value for patients [14, 61, 97,98,99,100]. On the opposite M2 macrophages characterized by an anti-inflammatory phenotypes are associated with a poor prognosis [61, 101, 102].
Therefore, an organoid co-cultures with stromal and immune cells were established to recapitulate better the heterogeneity and microenvironment of a patient tumor, as well as provide a promising platform for assessing immunotherapy response and guiding treatment selection in patients who are unlikely to respond to current first-line therapy [103]. Optimization of immunologically representative scalable functional assays represent an important step toward increasing the value of PDOs as a preclinical model for immunotherapy screening. Several new technologies have been developed by different groups [104,105,106,107,108], in which PDOs are used to identify tumor-reactive T cells, assess a patient’s potential response to immune checkpoint blockade, and identify neoantigens in individual tumors, to which cell therapies could be applied. Selected studies are discussed below.
CAR-T cell therapy
Strategies to co-culture tumor organoids with autologous T cells have demonstrated clinically significant treatment optimization, demonstrating the potential and versatility of PDOs and T cell co-culture systems for immunotherapy screening. Chimeric antigen receptor-T (CAR-T) therapies consist of genetically modified T cells able to specifically recognize tumor antigens and eradicate cancer cells. CAR-T therapies are established FDA-approved treatment for several blood cancers, such a B cell leukemia or lymphoma, but still remain a challenge for solid tumors [109, 110]. Lack of efficacy of CAR-T therapy may be caused by the barrier function through the lack of adhesiveness of the tumor vasculature described earlier. In this line of thought it might be advantages to design CAR-T therapies against the tumor vasculature, as this may solve the vascular barrier problem [111, 112]. Another problem with CAR-T therapy in patients with solid tumors is discontinuation of therapy caused by on-target off-tumor toxicities [113, 114]. Those often occurring toxicities support the use of PDOs for pre-clinical development to prevent and resolve them by finding antigen exclusively expressed in tumor cells [109, 115]. In clinical settings, poor response to CAR-T-based treatments is also caused, among others, by the TME composition in solid tumors, therefore there is a need for adequate co-culture models with preserved TME [116]. Moreover, due to limitations in treating solid tumors with CAR-T cells, an unmet need remains to discover new targets and optimize combination strategies.
Schnalzger et al. proposed an original in vitro platform designed for pre-clinical testing of CAR NK cells using CRC PDOs as shown in Fig. 3 [52]. The authors demonstrated the response of CAR-NK cells to two clinically relevant cancer-associated antigens, namely epidermal growth factor receptor variant III (EGFRvIII) and the WNT receptor FRIZZLED (FZD). EGFRvIII-specific CAR-NK cells showed high selectivity for PDOs expressing mutant EGFR, with no off-target toxicity observed in healthy colon organoids. Conversely, FZD-specific CAR-NK cells showed non-specific killing of PDOs regardless of their origin (normal human colon organoids and primary fibroblasts derived from non-pathological mucosa collected either during preemptive colonoscopy or from tumor-adjacent normal colon after tumor resection) and FZD receptor status, suggesting that clinical targeting of FZD may lead to mucosal toxicity.
In CRC the expression of CD70 on CAFs has been identified as a novel negative prognostic marker and thus independently of the MMR status [117, 118]. The CD70 expression has been suggested to help tumor immune evasion and accelerate tumor growth. Of note, a higher CD70 expression by CAFs compared to tumor cells was showed in CRC cases (14,9% vs. 2,2%) [118]. Recently, Van den Eynde et al. have demonstrated that CD70 in CRC and pancreatic ductal adenocarcinoma patients could be a potential therapeutic target for both tumor cells and tumor promoting CAFs [119]. Using PDOs co-cultured with CAFs they showed that CD70-CAR-NK cells in combination with IL-15 is needed for an effective eradication of low- and high-expressing CD70 + tumor cells and CAFs.
The simultaneous administration of CAR-NK cells and CAR-T cells, could also increase the effectiveness of treatment, therefore the use of PDO can lead to a rapid identification CAR T/NK new target and test of their efficacy [120].
Immune checkpoint inhibitors (ICIs)
The use of immune checkpoint blockers such as PD-1/PD-L1 inhibitors, has emerged as a cornerstone in cancer treatment. The PD1 expression has been mainly described on cell surface of activated lymphocytes populations and particularly CD8+ T cell [121,122,123]. Recently, PD1 expression has also been described in a variety of immune cells (DCs, macrophages, NK cells) and tumor cells [124, 125]. The interaction of PD1 with its ligand PD-L1 impair T cell activation, cytotoxicity and induce an immune tolerance favorable for tumor cell onset [126, 127].
In 2017, FDA approved pembrolizumab and nivolumab, anti-PD1 monoclonal antibodies (mAb), for the treatment of adult and pediatric patients unresectable or metastatic CRC with mismatch repair deficient and microsatellite instability high (dMMR/MSI-H) that has progressed prior a fluoropyrimidine, oxaliplatin, and irinotecan treatment [128, 129]. In 2020, pembrolizumab has been approved as a first-line treatment for patients with unresectable or metastatic MSI-H or dMMR colorectal cancer [130]. However, its efficacy remains limited to patients with dMMR/MSI-H tumors, which represents approximately 15% of CRC patients [131]. Enhancing the efficacy of anti-PD1 therapy through different pathways is pivotal, driving significant research efforts towards identifying novel combination strategies (Fig. 3).
One of the strategies used by Deng et al. was to combine small molecules with immune checkpoint blockade (Fig. 3). Using MC38 murine-derived organotypic tumor spheroids, the authors demonstrated that a combination of CDK4/6 inhibitor (trilaciclib or palbociclib) with anti-PD1 achieve a greater killing effect. They further validated the combination efficacy in vivo in both partially anti-PD1 responder mouse model (MC38) and anti-PD1 resistant mouse model (CT26) [132].
A study by Sui et al. highlighted that an elevated Dickkopf 1 (DKK1) expression and an elevated concentration in serum was associated with recurrence, decreased CD8+ T-cell infiltration and poor response to PD-1 blockade in dMMR CRCs patients [133]. In their CRC organoid model, they validated the potential of DKK1 inhibition in combination with anti-PD1 with a further increase of apoptotic cells proportion when both were combined. Another small molecule-based drug Atractylenolide I, was identified through an CRC organoid model that showed promising MHC-I-mediated antigen presentation on CRC cells inducing a T cell infiltration and cytotoxicity which was strengthen the efficacy of anti-PD-1 in combination [134]. These studies extend the possible choice of therapeutic combinations for patient management. In these cases, organoid models are used for validation, as opposed to screening, which could yield a broader pool of new combinations.
A clinical study involving patients with early-stage CRC receiving neoadjuvant ICI have been used to evaluate the correlations between PDO-specific T cell reactivity and clinical responses to immunotherapy [135]. Most PDOs were generated from pre-treatment biopsy samples. However, for three clinical non-responders, post-treatment resection specimens were used as a source material. PDOs and clinical responses were only partially correlated, with reactivity seen for only three of six responders. One of the limitations in this study was that patients received the combination of an anti-PD-1 antibody and an anti-CTLA4 antibody, whereas only an anti-PD-1 antibody was included in the PDOs treatment. An alternative explanation for this lack of correlation might also be the absence of other important TME components. The only presence of reactive T cell doesn’t mimic the full spectrum of CMS, additional immune population like DCs or NK cells that are associated with a good response or Treg, MDSCs that are associated with poor response to immunotherapy can bring more valuable and relevant results [15, 136, 137].
For instance, in an enrichment model of HT29 spheroid co-cultured with T and NK cells from from healthy donors PBMCs, Coureau et al. showed that stimulating TILs and NK cells infiltrate induced an immune response capable of destroying tumor organoid structures. In their study they inhibited NKG2A a powerful inhibitory signal in both T and NK cells and the major histocompatibility complex-class I chain related proteins A and B (MICA/B) to avoid shedding on cancer cells and escape from NKG2D recognition by NK cells [138]. Their results highlight the powerful anti-tumor potential of IL-15-based treatments combined with immune modulatory anti-MICA/B and anti-NKG2A (Fig. 3) for cancer treatment for which co-cultured spheroids deeply help to characterize the efficacy and mode of action [139]. Although this study was performed in a spheroid model, similar experiments could very well be performed in this more complex model. With the enrichment of the PDOs model with myeloid-derived suppressor cells (MDSCs), Chen et al. showcased a critical role of those tumor-infiltrating MDSCs in resistance to PD-1 blockade in CRC. The authors demonstrated that a co-culture of CRC organoids expressing PD-L1 in the presence of MDSCs did not respond to anti-PD-1 therapy. However, when combining a simultaneous T cell activation and a depletion of MDSCs by IFN-α/β and TNF-α treatment (Fig. 3) in co-culture conditions, this led to an increased immunogenic organoids cell death following anti-PD-1 therapy [140].
In the last few years, the implication of myeloid compartment on immunotherapies response have been highlighted in several cancer types [137, 141,142,143,144], including CRC. This large population of myeloid cells are now considered as a clinical predictive biomarkers for ICIs [144]. Emerging strategies include combinatorial approaches with myeloid-targeting agents to improve ICIs response. A wide range of approaches is used to target this subsets, by either depleting them, preventing their recruitment, their differentiation and by inhibiting or reprogramming their immunosuppressive functions [142, 145]. However, their implication in primary or acquired resistance as well as the reciprocal interactions between tumor cells, dendritic cells or lymphocytes is still under investigation. For this purpose, co-culture of these cells in a complex 3D environment will provide further additional insight on the orchestration of their interactions and the resulting biological effect.
Other immune therapy-based combinations
Bispecific antibodies (BsAb) result of constant advances in engineering antibody field. The main benefit of BsAb is the dual targeting capability and the large number of combinations that can be generated [146]. The main benefit of BsAb is the dual targeting capability and the large number of combinations that can be generated [146]. A functional screening of more than 500 BsAbs on CRC PDOs has been done by Herpers et al., from the screening they identified MCLA-158 BsAb targeting EGFRxLGR5 that target highly mitotic leucine-rich repeat-containing G-protein-coupled receptor 5-positive (LGR5+) CSC population supporting organoid growth found in primary and metastatic tumors. This BsAb EGFRxLGR5 (Fig. 3) showed a higher efficacy demonstrated by lower IC50 on tumor PDOs and minimal toxicity through a higher IC50 on adjacent healthy mucosal tissue PDOs than Cetuximab. As well in vivo in an PDX orthotopic CRC model the BsAb EGFRxLGR5 showed a greater reduction of tumor size and weight compared to control [147]. Around 70% of CRC cases harbor multiple genetic alteration of the EGFR signaling pathway implicated in the resistance of targeted therapy such as anti-EGFR [148]. The combination strategies appears crucial to overcome either primary and acquired anti-EGFR therapy resistance [149]. Rau et al. demonstrated by using a newly developed bispecific EGFRxHER3-targeting antibody together with trastuzumab (Fig. 3) the efficacy of multiple targeting by CellTiterGlo® assays on two different CRC cell line spheroids and PDOs [150]. These two studies highlighted that a better understanding of resistance mechanism combined with computational engineering approaches, could therefore facilitate the future of combined drug development.
A new immunotherapy known as CEA-TCB, an IgG-based T-cell bispecific antibody (TCB) reroutes T cells to tumor cells that express the carcinoembryonic antigen (CEA) glycoprotein on their cell surface, regardless of the T cell receptor specificity of the redirected T cells. Cibisatamab contains two antigen-recognition sites: one for human CD3, a T-cell surface antigen, and one for human CEA, a tumor-associated antigen that is specifically expressed on certain tumor cells [151]. Gonzalez-Exposito et al. showed that the heterogeneity of CEA expression contributed to resistance to cibisatamab in the T cell and CRC PDOs co-culture systems [152]. By exploring the pathways regulating CEA expression the authors found that a combination of cibisatamab and the WNT/β-catenin inhibitors (porcupine inhibitor LGK-974 and tankyrase inhibitor compound 21) (Fig. 3) can increase the drug sensitivity to cibisatamab in PDOs established with metastatic chemotherapy-resistant CRC patient samples. Thus, it became clear that this co-culture model might be used for the new prognostic biomarkers, as well as an approach to improve immunotherapy sensitivity in clinical settings.
Targeting tumor metabolism to promote immunity
Another recognized hallmark of cancer that gain attention over the years is the tumor metabolism [68, 153]. Deregulation of metabolism has been shown to promote tumor progression [154], influence the tumor immune microenvironment [155] and induce drug resistance [156, 157]. Targeting metabolic pathways have shown to promote immunity by enhancing anti-tumor T cells, reducing immunosuppressive populations and thus enhance immunotherapy response [158].
One of the metabolic pathways investigated by Conche et al. is the ferroptosis, i.e.an iron-dependent cell death, which cancer cells can bypass [159, 160]. They used a specific CRC organoids model harboring ACP, Trp53, Tgfbr2, K-rasG12D(APTK) mutations, similarly to the human mesenchymal CRC subtype (CMS4). The authors transplanted those organoids in immunocompetent mice. In vivo, the combination of ferroptosis induction by Glutathione peroxidase 4 inhibition (Withaferin A) along with PD-1 and CXCR2 inhibitor (SB225002) a MDSC blocker effective for CRC liver metastasis inhibition (Fig. 3), whereas primary CRC tumor was resistant to this combinatorial treatment [161]. This study suggests that PDOs can be used to identify an additional combination therapy effective for advanced mCRC. Furthermore, the authors demonstrated that targeting tumor metabolism influences T cell activation and cytotoxicity toward liver metastasis.
In the CMS3 subtype of CRC, the presence of KRAS-mutant is associated with low immune infiltration [12, 13, 15]. Emerging evidence describes the glutaminolysis process promoted by KRAS-mutant CRC generating critical metabolites and epigenetic deregulation to support cancer cell proliferation, stemness and chemotherapy resistance [162]. Zhou et al. identified SLC25A22 a mediator implicated in the glutaminolysis process. They took benefit of CRC organoids with APC-KRAS mutations and colon-specific Slc25a22 knockout to inject them in immunocompetent mice. Similarly, they observed a reverse KRAS-mediated immunosuppressive phenotype hampering MDMCs recruitment, increasing cytotoxic T cell activation and finally synergy with anti-PD1 therapy [163].
As listed in Table 1 drug development and screening using immune co-culture models of PDOs are emerging bringing a wide range of new combination strategies. However limitations remain, the current co-culture models of PDOs fail to fully reproduce the original TME as in vivo system, limiting their application to target accessory immune and microenvironment cells. Some studies lack immune cell components cultured together in PDOs, others use non-autologous immune cells which could lead to unreliable prediction. Clinical application of combined immunotherapeutic regimen needs extensive trials and higher number of patient samples due to individual differences and the distinct roles of various immune cells in the immune system. Furthermore, toxicity of the novel combined therapies is not consistently addressed which could provide valuable insight to identify off-target effects and assess the safety profile for future clinical application.
Challenges and future perspectives
In the chapters above, we discussed advancements in the use of human patient-derived organoids in new combinatory treatments discovery for CRC. Even though organoids are more and more available in academic and industrial institutions, their use for the identification of combination strategies is relatively low. This can be explained by several factors.
Firstly, there is the scientific culture, which frequently translates results from drug candidates tested in simple 2D in vitro models directly to in vivo animal models. To implement PDOs model tests in predictive/drug discovery clinical standard of care, it necessitates neighboring clinical infrastructure, established collaborative network between scientists and clinical partners, such as surgeons, clinical pathologists or clinical oncologists as well as necessary ethical agreements. To be introduced in a clinical routine in addition to the tumor molecular profiling, PDOs generation need to be achieved in a clinical timeframe for patient care. CRC studies report a timeframe from around 2 weeks up to 53 weeks [19, 21, 164, 165] with a median time of 9 weeks [164] for the entire process. Standardization and automatization could reduce the timeline and variability for PDOs establishment and drug selection for patient benefit.
Another, more technical points are the standardized and reproducible protocols for maintenance, culturing/seeding, cryopreservation and treatment of organoids, especially those enriched with various cell types [166]. Not only organoids are known to be heterogenous between the patients, but the addition of other cells type requires an optimized cell culture medium to maintain the cells alive and enable their proliferation. The protocols should include human factor, i.e. possible sources of errors, experience, lab-to-lab equipment differences, that all may greatly influence the result of experiments. Quality control plan should be available with each validated protocol to secure the most reproducible and reliable outcome. In addition, the use of organoids technology in high-throughput drug-screening requires a standardized step of seeding, for the size of the organoids should be clinically relevant in order to obtain relevant and reproducible results [19]. Standardization of in vitro drug screening assays in organoids model is also to consider. There are multiple assays based on luminescence, colorimetry or fluorescence to assess the cell viability in PDOs. Even if the most used readout in the literature is the CellTiter-Glo®, that reflects cell viability based on the ATP quantification, other tests related to cytotoxicity are also used and should be considered or standardized using a decision tree based on the PDOs model and culture settings. Optimization of medium during co-culture appears essential for all cell types maintenance [167], as shown by Neal et al., by IL-2 addition for immune components maintenance viability [108]. The use of automated platforms in cell culture has widely emerged and allowed to perform high-throughput screenings in less time and effort. However, it comes with different type of challenges such as inconsistent volume measurements due to various viscosities of the solutions used, sample contamination, and even damage can result from poor pipetting techniques. This could lead to an accumulation of errors, inadequate data, poor reproducibility, and enhanced costs [168, 169].
As seen in the studies discussed above, only a few of them [19, 35, 52, 147] use simultaneously non-cancerous organoids (colon organoids) to evaluate safety of drug candidates. This could very well be due to the fact that the isolation and maintenance of colon organoids requires different conditions than CRC organoids, therefore makes the entire process even more complicated. It should be underlined however that the efficacy of combination therapy may come along with more important side effects [170]. Therefore, mathematical models are being developed alongside screening tests to evaluate drug-drug interactions, as well as supporting data for dose and drug selection to achieve synergistic combinations [19, 170, 171]. The recent development of multi-organ organoids is particularly useful in studying drug-induced liver, heart, kidney, gastrointestinal, and brain toxicities [172,173,174,175]. Ongoing and future advancements in organoid-based technology has the potential to provide more accurate and reliable models for drug safety profiling, ultimately leading to safer and more effective therapeutic strategies.
Optimization of combinatory approaches containing 3 or 4 drugs based on drug synergy remains rather rare. This is a consequence of the current development of methods for multi-drug combinations and the assessment of their interactions. It is known that anti-cancer drugs as single agents have high attrition rate, where only 5% of the drug-candidates make it to Phase III clinical trials [176,177,178]. Surprisingly, this rate has not improved in a decade despite the major advances in experimental research [179]. This might be due to the lack of relevant pre-clinical models that would recapitulate the complex and heterogenous nature of the human physiology. When it comes to drug combinations, the issue is not simpler, even though most drug candidates consisting of these drug mixtures are already used in the clinics. This is mainly due to a drug-drug interaction. This parameter is not always evaluated and depends highly on the complexity of the model used and changes from a model to another (i.e. PDOs vs. PDOs enriched with immune cells). This leads to a more challenging evaluation of the activity and toxicity of a drug combination, and therefore a more difficult translation from in vitro to in vivo. Other challenges for immunotherapy-based combinations are being studied as are the immune CAR-T or NK cell generation, constantly under development to reduce the production timeframe and improve their stability overtime, as well as the high cost of manufacturing that restrains their use in the clinics [180].
Cancer organoids do not present the complexity of living organism limiting the tests of several therapies, e.g. anti-angiogenic therapies or those that require in vivo metabolization [181, 182]. A crucial tumoral parameter is its heterogeneity in terms of phenotype, genotype or cell composition, which underscores the importance of using combination therapies. Building a complex co-cultured organoids model is a way to fill the gap between the primary tissue [108]. New technologies such as spatial transcriptomics and multiplex imaging have highlighted the significant impact of exploring the crosstalk between stromal, immune and cancer cells [183,184,185,186]. For example, Li et al. by mapping cell distribution of CRC patient samples, mouse bearing CRC tumor and co-culture model of CRC PDOs revealed the advantage of PDOs to investigate the functional interactions between CRC and TME [187]. Intricate interaction between ECs and immune cells have been described in tumor [188, 189], the investigation of such interactions in PDOs could enhance therapeutic strategies for CRC patients. The use of complex PDOs therefore could provide dynamic insight revealing the optimal drug administration regimen and the cells crosstalk in real-time leading to drug response or resistance.
Another aspect developed recently is the mimicking of the organ architecture. A mini-intestine organoid model developed by Lutolf et al., can conserve intestinal characteristics and survive couple of weeks with microfluidics perfusion [190]. This model could allow even more relevant and physiological interactions between various cells. Another study reports a successful addition of immune cells and microbiome into an Intestine-on-Chip model [191]. The combination of both chips would be an accurate representation of the in vivo situation to further study CRC without the necessity of actual in vivo models and compatible with high-throughput screening. Challenges related to the implementation of organoids in CRC research are listed in Table 2.
Finally, PDOs co-culture with various cell types, combined with multi-omics analysis, could provide comprehensive insights into tumor biology, heterogeneity, and therapeutic responses [90, 192]. The results can enhance the ability to identify novel pharmacological targets, diagnostic biomarkers as well as further investigation of mechanism of action or resistance for a better clinical translation [193].
Conclusion
CRC organoids are one of the earliest and well established pre-clinical models that has been widely used in translational research. Crucial research findings are consistently arising, and these organoid systems offer promising avenues for application. The ability to detect non-responders would save patients from the adverse side effects of an inadequate therapy, and PDOs from non-responders may benefit of a drug screening pipeline to uncover novel drug combination regimens. Challenges remain in the reproducibility and consistency of organoid culture, the lack of standardized processes, and the incomplete replication of the tumor microenvironment. Further validation and refinement of organoid-based approaches are warranted to maximize their clinical utility and impact on improving outcomes for CRC patients.
Data availability
All data generated or analysed during this study are included in this published article.
Abbreviations
- 2D:
-
Two-dimensional
- 3D:
-
Three-dimensional
- ALI:
-
Air-liquid interface
- CAFs:
-
Cancer-associated fibroblasts
- CAM:
-
Cell adhesion molecules
- CAR:
-
T-Chimeric antigen receptor-T
- CEA:
-
Carcinoembryonic antigen
- cDC2s:
-
Conventional DC type 2
- CK:
-
Cytokeratin
- CMS:
-
Consensus molecular subtype
- CRC:
-
Colorectal cancer
- CRIS:
-
A-CRC intrinsic subtype A
- DCs:
-
Dendritic cells
- DKK1:
-
Dickkopf 1
- ECs:
-
Endothelial cells
- EGFRvIII:
-
Epidermal growth factor receptor variant III
- EMT:
-
Epithelial-to-mesenchymal transition
- FZD:
-
FRIZZLED
- GM:
-
CSF-Granulocyte-macrophage colony-stimulating factor
- HUVEC:
-
Human umbilical vein endothelial cells
- ICIs:
-
Immune Checkpoint inhibitors
- ICAMs:
-
Intercellular adhesion molecules
- IPSC:
-
Induced pluripotent stem cells
- LGR5+:
-
Leucine-rich repeat-containing G-protein-coupled receptor 5-positive
- M-CSF:
-
Macrophage colony stimulating factor
- mCRC:
-
Metastatic colorectal cancer
- MDSCs:
-
Myeloid-derived suppressor cells
- MICA/B:
-
Major histocompatibility complex-class I chain related proteins A and B
- MMR:
-
Mismatch repair
- MSI-h:
-
Microsatellite instability-high
- MoDCs:
-
Monocyte-derived dendritic cells
- MMS:
-
Microsatellite stablility
- NK cell:
-
Natural Killer cell
- ODC:
-
Optimized drug combination
- PBMCs:
-
Peripheral blood mononuclear cells
- SIRT1:
-
Sirtuin 1
- TAM:
-
Tumor-associated macrophages
- TCB:
-
T-cell bispecific antibody
- PDOs:
-
Patient-derived organoids
- TECs:
-
Tumor endothelial cells
- TGMO:
-
Therapeutically guided multidrug optimization
- TME:
-
Tumor microenvironment
- VCAMs:
-
Vascular cell adhesion molecules
References
Hubrecht RC, Carter E. The 3Rs and Humane experimental technique: implementing change. Anim Open Access J MDPI. sept 2019;30(10):754.
Wadman M. FDA no longer has to require animal testing for new drugs. Sci 13 janv. 2023;379(6628):127–8.
Jensen C, Teng Y. Is it Time to start transitioning from 2D to 3D cell culture? Front Mol Biosci 6 mars 2020;7.
Ramzy GM, Koessler T, Ducrey E, McKee T, Ris F, Buchs N et al. Patient-Derived In Vitro Models for Drug Discovery in Colorectal Carcinoma. Cancers [Internet]. juin 2020 [cité 23 mai 2024];12(6). Disponible sur: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7352800/
Kapałczyńska M, Kolenda T, Przybyła W, Zajączkowska M, Teresiak A, Filas V, et al. 2D and 3D cell cultures – a comparison of different types of cancer cell cultures. Arch Med Sci [Internet]. 2016. https://doi.org/10.5114/aoms.2016.63743. [cité 23 mai 2024]; Disponible sur:. https://www.termedia.pl/doi/.
Sato T, Stange DE, Ferrante M, Vries RGJ, Van Es JH, Van den Brink S, et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterol Nov. 2011;141(5):1762–72.
Zhao Z, Chen X, Dowbaj AM, Sljukic A, Bratlie K, Lin L, et al. Organoids Nat Rev Methods Primer 1 déc. 2022;2(1):94.
Engel RM, Jardé T, Oliva K, Kerr G, Chan WH, Hlavca S, et al. Modeling colorectal cancer: a bio-resource of 50 patient-derived organoid lines. J Gastroenterol Hepatol Mai. 2022;37(5):898–907.
Lv T, Shen L, Xu X, Yao Y, Mu P, Zhang H, et al. Patient-derived tumor organoids predict responses to irinotecan‐based neoadjuvant chemoradiotherapy in patients with locally advanced rectal cancer. Int J Cancer févr. 2023;152(3):524–35.
Cho YW, Min DW, Kim HP, An Y, Kim S, Youk J, et al. Patient-derived organoids as a preclinical platform for precision medicine in colorectal cancer. Mol Oncol juin. 2022;16(12):2396–412.
Drost J, Clevers H. Organoids in cancer research. Nat Rev Cancer 24 Juill. 2018;18(7):407–18.
Picard E, Verschoor CP, Ma GW, Pawelec G. Relationships between Immune landscapes, genetic subtypes and responses to Immunotherapy in Colorectal Cancer. Front Immunol 6 mars. 2020;11:369.
Dienstmann R, Vermeulen L, Guinney J, Kopetz S, Tejpar S, Tabernero J. Consensus molecular subtypes and the evolution of precision medicine in colorectal cancer. Nat Rev Cancer févr. 2017;17(2):79–92.
Zheng Z, Wieder T, Mauerer B, Schäfer L, Kesselring R, Braumüller H. T cells in Colorectal Cancer: unravelling the function of different T cell subsets in the Tumor Microenvironment. Int J Mol Sci. 2023.
Roelands J, Kuppen P, Vermeulen L, Maccalli C, Decock J, Wang E, et al. Immunogenomic classification of Colorectal Cancer and therapeutic implications. Int J Mol Sci. oct 2017;24(10):2229.
He X, Jiang Y, Zhang L, Li Y, Hu X, Hua G et al. Patient-derived organoids as a platform for drug screening in metastatic colorectal cancer. Front Bioeng Biotechnol 22 mai 2023;11.
Wang T, Pan W, Zheng H, Zheng H, Wang Z, Li JJ, et al. Accuracy of using a patient-derived Tumor Organoid Culture Model to predict the response to Chemotherapy regimens in Stage IV Colorectal Cancer: a blinded study. Dis Colon Rectum 1 Juill. 2021;64(7):833–50.
Sakshaug BC, Folkesson E, Haukaas TH, Visnes T, Flobak Å. Systematic review: predictive value of organoids in colorectal cancer. Sci Rep 23 oct. 2023;13(1):18124.
Ramzy GM, Norkin M, Koessler T, Voirol L, Tihy M, Hany D, et al. Platform combining statistical modeling and patient-derived organoids to facilitate personalized treatment of colorectal carcinoma. J Exp Clin Cancer Res 3 avr. 2023;42(1):79.
Yao Y, Xu X, Yang L, Zhu J, Wan J, Shen L, et al. Patient-derived Organoids Predict Chemoradiation responses of locally advanced rectal Cancer. Cell Stem Cell 2 janv. 2020;26(1):17–e266.
Vlachogiannis G, Hedayat S, Vatsiou A, Jamin Y, Fernández-Mateos J, Khan K, et al. Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Sci 23 févr. 2018;359(6378):920–6.
Mo S, Tang P, Luo W, Zhang L, Li Y, Hu X, et al. Patient-derived Organoids from Colorectal Cancer with Paired Liver Metastasis reveal Tumor heterogeneity and predict response to Chemotherapy. Adv Sci Weinh Baden-Wurtt Ger Nov. 2022;9(31):e2204097.
Martini G, Belli V, Napolitano S, Ciaramella V, Ciardiello D, Belli A, et al. Establishment of patient-derived tumor organoids to functionally inform treatment decisions in metastatic colorectal cancer. ESMO Open juin. 2023;8(3):101198.
Ooft SN, Weeber F, Schipper L, Dijkstra KK, McLean CM, Kaing S, et al. Prospective experimental treatment of colorectal cancer patients based on organoid drug responses. ESMO Open juin. 2021;6(3):100103.
Bayat Mokhtari R, Homayouni TS, Baluch N, Morgatskaya E, Kumar S, Das B, et al. Combination therapy in combating cancer. Oncotarget 6 juin. 2017;8(23):38022–43.
Özdemir D, Büssgen M. Effectiveness and cost-effectiveness of combination therapy versus monotherapy in malignant melanoma. J Pharm Policy Pract 25 sept. 2023;16:106.
Antolin A, Workman P, Mestres J, Al-Lazikani B. Polypharmacology in Precision Oncology: current applications and future prospects. Curr Pharm Des 24 janv. 2017;22(46):6935–45.
Yang J, Kang H, Lyu L, Xiong W, Hu Y. A target map of clinical combination therapies in oncology: an analysis of clinicaltrials.gov. Discov Oncol 21 août. 2023;14(1):151.
Makhoba XH, Viegas C Jr, Mosa RA, Viegas FPD, Pooe OJ. Potential impact of the Multi-target Drug Approach in the treatment of some Complex diseases. Drug Des Devel Ther 11 août. 2020;14:3235–49.
Matsubara H, Miyoshi H, Kakizaki F, Morimoto T, Kawada K, Yamamoto T, et al. Efficacious combination Drug Treatment for Colorectal Cancer that overcomes resistance to KRAS G12C inhibitors. Mol Cancer Ther 3 avr. 2023;22(4):529–38.
Benson AB, Venook AP, Al-Hawary MM, Arain MA, Chen YJ, Ciombor KK, et al. Colon cancer, Version 2.2021, NCCN Clinical Practice guidelines in Oncology. J Natl Compr Canc Netw mars. 2021;19(3):329–59.
Weiss A, Nowak-Sliwinska P. Current trends in Multidrug optimization: an Alley of future successful treatment of Complex disorders. SLAS Technol juin. 2017;22(3):254–75.
Bahar ME, Kim HJ, Kim DR. Targeting the RAS/RAF/MAPK pathway for cancer therapy: from mechanism to clinical studies. Signal Transduct Target Ther 18 déc. 2023;8(1):1–38.
Shahi Thakuri P, Lamichhane A, Singh S, Gupta M, Luker GD, Tavana H. Modeling Adaptive Resistance of KRAS Mutant Colorectal Cancer to MAPK Pathway Inhibitors with a Three-Dimensional Tumor Model. ACS Pharmacol Transl Sci. 11 déc. 2020;3(6):1176–87.
Zoetemelk M, Ramzy GM, Rausch M, Koessler T, van Beijnum JR, Weiss A, et al. Optimized low-dose combinatorial drug treatment boosts selectivity and efficacy of colorectal carcinoma treatment. Mol Oncol 1 nov. 2020;14(11):2894–919.
Atanasova VS, Riedl A, Strobl M, Flandorfer J, Unterleuthner D, Weindorfer C et al. Selective Eradication of Colon Cancer Cells Harboring PI3K and/or MAPK Pathway Mutations in 3D Culture by Combined PI3K/AKT/mTOR Pathway and MEK Inhibition. Int J Mol Sci. 14 janv. 2023;24(2):1668.
Weiss A, Le Roux-Bourdieu M, Zoetemelk M, Ramzy GM, Rausch M, Harry D, et al. Identification of a synergistic multi-drug combination active in Cancer cells via the Prevention of Spindle Pole Clustering. Cancers. oct 2019;22(10):1612.
Thng DKH, Hooi L, Siew BE, Lee KY, Tan IJW, Lieske B et al. A functional personalised oncology approach against metastatic colorectal cancer in matched patient derived organoids. Npj Precis Oncol. 27 févr. 2024;8(1):52.
Bubna AK. Vorinostat—An overview. Indian J Dermatol. 2015;60(4):419.
Mertens S, Huismans MA, Verissimo CS, Ponsioen B, Overmeer R, Proost N, et al. Drug-repurposing screen on patient-derived organoids identifies therapy-induced vulnerability in KRAS-mutant colon cancer. Cell Rep 25 avr. 2023;42(4):112324.
Bitar L, Zouein J, Haddad FG, Eid R, Kourie HR. HER2 in metastatic colorectal cancer: a new to target to remember. Biomark Med févr. 2021;15(2):135–8.
Zeiser R, Andrlová H, Meiss F. Trametinib (GSK1120212). Recent Results Cancer Res Fortschr Krebsforsch Progres Dans Rech Sur Cancer. 2018;211:91–100.
Wang Q, Shen X, Chen G, Du J. Drug Resistance in Colorectal Cancer: From Mechanism to Clinic. Cancers. 14 juin. 2022;14(12):2928.
Guo Y, Wang M, Zou Y, Jin L, Zhao Z, Liu Q et al. Mechanisms of chemotherapeutic resistance and the application of targeted nanoparticles for enhanced chemotherapy in colorectal cancer. J Nanobiotechnology. 11 août. 2022;20(1):371.
Van der Jeught K, Xu HC, Li YJ, Lu XB, Ji G. Drug resistance and new therapies in colorectal cancer. World J Gastroenterol. sept 2018;14(34):3834–48.
Zoetemelk M, Ramzy GM, Rausch M, Nowak-Sliwinska P. Drug-drug interactions of Irinotecan, 5-Fluorouracil, Folinic Acid and Oxaliplatin and its activity in Colorectal Carcinoma Treatment. Molecules 4 juin. 2020;25(11):2614.
Ramzy GM, Boschung L, Koessler T, Delucinge-Vivier C, Docquier M, McKee TA et al. FOLFOXIRI Resistance induction and characterization in human colorectal Cancer cells. Cancers. 1 oct 2022;14(19).
Boos SL, Loevenich LP, Vosberg S, Engleitner T, Öllinger R, Kumbrink J, et al. Disease modeling on Tumor Organoids implicates AURKA as a therapeutic target in Liver metastatic colorectal Cancer. Cell Mol Gastroenterol Hepatol. 2022;13(2):517–40.
Liewer S, Huddleston A. Alisertib: a review of pharmacokinetics, efficacy and toxicity in patients with hematologic malignancies and solid tumors. Expert Opin Investig Drugs janv. 2018;27(1):105–12.
Usui T, Sakurai M, Umata K, Elbadawy M, Ohama T, Yamawaki H et al. Hedgehog signals mediate Anti-cancer Drug Resistance in three-Dimensional Primary Colorectal Cancer Organoid Culture. Int J Mol Sci 6 avr 2018;19(4).
Shen X, Zhang Y, Xu Z, Gao H, Feng W, Li W, et al. KLF5 inhibition overcomes oxaliplatin resistance in patient-derived colorectal cancer organoids by restoring apoptotic response. Cell Death Dis 5 avr. 2022;13(4):303.
Schnalzger TE, de Groot MH, Zhang C, Mosa MH, Michels BE, Röder J et al. 3D model for CAR-mediated cytotoxicity using patient-derived colorectal cancer organoids. EMBO J 17 juin 2019;38(12).
Fang H, Huang Y, Luo Y, Tang J, Yu M, Zhang Y, et al. SIRT1 induces the accumulation of TAMs at colorectal cancer tumor sites via the CXCR4/CXCL12 axis. Cell Immunol janv. 2022;371:104458.
Subtil B, Iyer KK, Poel D, Bakkerus L, Gorris MAJ, Escalona JC et al. Dendritic cell phenotype and function in a 3D co-culture model of patient-derived metastatic colorectal cancer organoids. Front Immunol 25 janv 2023;14.
Katopodi T, Petanidis S, Charalampidis C, Chatziprodromidou I, Eskitzis P, Tsavlis D, et al. Tumor-infiltrating dendritic cells: decisive roles in Cancer Immunosurveillance, Immunoediting, and Tumor T Cell Tolerance. Cells. oct 2022;11(20):3183.
Subtil B, van der Hoorn IAE, Cuenca-Escalona J, Becker AMD, Alvarez-Begue M, Iyer KK et al. cDC2 plasticity and acquisition of a DC3-like phenotype mediated by IL-6 and PGE2 in a patient-derived colorectal cancer organoids model. Eur J Immunol. n/a(n/a):2350891.
Wolf NK, Kissiov DU, Raulet DH. Roles of natural killer cells in immunity to cancer, and applications to immunotherapy. Nat Rev Immunol févr. 2023;23(2):90–105.
Sentman CL, Barber MA, Barber A, Zhang T. NK Cell receptors as Tools in Cancer Immunotherapy. Advances in Cancer Research [Internet]. Elsevier; 2006. pp. 249–92. Disponible sur:. https://linkinghub.elsevier.com/retrieve/pii/S0065230X06950076.
Moretta A, Marcenaro E, Parolini S, Ferlazzo G, Moretta L. NK cells at the interface between innate and adaptive immunity. Cell Death Differ févr. 2008;15(2):226–33.
Mujal AM, Delconte RB, Sun JC. Natural killer cells: from innate to adaptive features. Annu Rev Immunol 26 avr. 2021;39(1):417–47.
Bruni D, Angell HK, Galon J. The immune contexture and immunoscore in cancer prognosis and therapeutic efficacy. Nat Rev Cancer Nov. 2020;20(11):662–80.
Sconocchia G, Eppenberger S, Spagnoli GC, Tornillo L, Droeser R, Caratelli S, et al. NK cells and T cells cooperate during the clinical course of colorectal cancer. OncoImmunology 3 août. 2014;3(8):e952197.
Melaiu O, Lucarini V, Cifaldi L, Fruci D. Influence of the Tumor Microenvironment on NK cell function in solid tumors. Front Immunol 21 janv. 2020;10:3038.
Cheng M, Chen Y, Xiao W, Sun R, Tian Z. NK cell-based immunotherapy for malignant diseases. Cell Mol Immunol Mai. 2013;10(3):230–52.
Lanuza PM, Vigueras A, Olivan S, Prats AC, Costas S, Llamazares G, et al. Activated human primary NK cells efficiently kill colorectal cancer cells in 3D spheroid cultures irrespectively of the level of PD-L1 expression. OncoImmunology 3 avr. 2018;7(4):e1395123.
Neal JT, Li X, Zhu J, Giangarra V, Grzeskowiak CL, Ju J, et al. Organoid modeling of the Tumor Immune Microenvironment. Cell 13 déc. 2018;175(7):1972–e198816.
Zhao Y, Shen M, Wu L, Yang H, Yao Y, Yang Q, et al. Stromal cells in the tumor microenvironment: accomplices of tumor progression? Cell Death Dis 4 sept. 2023;14(9):587.
Hanahan D. Hallmarks of Cancer: New dimensions. Cancer Discov 1 janv. 2022;12(1):31–46.
Dudley AC, Griffioen AW. Pathological angiogenesis: mechanisms and therapeutic strategies. Angiogenesis août. 2023;26(3):313–47.
Liu ZL, Chen HH, Zheng LL, Sun LP, Shi L. Angiogenic signaling pathways and anti-angiogenic therapy for cancer. Signal Transduct Target Ther. 11 mai. 2023;8(1):198.
Van Beijnum JR, Huijbers EJM, Van Loon K, Blanas A, Akbari P, Roos A, et al. Extracellular vimentin mimics VEGF and is a target for anti-angiogenic immunotherapy. Nat Commun 23 mai. 2022;13(1):2842.
Griffioen AW, Damen CA, Martinotti S, Blijham GH, Groenewegen G. Endothelial intercellular adhesion molecule-1 expression is suppressed in human malignancies: the role of angiogenic factors. Cancer Res 1 mars. 1996;56(5):1111–7.
Nowak-Sliwinska P, Van Beijnum JR, Griffioen CJ, Huinen ZR, Sopesens NG, Schulz R, et al. Proinflammatory activity of VEGF-targeted treatment through reversal of tumor endothelial cell anergy. Angiogenesis mai. 2023;26(2):279–93.
Huinen ZR, Huijbers EJM, van Beijnum JR, Nowak-Sliwinska P, Griffioen AW. Anti-angiogenic agents — overcoming tumour endothelial cell anergy and improving immunotherapy outcomes. Nat Rev Clin Oncol 8 août. 2021;18(8):527–40.
Huijbers EJM, Khan KA, Kerbel RS, Griffioen AW. Tumors resurrect an embryonic vascular program to escape immunity. Sci Immunol 14 janv. 2022;7(67):eabm6388.
Strobel HA, Moss SM, Hoying JB. Vascularized Tissue Organoids Bioeng 17 janv. 2023;10(2):124.
Wang X, Bijonowski B, Kurniawan N. Vascularizing Organoids to Promote Long-Term Organogenesis on a chip. Organoids 7 déc. 2023;2(4):239–55.
Zhao X, Xu Z, Xiao L, Shi T, Xiao H, Wang Y, et al. Review on the vascularization of Organoids and Organoids-on-a-Chip. Front Bioeng Biotechnol 12 avr. 2021;9:637048.
Chung M, Ahn J, Son K, Kim S, Jeon NL. Biomimetic Model of Tumor Microenvironment on Microfluidic platform. Adv Healthc Mater août. 2017;6(15):1700196.
Biselli E, Agliari E, Barra A, Bertani FR, Gerardino A, De Ninno A, et al. Organs on chip approach: a tool to evaluate cancer -immune cells interactions. Sci Rep 6 oct. 2017;7(1):12737.
Homan KA, Gupta N, Kroll KT, Kolesky DB, Skylar-Scott M, Miyoshi T, et al. Flow-enhanced vascularization and maturation of kidney organoids in vitro. Nat Methods mars. 2019;16(3):255–62.
Nwokoye PN, Abilez OJ. Bioengineering methods for vascularizing organoids. Cell Rep Methods juin. 2024;4(6):100779.
Truelsen SLB, Mousavi N, Wei H, Harvey L, Stausholm R, Spillum E et al. The cancer angiogenesis co-culture assay: In vitro quantification of the angiogenic potential of tumoroids. PLOS ONE. 7 juill. 2021;16(7):e0253258.
Li J, Chen D, Shen M. Tumor Microenvironment shapes Colorectal Cancer Progression, Metastasis, and treatment responses. Front Med 23 mars 2022;9:869010.
Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM, et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer mars. 2020;20(3):174–86.
Kobayashi H, Enomoto A, Woods SL, Burt AD, Takahashi M, Worthley DL. Cancer-associated fibroblasts in gastrointestinal cancer. Nat Rev Gastroenterol Hepatol Mai. 2019;16(5):282–95.
Calon A, Lonardo E, Berenguer-Llergo A, Espinet E, Hernando-Momblona X, Iglesias M, et al. Stromal gene expression defines poor-prognosis subtypes in colorectal cancer. Nat Genet avr. 2015;47(4):320–9.
Guinney J, Dienstmann R, Wang X, de Reyniès A, Schlicker A, Soneson C, et al. The consensus molecular subtypes of colorectal cancer. Nat Med Nov. 2015;21(11):1350–6.
Xi Y, Xu P. Global colorectal cancer burden in 2020 and projections to 2040. Transl Oncol oct. 2021;14(10):101174.
Strating E, Verhagen MP, Wensink E, Dünnebach E, Wijler L, Aranguren I et al. Co-cultures of colon cancer cells and cancer-associated fibroblasts recapitulate the aggressive features of mesenchymal-like colon cancer. Front Immunol 16 mai 2023;14.
Luo X, Fong ELS, Zhu C, Lin QXX, Xiong M, Li A, et al. Hydrogel-based colorectal cancer organoid co-culture models. Acta Biomater Sept. 2021;132:461–72.
Zhu J, Ji L, Chen Y, Li H, Huang M, Dai Z, et al. Organoids and organs-on-chips: insights into predicting the efficacy of systemic treatment in colorectal cancer. Cell Death Discov 22 févr. 2023;9(1):72.
Strelez C, Chilakala S, Ghaffarian K, Lau R, Spiller E, Ung N, et al. Human colorectal cancer-on-chip model to study the microenvironmental influence on early metastatic spread. iScience mai. 2021;24(5):102509.
McAleer CW, Long CJ, Elbrecht D, Sasserath T, Bridges LR, Rumsey JW et al. Multi-organ system for the evaluation of efficacy and off-target toxicity of anticancer therapeutics. Sci Transl Med. 19 juin. 2019;11(497):eaav1386.
Jenkins RW, Aref AR, Lizotte PH, Ivanova E, Stinson S, Zhou CW, et al. Ex vivo profiling of PD-1 blockade using Organotypic Tumor spheroids. Cancer Discov 1 févr. 2018;8(2):196–215.
Vunjak-Novakovic G, Ronaldson-Bouchard K, Radisic M. Organs-on-a-chip models for biological research. Cell sept. 2021;184(18):4597–611.
Elomaa H. Prognostic significance of spatial and density analysis of T lymphocytes in colorectal cancer. Br J Cancer. 2022.
Wang Q, Shen X, An R, Bai J, Dong J, Cai H, et al. Peritumoral tertiary lymphoid structure and tumor stroma percentage predict the prognosis of patients with non-metastatic colorectal cancer. Front Immunol 16 sept. 2022;13:962056.
Posch F, Silina K, Leibl S, Mündlein A, Moch H, Siebenhüner A, et al. Maturation of tertiary lymphoid structures and recurrence of stage II and III colorectal cancer. OncoImmunology 1 févr. 2018;7(2):e1378844.
Bindea G, Mlecnik B, Tosolini M, Kirilovsky A, Waldner M, Obenauf AC, et al. Spatiotemporal Dynamics of Intratumoral Immune Cells Reveal the Immune Landscape in Human Cancer. Immun Oct. 2013;39(4):782–95.
Yang Z. The prognostic and clinicopathological value of tumor-associated macrophages in patients with colorectal cancer: a systematic review and meta-analysis. Int J Colorectal Dis. 2020.
Väyrynen JP, Haruki K, Lau MC, Väyrynen SA, Zhong R, Dias Costa A, et al. The Prognostic Role of Macrophage polarization in the Colorectal Cancer Microenvironment. Cancer Immunol Res 1 janv. 2021;9(1):8–19.
Homicsko K. Organoid technology and applications in cancer immunotherapy and precision medicine. Curr Opin Biotechnol oct. 2020;65:242–7.
Dijkstra KK, Cattaneo CM, Weeber F, Chalabi M, Van De Haar J, Fanchi LF, et al. Generation of Tumor-reactive T cells by co-culture of Peripheral Blood lymphocytes and Tumor Organoids. Cell sept. 2018;174(6):1586–e159812.
Cattaneo CM, Dijkstra KK, Fanchi LF, Kelderman S, Kaing S, Van Rooij N, et al. Tumor organoid–T-cell coculture systems. Nat Protoc Janv. 2020;15(1):15–39.
Votanopoulos KI, Forsythe S, Sivakumar H, Mazzocchi A, Aleman J, Miller L, et al. Model of patient-specific Immune-enhanced Organoids for Immunotherapy Screening: feasibility study. Ann Surg Oncol juin. 2020;27(6):1956–67.
Dekkers JF, Alieva M, Cleven A, Keramati F, Wezenaar AKL, van Vliet EJ, et al. Uncovering the mode of action of engineered T cells in patient cancer organoids. Nat Biotechnol Janv. 2023;41(1):60–9.
Neal JT, Li X, Zhu J, Giangarra V, Grzeskowiak CL, Ju J, et al. Organoid modeling of the Tumor Immune Microenvironment. Cell déc. 2018;175(7):1972–e198816.
Sterner RC, Sterner RM. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J 6 avr. 2021;11(4):69.
Dagar G, Gupta A, Masoodi T, Nisar S, Merhi M, Hashem S et al. Harnessing the potential of CAR-T cell therapy: progress, challenges, and future directions in hematological and solid tumor treatments. J Transl Med. 7 juill. 2023;21(1):449.
Akbari P, Huijbers EJM, Themeli M, Griffioen AW, Van Beijnum JR. The tumor vasculature an attractive CAR T cell target in solid tumors. Angiogenesis Nov. 2019;22(4):473–5.
Akbari P, Katsarou A, Daghighian R, Van Mil LWHG, Huijbers EJM, Griffioen AW, et al. Directing CAR T cells towards the tumor vasculature for the treatment of solid tumors. Biochim Biophys Acta BBA - Rev Cancer Mai. 2022;1877(3):188701.
Kennedy LB, Salama AKS. A review of cancer immunotherapy toxicity. CA Cancer J Clin mars. 2020;70(2):86–104.
Brudno JN, Kochenderfer JN. Recent advances in CAR T-cell toxicity: mechanisms, manifestations and management. Blood Rev mars. 2019;34:45–55.
Rojas-Quintero J, Díaz MP, Palmar J, Galan-Freyle NJ, Morillo V, Escalona D, et al. Car T cells in solid tumors: overcoming obstacles. Int J Mol Sci 10 avr. 2024;25(8):4170.
Ning RX, Liu CY, Wang SQ, Li WK, Kong X, He ZW. Application status and optimization suggestions of tumor organoids and CAR-T cell co-culture models. Cancer Cell Int 5 mars. 2024;24(1):98.
Jacobs J, Deschoolmeester V, Zwaenepoel K, Flieswasser T, Deben C, Van Den Bossche J, et al. Unveiling a CD70-positive subset of cancer-associated fibroblasts marked by pro-migratory activity and thriving regulatory T cell accumulation. OncoImmunology 3 Juill. 2018;7(7):e1440167.
Inoue S, Ito H, Tsunoda T, Murakami H, Ebi M, Ogasawara N, et al. CD70 expression in tumor-associated fibroblasts predicts worse survival in colorectal cancer patients. Virchows Arch Oct. 2019;475(4):425–34.
Van den Eynde A, Gehrcken L, Verhezen T, Lau HW, Hermans C, Lambrechts H, Flieswasser T, Quatannens D, Roex G, Zwaenepoel K, Marcq E, Philippe Joye, Edgar Cardenas De La Hoz, Christophe Deben, Alessia Gasparini, Pierre Montay-Gruel, Maxim Le Compte, Eva Lion, Filip Lardon, Steven, Jonas RM, Van Audenaerde. IL-15-secreting CAR natural killer cells directed toward the pan-cancer target CD70 eliminate both cancer cells and cancer-associated fibroblasts. 2024.
Wang H, Pan W. Challenges of chimeric antigen receptor-T/natural killer cell therapy in the treatment of solid tumors: focus on colorectal cancer and evaluation of combination therapies. Mol Cell Biochem Mai. 2023;478(5):967–80.
Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol août. 2015;15(8):486–99.
van der Leun AM, Thommen DS, Schumacher TN. CD8 + T cell states in human cancer: insights from single-cell analysis. Nat Rev Cancer avr. 2020;20(4):218–32.
Thommen DS, Koelzer VH, Herzig P, Roller A, Trefny M, Dimeloe S, et al. A transcriptionally and functionally distinct PD-1 + CD8 + T cell pool with predictive potential in non-small-cell lung cancer treated with PD-1 blockade. Nat Med Juill. 2018;24(7):994–1004.
Kim MJ, Ha SJ. Differential Role of PD-1 expressed by various Immune and Tumor cells in the Tumor Immune Microenvironment: expression, function, therapeutic efficacy, and resistance to Cancer Immunotherapy. Front Cell Dev Biol 22 Nov. 2021;9:767466.
Gordon SR, Maute RL, Dulken BW, Hutter G, George BM, McCracken MN, et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nat Mai. 2017;545(7655):495–9.
Lin KX, Istl AC, Quan D, Skaro A, Tang E, Zheng X. PD-1 and PD-L1 inhibitors in cold colorectal cancer: challenges and strategies. Cancer Immunol Immunother déc. 2023;72(12):3875–93.
Pauken KE, Torchia JA, Chaudhri A, Sharpe AH, Freeman GJ. Emerging concepts in PD-1 checkpoint biology. Semin Immunol févr. 2021;52:101480.
Research C, for DE. and. FDA grants nivolumab accelerated approval for MSI-H or dMMR colorectal cancer. FDA [Internet]. 2 sept 2019 [cité 16 avr 2024]; Disponible sur: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-nivolumab-accelerated-approval-msi-h-or-dmmr-colorectal-cancer
Research C, for DE. and. FDA grants accelerated approval to pembrolizumab for first tissue/site agnostic indication. FDA [Internet]. 2 sept 2019 [cité 16 avr 2024]; Disponible sur: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-pembrolizumab-first-tissuesite-agnostic-indication
Research C, for DE. and. FDA approves pembrolizumab for first-line treatment of MSI-H/dMMR colorectal cancer. FDA [Internet]. 30 juin 2020 [cité 16 avr 2024]; Disponible sur: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-pembrolizumab-first-line-treatment-msi-hdmmr-colorectal-cancer
Ganesh K, Stadler ZK, Cercek A, Mendelsohn RB, Shia J, Segal NH, et al. Immunotherapy in colorectal cancer: rationale, challenges and potential. Nat Rev Gastroenterol Hepatol juin. 2019;16(6):361–75.
Deng J, Wang ES, Jenkins RW, Li S, Dries R, Yates K, et al. CDK4/6 inhibition augments Antitumor Immunity by enhancing T-cell activation. Cancer Discov févr. 2018;8(2):216–33.
Sui Q, Liu D, Jiang W, Tang J, Kong L, Han K, et al. Dickkopf 1 impairs the tumor response to PD-1 blockade by inactivating CD8 + T cells in deficient mismatch repair colorectal cancer. J Immunother Cancer mars. 2021;9(3):e001498.
Xu H, Van Der Jeught K, Zhou Z, Zhang L, Yu T, Sun Y, et al. Atractylenolide I enhances responsiveness to immune checkpoint blockade therapy by activating tumor antigen presentation. J Clin Invest 17 mai. 2021;131(10):e146832.
Chalabi M, Fanchi LF, Dijkstra KK, Van Den Berg JG, Aalbers AG, Sikorska K, et al. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nat Med avr. 2020;26(4):566–76.
Zheng X, Gao Y, Yu C, Fan G, Li P, Zhang M, et al. Identification of immune-related subtypes of colorectal cancer to improve antitumor immunotherapy. Sci Rep. sept 2021;30(1):19432.
Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A. Primary, adaptive, and Acquired Resistance to Cancer Immunotherapy. Cell févr. 2017;168(4):707–23.
De Ferrari L, Kumar S, Luoma AM, Ito Y, Alves Da Silva PH, Pan D, et al. Inhibition of MICA and MICB Shedding elicits NK-Cell–mediated immunity against tumors resistant to cytotoxic T cells. Cancer Immunol Res 1 juin. 2020;8(6):769–80.
Courau T, Bonnereau J, Chicoteau J, Bottois H, Remark R, Assante Miranda L, et al. Cocultures of human colorectal tumor spheroids with immune cells reveal the therapeutic potential of MICA/B and NKG2A targeting for cancer treatment. J Immunother Cancer déc. 2019;7(1):74.
Chen J, Sun HW, Yang YY, Chen HT, Yu XJ, Wu WC et al. Reprogramming immunosuppressive myeloid cells by activated T cells promotes the response to anti-PD-1 therapy in colorectal cancer. Signal Transduct Target Ther. 8 janv. 2021;6(1):4.
Arlauckas SP, Garris CS, Kohler RH, Kitaoka M, Cuccarese MF, Yang KS et al. In vivo imaging reveals a tumor-associated macrophage–mediated resistance pathway in anti–PD-1 therapy. Sci Transl Med. 10 mai. 2017;9(389):eaal3604.
Pittet MJ, Michielin O, Migliorini D. Clinical relevance of tumour-associated macrophages. Nat Rev Clin Oncol juin. 2022;19(6):402–21.
Engblom C, Pfirschke C, Pittet MJ. The role of myeloid cells in cancer therapies. Nat Rev Cancer Juill. 2016;16(7):447–62.
Peranzoni E, Ingangi V, Masetto E, Pinton L, Marigo I. Myeloid cells as clinical biomarkers for Immune Checkpoint Blockade. Front Immunol 24 Juill. 2020;11:1590.
Johnson B. Targeting myeloid-derived suppressor cell trafficking as a Novel Immunotherapeutic Approach in microsatellite stable colorectal Cancer. Cancers 20 nov. 2023;15(22):5484.
Wei J, Yang Y, Wang G, Liu M. Current landscape and future directions of bispecific antibodies in cancer immunotherapy. Front Immunol. oct 2022;28:13:1035276.
Herpers B, Eppink B, James MI, Cortina C, Cañellas-Socias A, Boj SF, et al. Functional patient-derived organoid screenings identify MCLA-158 as a therapeutic EGFR × LGR5 bispecific antibody with efficacy in epithelial tumors. Nat Cancer 25 avr. 2022;3(4):418–36.
Misale S, Di Nicolantonio F, Sartore-Bianchi A, Siena S, Bardelli A. Resistance to Anti-EGFR Therapy in Colorectal Cancer: from heterogeneity to Convergent Evolution. Cancer Discov 1 nov. 2014;4(11):1269–80.
Dienstmann R, Salazar R, Tabernero J. Overcoming resistance to Anti-EGFR Therapy in Colorectal Cancer. Am Soc Clin Oncol Educ Book Mai 2015;(35):e149–56.
Rau A, Janssen N, Kühl L, Sell T, Kalmykova S, Mürdter TE, et al. Triple targeting of HER receptors overcomes heregulin-mediated resistance to EGFR Blockade in Colorectal Cancer. Mol Cancer Ther 4 mai. 2022;21(5):799–809.
Bacac M, Fauti T, Sam J, Colombetti S, Weinzierl T, Ouaret D, et al. A Novel Carcinoembryonic Antigen T-Cell bispecific antibody (CEA TCB) for the treatment of solid tumors. Clin Cancer Res 1 Juill. 2016;22(13):3286–97.
Gonzalez-Exposito R, Semiannikova M, Griffiths B, Khan K, Barber LJ, Woolston A, et al. CEA expression heterogeneity and plasticity confer resistance to the CEA-targeting bispecific immunotherapy antibody cibisatamab (CEA-TCB) in patient-derived colorectal cancer organoids. J Immunother Cancer déc. 2019;7(1):101.
Pavlova NN, Zhu J, Thompson CB. The hallmarks of cancer metabolism: still emerging. Cell Metab mars. 2022;34(3):355–77.
Cantor JR, Sabatini DM. Cancer Cell Metabolism: one Hallmark, many faces. Cancer Discov 1 oct. 2012;2(10):881–98.
Ramapriyan R, Caetano MS, Barsoumian HB, Mafra ACP, Zambalde EP, Menon H, et al. Altered cancer metabolism in mechanisms of immunotherapy resistance. Pharmacol Ther mars. 2019;195:162–71.
Bader JE, Voss K, Rathmell JC. Targeting metabolism to improve the Tumor Microenvironment for Cancer Immunotherapy. Mol Cell juin. 2020;78(6):1019–33.
Jiang Z, Hsu JL, Li Y, Hortobagyi GN, Hung MC. Cancer Cell Metabolism bolsters Immunotherapy Resistance by promoting an immunosuppressive Tumor Microenvironment. Front Oncol 22 Juill. 2020;10:1197.
Leone RD, Powell JD. Fueling the revolution: targeting metabolism to Enhance Immunotherapy. Cancer Immunol Res 1 mars. 2021;9(3):255–60.
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: An Iron-Dependent form of nonapoptotic cell death. Cell mai. 2012;149(5):1060–72.
Gong D, Chen M, Wang Y, Shi J, Hou Y. Role of ferroptosis on tumor progression and immunotherapy. Cell Death Discov. oct 2022;26(1):427.
Conche C, Finkelmeier F, Pešić M, Nicolas AM, Böttger TW, Kennel KB, et al. Combining ferroptosis induction with MDSC blockade renders primary tumours and metastases in liver sensitive to immune checkpoint blockade. Gut sept. 2023;72(9):1774–82.
Wong CC, Xu J, Bian X, Wu JL, Kang W, Qian Y, et al. In colorectal Cancer cells with mutant KRAS, SLC25A22-Mediated glutaminolysis reduces DNA demethylation to increase WNT signaling, stemness, and Drug Resistance. Gastroenterol déc. 2020;159(6):2163–e21806.
Zhou Q, Peng Y, Ji F, Chen H, Kang W, Chan LS, et al. Targeting of SLC25A22 boosts the immunotherapeutic response in KRAS-mutant colorectal cancer. Nat Commun 4 août. 2023;14(1):4677.
Tan T, Mouradov D, Lee M, Gard G, Hirokawa Y, Li S, et al. Unified framework for patient-derived, tumor-organoid-based predictive testing of standard-of-care therapies in metastatic colorectal cancer. Cell Rep Med déc. 2023;4(12):101335.
Jensen LH, Rogatto SR, Lindebjerg J, Havelund B, Abildgaard C, do Canto LM, et al. Precision medicine applied to metastatic colorectal cancer using tumor-derived organoids and in-vitro sensitivity testing: a phase 2, single-center, open-label, and non-comparative study. J Exp Clin Cancer Res 5 mai. 2023;42(1):115.
Papaccio F, Cabeza-Segura M, Garcia-Micò B, Tarazona N, Roda D, Castillo J, et al. Will Organoids fill the gap towards functional Precision Medicine? J Pers Med. nov 2022;21(11):1939.
Zhou C, Wu Y, Wang Z, Liu Y, Yu J, Wang W, et al. Standardization of organoid culture in cancer research. Cancer Med Juill. 2023;12(13):14375–86.
Daniszewski M, Crombie DE, Henderson R, Liang HH, Wong RCB, Hewitt AW, et al. Automated Cell Culture systems and their applications to human pluripotent stem cell studies. SLAS Technol août. 2018;23(4):315–25.
Louey A, Hernández D, Pébay A, Daniszewski M. Automation of organoid cultures: current protocols and applications. SLAS Discov oct. 2021;26(9):1138–47.
Küçükosmanoglu A, Scoarta S, Houweling M, Spinu N, Wijnands T, Geerdink N, et al. A real-world toxicity Atlas shows that adverse events of Combination therapies commonly result in additive interactions. Clin Cancer Res 15 avr. 2024;30(8):1685–95.
Ianevski A, Timonen S, Kononov A, Aittokallio T, Giri AK, SynToxProfiler. An interactive analysis of drug combination synergy, toxicity and efficacy. Marz M, éditeur. PLOS Comput Biol 3 févr. 2020;16(2):e1007604.
Matsui T, Shinozawa T. Human organoids for Predictive Toxicology Research and Drug Development. Front Genet 1 nov. 2021;12:767621.
Antón-Bolaños N, Faravelli I, Faits T, Andreadis S, Kastli R, Trattaro S, et al. Brain chimeroids reveal individual susceptibility to neurotoxic triggers. Nat 4 Juill. 2024;631(8019):142–9.
Zhang CJ, Meyer SR, O’Meara MJ, Huang S, Capeling MM, Ferrer-Torres D, et al. A human liver organoid screening platform for DILI risk prediction. J Hepatol Mai. 2023;78(5):998–1006.
Kim H, Im I, Jeon JS, Kang EH, Lee HA, Jo S, et al. Development of human pluripotent stem cell-derived hepatic organoids as an alternative model for drug safety assessment. Biomaterials Juill. 2022;286:121575.
Hutchinson L, Kirk R. High drug attrition rates—where are we going wrong? Nat Rev Clin Oncol avr. 2011;8(4):189–90.
Harrison RK. Phase II and phase III failures: 2013–2015. Nat Rev Drug Discov 4 Nov. 2016;15(12):817–8.
Waring MJ, Arrowsmith J, Leach AR, Leeson PD, Mandrell S, Owen RM, et al. An analysis of the attrition of drug candidates from four major pharmaceutical companies. Nat Rev Drug Discov Juill. 2015;14(7):475–86.
Jentzsch V, Osipenko L, Scannell JW, Hickman JA. Costs and causes of Oncology Drug Attrition with the example of insulin-like Growth Factor-1 receptor inhibitors. JAMA Netw Open 28 Juill. 2023;6(7):e2324977.
Mitra A, Barua A, Huang L, Ganguly S, Feng Q, He B. From bench to bedside: the history and progress of CAR T cell therapy. Front Immunol 15 mai. 2023;14:1188049.
Fior R, Póvoa V, Mendes RV, Carvalho T, Gomes A, Figueiredo N et al. Single-cell functional and chemosensitive profiling of combinatorial colorectal therapy in zebrafish xenografts. Proc Natl Acad Sci [Internet] 26 sept 2017 [cité 15 juill 2024];114(39). Disponible sur: https://doi.org/10.1073/pnas.1618389114
Haldi M, Ton C, Seng WL, McGrath P. Human melanoma cells transplanted into zebrafish proliferate, migrate, produce melanin, form masses and stimulate angiogenesis in zebrafish. Angiogenesis 26 oct. 2006;9(3):139–51.
Peng Z, Ye M, Ding H, Feng Z, Hu K. Spatial transcriptomics atlas reveals the crosstalk between cancer-associated fibroblasts and tumor microenvironment components in colorectal cancer. J Transl Med déc. 2022;20(1):302.
Xiao J, Yu X, Meng F, Zhang Y, Zhou W, Ren Y, et al. Integrating spatial and single-cell transcriptomics reveals tumor heterogeneity and intercellular networks in colorectal cancer. Cell Death Dis 10 mai. 2024;15(5):326.
Lin JR, Wang S, Coy S, Chen YA, Yapp C, Tyler M, et al. Multiplexed 3D atlas of state transitions and immune interaction in colorectal cancer. Cell janv. 2023;186(2):363–e38119.
Allam M, Cai S, Coskun AF. Multiplex bioimaging of single-cell spatial profiles for precision cancer diagnostics and therapeutics. Npj Precis Oncol 1 mai. 2020;4(1):11.
Li N, Zhu Q, Tian Y, Ahn KJ, Wang X, Cramer Z, et al. Mapping and modeling human colorectal carcinoma interactions with the tumor microenvironment. Nat Commun 30 nov. 2023;14(1):7915.
Fang J, Lu Y, Zheng J, Jiang X, Shen H, Shang X, et al. Exploring the crosstalk between endothelial cells, immune cells, and immune checkpoints in the tumor microenvironment: new insights and therapeutic implications. Cell Death Dis 4 sept. 2023;14(9):586.
Xu Y, Miller CP, Tykodi SS, Akilesh S, Warren EH. Signaling crosstalk between tumor endothelial cells and immune cells in the microenvironment of solid tumors. Front Cell Dev Biol 25 avr. 2024;12:1387198.
Nikolaev M, Mitrofanova O, Broguiere N, Geraldo S, Dutta D, Tabata Y, et al. Homeostatic mini-intestines through scaffold-guided organoid morphogenesis. Nat 24 sept. 2020;585(7826):574–8.
Kim HJ, Li H, Collins JJ, Ingber DE. Contributions of microbiome and mechanical deformation to intestinal bacterial overgrowth and inflammation in a human gut-on-a-chip. Proc Natl Acad Sci U S 5 janv. 2016;113(1):E7–15.
Papaccio F, García-Mico B, Gimeno-Valiente F, Cabeza-Segura M, Gambardella V, Gutiérrez-Bravo MF, et al. Proteotranscriptomic analysis of advanced colorectal cancer patient derived organoids for drug sensitivity prediction. J Exp Clin Cancer Res 6 janv. 2023;42(1):8.
Xiang D, He A, Zhou R, Wang Y, Xiao X, Gong T, et al. Building consensus on the application of organoid-based drug sensitivity testing in cancer precision medicine and drug development. Theranostics. 2024;14(8):3300–16.
Acknowledgements
The authors acknowledge our lab members and Prof. Patrick Meraldi for constructive discussions.
Funding
This study was supported by the Swiss National Science Foundation (310030_197878 to P.N.-S.)
Open access funding provided by University of Geneva
Author information
Authors and Affiliations
Contributions
VB, GMR, SJ and PN-S wrote the original draft and the revised manuscript. VB, GMR and PN-S contributed to the conceptualization. VB and PN-S contributed to the supervision. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors agreed to the publication of the article to the journal.
Competing interests
P.N.-S. is inventor of a patent of drug combination optimization methods. The other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Benboubker, V., Ramzy, G.M., Jacobs, S. et al. Challenges in validation of combination treatment strategies for CRC using patient-derived organoids. J Exp Clin Cancer Res 43, 259 (2024). https://doi.org/10.1186/s13046-024-03173-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13046-024-03173-x