Abstract
Background
The endostyle is an epithelial exocrine gland found in non-vertebrate chordates (amphioxi and tunicates) and the larvae of modern lampreys. It is generally considered to be an evolutionary precursor of the thyroid gland of vertebrates. Transformation of the endostyle into the thyroid gland during the metamorphosis of lampreys is thus deemed to be a recapitulation of a past event in vertebrate evolution. In 1906, Stockard reported that the thyroid gland in hagfish, the sister cyclostome group of lampreys, develops through an endostyle-like primordium, strongly supporting the plesiomorphy of the lamprey endostyle. However, the findings in hagfish thyroid development were solely based on this single study, and these have not been confirmed by modern molecular, genetic, and morphological data pertaining to hagfish thyroid development over the last century.
Results
Here, we showed that the thyroid gland of hagfish undergoes direct development from the ventrorostral pharyngeal endoderm, where the previously described endostyle-like primordium was not found. The developmental pattern of the hagfish thyroid, including histological features and regulatory gene expression profiles, closely resembles that found in modern jawed vertebrates (gnathostomes). Meanwhile, as opposed to gnathostomes but similar to non-vertebrate chordates, lamprey and hagfish share a broad expression domain of Nkx2-1/2-4, a key regulatory gene, in the pharyngeal epithelium during early developmental stages.
Conclusions
Based on the direct development of the thyroid gland both in hagfish and gnathostomes, and the shared expression profile of thyroid-related transcription factors in the cyclostomes, we challenge the plesiomorphic status of the lamprey endostyle and propose an alternative hypothesis where the lamprey endostyle could be obtained secondarily in crown lampreys.
Similar content being viewed by others
Background
The endostyle or thyroid is a defining characteristic of chordates [1,2,3] (Additional file 1: Fig. S1) [4,5,6,7]. The thyroid gland is a major endocrine organ that produces and secretes thyroid hormones (i.e., triiodothyronine and thyroxine) in all vertebrates. The thyroid hormones exert important pleiotropic effects on development, growth, energy metabolism, and metamorphosis. Although the general anatomy of the vertebrate thyroid gland is highly diverse (Additional file 1: Fig. S1A), the thyroid follicle is a common functional unit and an evolutionary novelty of vertebrates [3]. Acquisition of the follicular thyroid gland enables the efficient uptake and storage of iodine, which is notoriously scarce in freshwater environments, likely contributing to the early radiation of aquatic vertebrates [8].
The endostyle is the postulated evolutionary precursor of the vertebrate thyroid, occurring in filter-feeding non-vertebrate chordates in an anatomical position similar to that of the thyroid [8,9,10] (Additional file 1: Fig. S1B). The evolutionary link between the thyroid and the endostyle is supported by many shared morphological, physiological, and developmental traits including accumulation of iodide and peroxidase activity [8, 11], endodermal origin [9], and gene expression profiles [12, 13]. All extant non-vertebrate chordates are microphagous suspension feeders that secrete mucus from the endostyle to capture small particles, such as phytoplankton or detritus. The endostyle is also found in the filter-feeding ammocoete larvae of modern lampreys, and a portion of the epithelial cells of the ammocoete endostyle re-differentiates and transforms into thyroid follicles during metamorphosis [14] (Fig. 1A). Thus, the lamprey endostyle was considered to represent a state of non-vertebrate chordates [1, 8].
Transition of the endostyle to the thyroid in lampreys and classical evolutionary hypothesis of the vertebrate thyroid gland. A Larval lampreys (lateral view) possess an exocrine endostyle (en), which transforms into thyroid follicles (thy) in the adult (ventral view). p, pharynx; ht, heart; or, orifice; bb, branchial basket; va, ventral aorta; hbm, hypobranchial muscle; tm, tongue muscle. Bars, 100 μm. B Classical evolutionary hypothesis of the relationship between the endostyle and the thyroid based on the previous studies. Although an endostyle-like thyroid primordium was observed in developing hagfish (Eptatretus stoutii) embryos in 1906, it has not been verified ever since [15]. The primitive endostyle has been retained only in modern lampreys but was independently lost in the gnathostome and possibly in hagfish.
Considering the monophyly of cyclostomes [16, 17], further studies on the development of the thyroid in hagfish, the other group of extant cyclostomes, may enhance our knowledge regarding the evolution of the vertebrate thyroid and endostyle by providing a clearer understanding of the last common ancestor (LCA) of vertebrates. In contrast to the many developmental studies in lampreys, the only study on hagfish thyroid development was conducted by Stockard [15], who used Eptatretus stoutii (formerly Bdellostoma stouti) embryos. He described that the thyroid first appeared as a longitudinal “trough” in the pharyngeal floor [15] (Additional file 1: Fig. S2A-D). The presence of this structure, which was described as a rudiment of the endostyle almost a century later [10], and the broad pharyngeal origin of hagfish thyroid follicles would be consistent with the classical assumption that considers the endostyle a plesiomorphic trait retained in cyclostomes (Fig. 1B). However, mainly owing to the difficulty in obtaining and maintaining fertilized hagfish eggs [18], no molecular, genetic, or morphological data pertaining to hagfish thyroid development have been updated over the last century.
In gnathostomes, the shared developmental origin of the thyroid is an endodermal thickening at the level of the first pharyngeal pouch close to the endothelial lining of the anterior end of the fused endocardial tubes (aortic sac) [3, 19]. Loss-of-function studies in mice and zebrafish have revealed several transcription factors that are crucial for thyroid development. Among them, Nkx2-1 (previously termed thyroid transcription factor-1), Pax8, and Hhex form a mutually dependent gene regulatory network (GRN) [19, 20]. Although these transcription factors are not required for thyroid specification, the deletion of any of these resulted in athyreosis or severe thyroid hypoplasia, suggesting that the formation of the GRN is essential for the thyroid organogenesis [19, 21]. Foxe1 (previously termed thyroid transcription factor-2), downstream of the GRN, is also known to be essential for thyroid development in mammals, whereas the thyroid develops normally (with no changes in its morphology) in knockdown experiments in zebrafish embryos injected with a Foxe1 morpholino [22]. This suggests that the regulatory roles of Foxe1 in thyroid development may not be conserved among gnathostomes [19].
Here, we observed staged embryos of the Japanese inshore hagfish (Eptatretus burgeri), provided an updated description of thyroid development in detail, and deduced the direct development of the thyroid as in gnathostomes. Furthermore, to compare molecular developmental signatures between the development of thyroids in hagfish and gnathostome and that of the endostyle in lamprey, we examined expression profiles of orthologs of the abovementioned thyroid transcription factor genes (Nkx2-1, Pax8, and Hhex) during the development of E. burgeri, the cloudy catshark (Scyliorhinus torazame), and the Arctic lamprey (Lethenteron camtschaticum).
Results and discussion
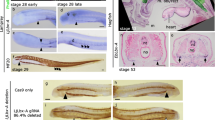
Contrary to previous observations of E. stoutii, we did not observe a trough-like structure during any of the developmental stages of E. burgeri (Fig. 2 and Additional file 1: Fig. S2E-G). Instead, we found a putative thyroid anlage in the form of a thickened epithelium on the pharyngeal floor juxtaposed to the aortic sac during the early pharyngula stage [17, 23] (at stage 45; Fig. 2A–C’), which is considerably earlier than the stage wherein the trough-like structure was observed by Stockard (at stage 53) [15]. Since the ventral pharynx of the hagfish embryo is rostroventrally everted in early development, the position of the hagfish thyroid anlage is difficult to identify based on the level of the pharyngeal arches [10]. Nevertheless, this thyroid anlage arose from a focal region of the pharyngeal floor slightly posterior to the root of the lower lip anlage and did not extend throughout the entire gill region as previously reported [15] (Fig. 2B, B’, C). The collection of hagfish embryos prepared by Bashford Dean, which was subsequently used in Stockard’s histological studies, showed a frequent distortion of embryonic tissues [24, 25]. The trough-like organ described by Stockard was not a consistent observation; thus, it was likely an artifact due to flawed fixation.
Development of the thyroid gland in the Eptatretus burgeri embryo. Dorsal (A, D) and lateral views (G) of fixed embryos immersed in methyl benzoate prior to paraffin embedding. B, B’, E, E’, H, H’ Midline sagittal sections of the hagfish thyroid. B, B’ Sections were stained using hematoxylin and eosin. E, E’, H, H’ Sections including thyroid primordia were stained via in situ hybridization of Tbx1/10A (E, E’) and Lhx3/4A (H, H’) with nuclear fast red in a previous study [17]. The area surrounded by dashed lines indicates thyroid primordium. Lateral (C, F, I) and frontal (C’, F’, I’) views of 3D reconstructed images. The ectoderm and endoderm are colored light blue and yellow, respectively. Dashed lines indicate the boundary between stomodeum and pharynx. Thyroid is colored red. Both cardiac field and blood vessel are transparently colored light red. pp, pharyngeal pouch; ect, ectoderm; nc, notochord; ot, otic vesicle; end, endoderm; heart, ht; ventral aorta, va; bs, bifurcation site of ventral aorta. See Fig. 1 for other abbreviations. Bars, 1 mm (B, E, H) and 200 μm (B’, E’, H’)
The position of the thyroid primordium in the hagfish embryo from stages 45 to 51 was continuously associated with a bifurcation site of the ventral aorta probably including the precardiac mesoderm (Fig. 2C’, F’, I’). In gnathostomes, the thyroid develops from an endodermal thickening of the pharyngeal floor in close vicinity to the aortic sac [3, 19], as was observed in the cloudy catshark (Scyliorhinus torazame; Fig. 3X; Additional file 1: Fig. S3A’-E’). Furthermore, in mice, frogs, and zebrafish, extrinsic factors, such as FGF2 and BMP4, presumably secreted from the precardiac mesoderm, have been shown to induce thyroid fate [19, 26]. Unlike the initiation of the thyroid development in hagfish and gnathostomes, evagination of the lamprey endostyle primordium from the midline pharyngeal endoderm occurs from the 2nd to 4th pharyngeal arches, and only the posterior end of the endostyle is in contact with the bifurcation site of the aortic sac [2] (Fig. 3W). Thus, the comparable topological relationships of the developing thyroid and the precardiac mesoderm, a possible specifier of thyroid origins, between hagfish and gnathostomes implies that the direct development of the thyroid gland may represent an ancestral condition in the LCA of modern vertebrates.
Expression profiles of the genes involved in thyroid development in modern cyclostomes. A–I and J–U’ hagfish and lamprey embryos, respectively. V, W, X schematic of the Nkx2-1/2-4 expressing domain (colored red) in hagfish, lamprey, and catshark embryos when the expression of Nkx2-1/2-4 first detected in this study (stages 45, 23, and 24, respectively). A’, D’, G’, K’, L’, S’, T’ Filled and open arrowheads indicate the presence and absence of signals, respectively. K’, L’, M’, O’, P’, Q’, S’, T’, and U’ Transverse sections of hybridized embryos in the plane of the dashed lines. M’, Q’, U’ Diagram of gene expression in the endostyle is shown in the boxes. as, aortic sac; opm, vestigial oropharyngeal membrane; MHB, midbrain-hindbrain boundary; ma, mandibular arch; liv, liver; hyp, hypothalamus; ve, velum; pn, pronephros. See Figs. 1 and 2 for other abbreviations. Bars, 200 μm
In hagfish, we found that the pharyngeal region co-expressing thyroid marker genes (Nkx2-1/2-4, Pax2/5/8, and Hhex) was confined to the thyroid anlage in stage 51 and 53 embryos (Fig. 3B, C, E, F, H, I), which is similar to the expression patterns observed in gnathostomes [19, 20] (Additional file 1: Fig. S3F-J). Co-expression of Nkx2-1/2-4, Pax2/5/8, and Hhex in hagfish and gnathostomes suggests that the conserved GRN for thyroid follicle formation was present in the LCA of modern vertebrates. However, unlike gnathostomes, in which Nkx2-1, Pax8, and Hhex are co-expressed in the earliest thyroid anlage [19], only Nkx2-1/2-4 was detected in the early primordia of the hagfish thyroid (stage 45; Fig. 3A’, D’, G’). Moreover, its expression domain extended rostrally towards the endodermal epithelium, up to the level of the oropharyngeal membrane, and was not confined to the prospective thyroid anlage (Fig. 3A, A’, V). In L. camtschaticum at Tahara’s stage 24 [27], the endostyle diverticulum was distinguishable by the broad medial expression of Nkx2-1/2-4A and Nkx2-1/2-4C genes in the pharynx (Fig. 3K, K’ and Additional file 1: Fig. S4J, J’), as previously reported [1, 2, 25]. Similar to the expression patterns in the hagfish, only the hybridized signal of Nkx2-1/2-4 paralogues was detected in the ventral pharynx of the lamprey before the initiation of endostyle formation (Tahara’s stage 23; Fig. 3J and Additional file 1: Fig. S4I). The spatiotemporal expression patterns of Nkx2-1/2-4 shared by hagfish and lamprey are similar to those of orthologous genes during the endostyle development in non-vertebrate chordates [13]. Furthermore, even in the hemichordate embryo which does not have an endostyle, the expression of Nkx2-1/2-4 is evident in the pharyngeal endoderm [28]. Nkx2-1 is focally expressed in the laryngotracheal diverticulum, in addition to the thyroid primordium; the laryngotracheal diverticulum is situated in the most caudal part of the pharynx during early development in mammals and birds [29, 30]. However, the lung and its homolog (the swim bladder) are presumed to be a synapomorphy of Osteichthyes (bony vertebrates), and thus, the expression domain of Nkx2-1 in these respiratory organs was likely acquired after Osteichthyes and Chondrichthyes split from each other and likely does not reflect an ancestral condition of gnathostomes. Thus, our findings suggest that the broad expression of Nkx2-1/2-4 orthologs in the ventral pharyngeal endoderm during the earliest stages of thyroid/endostyle development represents a plesiomorphic trait of chordates, which has been retained in cyclostomes, and may have been secondarily modified in crown gnathostomes where the Nkx2-1/2-4 expression domain was not broad and regionally confined to the thyroid primordium [19].
We identified an additional Pax2/5/8 paralogue in cyclostomes, termed Pax2/5/8B (Additional file 1: Fig. S4, S5). Expression signals of the lamprey Pax2/5/8A and Pax2/5/8B were not detected in the endostyle before stage 25 but were evident at stage 26 when the endostyle was almost differentiated (Fig. 3N–Q’ and Additional file 1: Fig. S4M-P). At stage 24, Hhex was expressed bilaterally at both ends of the diverticulum (Fig. 3S’). All thyroid marker genes were expressed in the endostyle of lamprey larvae only after stage 26 (Fig. 3M, Q, and U and Additional file 1: Fig. S4H, L, P), which were consistent with the previous studies [25, 31], but the expression of Pax2/5/8A and Hhex did not extend to the entire endostyle anlage in cross-sectional view (Fig. 3Q’, U’). The distributions of thyroid-related genes including Pax2/5/8 and Nkx2-1/2-4 orthologs in adult amphioxi and tunicates similarly do not extend to the entire endostyle in cross-sectional view. For example, the Nkx2-1/2-4 ortholog is not expressed in the thyroid-equivalent elements in the ascidian Ciona intestinalis, and the Pax2/5/8 ortholog is not expressed in part of the supporting elements in the cephalochordate Branchiostoma belcheri [12]. As co-expression of Nkx2-1/2-4, Pax2/5/8, and Hhex and their interactions are essential for the development of the thyroid gland in gnathostomes [19], the heterochronic, and heterogeneous expression of Nkx2-1/2-4, Pax2/5/8, and Hhex in the lamprey endostyle suggests that the core set of thyroid transcription factors, except for Nkx2-1/2-4, is not required for the endostyle development with regard to their organogenesis.
The expression of Nkx2-1/2-4, Pax2/5/8, and Hhex [32] orthologs in the endostyle of chordates, including the modern lampreys, may be responsible for thyroidal functions but not for its development. In the mammalian thyroid, Nkx2-1 and Pax8 genes are known to be involved both in organogenesis and physiological function [19, 21]. Thyroglobulin (Tg) and thyroid peroxidase (TPO) play critical roles in thyroid hormone synthesis, and the gene expression of Tg and TPO are coordinately regulated by Nkx2-1 and Pax8 in the thyroid [21, 33]. Likewise, TPO orthologs are co-expressed with Nkx2-1 and Pax8 in the endostyle of amphioxus and with Pax8 in that of tunicates, suggesting that these regulatory genes for thyroidal function are conserved among modern chordates [12, 13]. Thus, the regionally confined expression patterns of Nkx2-1/2-4, Pax2/5/8, and Hhex in the endostyles of the lamprey and non-vertebrate chordates imply that these transcription factors do not orchestrate a regulatory network that forms either the endostyle or thyroid follicle. Indeed, the thyroid follicles never occur prior to metamorphosis in the modern lampreys and throughout life in non-vertebrate chordates. In other words, our findings suggest that the developmental GRNs for the endostyle and thyroid organogenesis have an independent and distinct modular nature.
The life histories of hagfish and lamprey differ conspicuously. While all modern lampreys go through a larval phase for approximately 2–8 years and reach maturity only after metamorphosis [34], modern hagfishes develop into the adult morphology directly after hatching [18]. Recently, Miyashita et al. reported that fossil stem lampreys probably lacked the filter-feeding larval phase, since fossils of yolk-sac lampreys were found with prominent eyes and a circumoral feeding apparatus [35]. The fossil evidence suggested that they may have directly developed into macrophagous predators following the yolk-dependent lecithotrophic period. Although the presence of a rudimentary endostyle in these stem lampreys cannot be ruled out, these observations cast doubt on the prevailing view that the ammocoete larva best resembles the earliest vertebrates [36, 37]. In the context of thyroid evolution, the lack of a larval phase in stem lampreys suggests an alternative hypothesis, namely, that the primitive endostyle (which is retained in modern non-vertebrate chordates) was lost in the LCA of vertebrates, and the endostyle present in modern lampreys represents secondary acquisition.
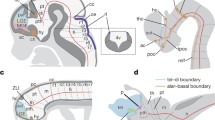
In the present study, we found that both gnathostomes and hagfish directly developed a thyroid. There is currently no evidence that ancestral species possessed an endostyle in the branches leading to these taxa. Considering the direct development (without metamorphosis) of stem lampreys [35], our findings further raise the possibility that the endostyle was acquired secondarily in crown lampreys. Thus, we propose the following alternative evolutionary scenario for the vertebrate thyroid and endostyle (Fig. 4). The broad expression of Nkx2-1/2-4 in the pharyngeal endoderm shared among extant cyclostomes, non-vertebrate chordates, and hemichordates represents a primitive trait of deuterostomes, and the expression domain was restricted to the ventral side of the pharynx in early chordates. A GRN involving unidentified transcription factors, instead of Pax2/5/8 and Hhex, which induces the formation of the primitive endostyle, may have first occurred downstream of Nkx2-1/2-4 (Fig. 4A). Before the acquisition of the follicular thyroid, Pax2/5/8 and Hhex orthologs were probably not coupled with Nkx2-1/2-4 and thus not involved in the endostyle or thyroid GRN, as observed in extant non-vertebrate chordates [12] (Fig. 4A). Subsequently, Nkx2-1/2-4, Pax2/5/8, and Hhex were organized in a colocalized expression domain, thereby establishing an interactive GRN to produce thyroid follicles in the earliest vertebrates (Fig. 4B). The acquisition of the thyroid and the subsequent loss of primitive endostyle occurred prior to the divergence of gnathostome and cyclostome lineages (Fig. 4C). An endostyle may have evolved secondarily at the base of crown lampreys in association with the filter-feeding larval stage. The cyclostome-specific (probably ancestral) broad and early expression domain of Nkx2-1/2-4 in the pharyngeal floor (Fig. 4D) may have facilitated the co-option of an ancestral endostyle GRN.
New alternative evolutionary scenario for the endostyle and thyroid gland. A–D Hypothetical independent evolutionary events that occurred during chordate evolution. Arrows indicate developmental sequence. A Acquisition of the primitive endostyle, which originated from the pharyngeal endoderm with broad expression of Nkx2-1/2-4 and subordinate endostyle GRN. B Thyroid follicles arise from a subset of epithelial cells in the endostyle with the acquisition of thyroid GRN including Nkx2-1/2-4, Pax2/5/8, and Hhex. Co-existence: the primitive endostyle and thyroid follicles are not mutually exclusive in this state. C Anatomy and developmental module of the primitive endostyle was lost in the LCA of modern vertebrates. Crown group of cyclostomes have retained the early and broad expression of Nkx2-1/2-4. C’ According to the classical hypothesis, the independent GRN of the primitive endostyle may have enabled the loss of its morphology without influencing thyroid development in each ancestor of gnathostomes, hagfishes, and possibly stem lampreys. D In the LCA of modern lampreys, an atavistic endostyle is secondarily acquired by recruiting a similar developmental module to form the endostyle. END, endostyle; THY, thyroid
Stem groups of vertebrates, such as Myllokunmingia, are considered to be microphagous suspension-feeders like modern non-vertebrate chordates [38], suggesting that the shared developmental mechanisms of the primitive endostyle were retained in these animals. Meanwhile, according to the alternative hypothesis, the fossil lamprey (Mesomyzon mengae) from the early Cretaceous has an ammocoete larval phase [39], suggesting that the endostyle in the modern lamprey lineage was likely acquired after the Palaeozoic.
The genetic basis of developmental mechanisms underlying endostyle formation remains largely unknown, and comparative single-cell transcriptomic analyses of the developing endostyles of non-vertebrate chordates and larval lampreys may elucidate similarities and differences in the GRN involved in morphogenesis. Furthermore, gene expression profiles in the endostyle of metamorphosing lampreys may also provide insights into the mechanisms of how the thyroid GRN is induced during the development. The novel hypothesis that the crown lamprey secondarily acquired the endostyle thus warrants further testing via future studies. According to the classical scenario, the independent nature of the GRN of the primitive endostyle from the thyroid gland may have enabled the loss of its morphology without influencing thyroid development in the respective ancestors of gnathostomes, hagfishes, and possibly some stem lampreys (Fig. 4C’). Alternatively, the LCA of modern vertebrates may have lost the primitive endostyle following the acquisition of thyroid follicles (Fig. 4C).
Conclusions
We described the early development of the thyroid and endostyle in two extant cyclostomes and found that the histological features and the regulatory gene expression of hagfish thyroid development are nearly identical to those in gnathostomes, namely direct development. Meanwhile, different from gnathostomes, the broad pharyngeal expression of Nkx2-1/2-4 prior to the initiation of the development of endostyle and thyroid was shared in lamprey and hagfish. Since the pharyngeal expression of Nkx2-1/2-4 orthologs is also conserved in the living non-vertebrate chordates and hemichordates, the expression domain in the ventral pharynx is likely a synapomorphy of chordates. The endostyles and thyroid are often lumped together as a homolog, but the heterochronic and heterogeneous expression of thyroid-related genes including the Nkx2-1/2-4 in the lamprey endostyle and nonvertebrate chordates suggests that the GRNs for the endostyle and thyroid organogenesis are likely distinct and independent as a separate developmental module. Although the plesiomorphy of lamprey endostyle remains elusive, we conclude that the spatiotemporal decoupling of these modules allowed the loss of the primitive endostyle in the vertebrate evolution. Furthermore, together with the recent fossil discovery, we question whether the lamprey endostyle is a bona fide plesiomorphic trait of vertebrates.
Methods
Sample collection and embryonic materials
Eptatretus burgeri is the only hagfish for which a method for obtaining fertilized eggs has been established [24]. Eptatretus burgeri embryos were obtained as per a previous protocol [24, 40] and fixed as described previously [17, 25, 41]. Eptatretus atami hatchling was sampled and fixed as described previously [41] (Additional file 1: Fig. S2H-I’). Fertilized eggs of Arctic lamprey (L. camtschaticum) were obtained via artificial fertilization, and the embryos were fixed with 4% paraformaldehyde. Adult cloudy catsharks (S. torazame) were collected from Ibaraki Prefecture, Japan. Adults and the deposited eggs were maintained in the same laboratory tank, and the embryos were fixed with 4% paraformaldehyde and Bouin’s fixative for in situ hybridization and histological observation, respectively. Embryos of cyclostomes and catshark used in this study were staged as described previously (Additional file 2: Table S1) [17, 23, 27, 42, 43].
Histology of hagfish and catshark embryos
The tissue sections of hagfish embryos were prepared in a previous study [17]. Several peri-midline sagittal sections previously stained by in situ hybridization were used for histological observations in the present study. A 3D reconstruction of the thyroid gland and blood vessels of hagfish was added to the image initially reconstructed by Oisi et al. [17] and is presented at different angles. The 3D images of catshark embryos reconstructed by Adachi et al. [44] were recolored and are shown at different angles. Reconstructed images were obtained using Avizo software, version 9.4 (Thermo Fisher Scientific, Waltham, MA, USA).
Molecular cloning
Previously reported partial nucleotide sequences of lamprey Pax2/5/8A and Nkx2-1/2-4 orthologues of hagfish and catshark were obtained from GenBank (Accession numbers: AB079852; AB747372; AB773852) [25, 31, 45]. The sequences of hagfish Pax2/5/8A, B, and Hhex, and lamprey Pax2/5/8B, and Hhex were obtained from the previously reported transcriptome data sets from the GenBank database (SRA accession numbers: SRX2541845-SRX2541849; SRX2847491-SRX2847498) [46]. The nucleotide sequences of catshark Pax2, 5, 8, and Hhex were obtained from assembled transcriptome database Squalomix (https://transcriptome.riken.jp/squalomix/) [47].
The total RNA was extracted from the frozen or fresh whole embryos using the ISOGEN reagent (Nippon Gene, Toyama, Japan). The fragments of each target gene amplified with KAPA TaqExtra DNA polymerase (KAPA Biosystems, Wilmington, MA, USA) using gene-specific primer sets (Additional file 3: Table S2) were cloned into the pGEM-T-Easy vector (Promega, Madison, WI, USA) and sequenced. Digoxigenin-labeled RNA probes were synthesized by using these plasmids. The plasmids containing lamprey Nkx2-1/2-4 paralogues (Nkx2-1/2-4A, B, C) generated in a previous study were used for probe synthesis [25].
Molecular phylogenetic analysis
The molecular phylogenetic trees of vertebrate Pax2/5/8 and Hhex genes were inferred via the maximum-likelihood method using PhyML3.0 (http://www.atgc-montpellier.fr/phyml/download.php) with the JTT+G4 model.
In situ hybridization
In situ hybridization on paraffin wax-embedded sections of hagfish embryos from stages 45 to 53 (n = 1 biological replication) was performed as previously described [17, 25]. Whole-mount in situ hybridization of lamprey embryos from stages 23 to 26 (n = 8 biological replicates) and shark embryos at stage 24 (n = 3 biological replicates) was performed according to Kusakabe et al. [48] and Sugahara et al. [49], respectively. As negative controls, sense strand probes were used for in situ hybridization in catshark and lamprey embryos. The hybridized lamprey embryos were fixed again and embedded in a mixture of 7.5% fish gelatin (Wako Pure Chemical Industries, Osaka, Japan) and 1.5% agarose (Nippon Gene); 40-μm-thick sections were obtained using a vibratome (LinearSlicer PRO7; Dosaka EM, Kyoto, Japan).
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its additional files. Newly identified cDNA sequences of catshark Pax2, 5, 8, Hhex, hagfish Pax2/5/8A, B, Hhex, and lamprey Pax2/5/8B have been deposited in GenBank under accession numbers LC495438 - LC495447.
Abbreviations
- LCA:
-
Last common ancestor
- GRN:
-
Gene regulatory network
- Tg:
-
Thyroglobulin
- TPO:
-
Thyroid peroxidase
References
Ogasawara M, Shigetani Y, Suzuki S, Kuratani S, Satoh N. Expression of thyroid transcription factor-1 (TTF-1) gene in the ventral forebrain and endostyle of the agnathan vertebrate, Lampetra japonica. Genesis. 2001;30:51–8.
Kluge B, Renault N, Rohr KB. Anatomical and molecular reinvestigation of lamprey endostyle development provides new insight into thyroid gland evolution. Dev Genes Evol. 2005;215:32–40.
Norris D, Carr J. Vertebrate endocrinology. 5th ed. New York: Academic; 2013.
Matsumoto A, Ishii S. Atlas of endocrine organs: vertebrates and invertebrates. Berlin: Springer-Verlag; 1992.
Baker KF, Berg O, Gorbman A, Nigrelli RF, Gordon M. Functional thyroid tumors in the kidneys of platyfish. Cancer Res. 1955;15:118–23.
Rähr H. The circulatory system of amphioxus (Branchiostoma lanceolatum (Pallas)): a light-microscopic investigation based on intravascular injection technique. Acta Zool. 1979;60:1–18.
Konrad MW. Blood circulation in the ascidian tunicate Corella inflata (Corellidae). PeerJ. 2016;4:e2771.
Eales JG. Iodine metabolism and thyroid-related functions in organisms lacking thyroid follicles: are thyroid hormones also vitamins? Proc Soc Exp Biol Med. 1997;214:302–17.
Müller W. Über die Hypobranchialrinne der Tunicaten und deren Vorhandensein bei Amphioxus und den Cyclostomen. Z Med Naturwiss. 1873;7:327–32.
Gorbman A. Hagfish development. Zool Sci. 1997;14:375–90.
Paris M, Escriva H, Schubert M, Brunet F, Brtko J, Ciesielski F, et al. Amphioxus postembryonic development reveals the homology of chordate metamorphosis. Curr Biol. 2008;18:825–30.
Hiruta J, Mazet F, Yasui K, Zhang P, Ogasawara M. Comparative expression analysis of transcription factor genes in the endostyle of invertebrate chordates. Dev Dyn. 2005;233:1031–7.
Ogasawara M. Overlapping expression of amphioxus homologs of the thyroid transcription factor-1 gene and thyroid peroxidase gene in the endostyle: insight into evolution of the thyroid gland. Dev Genes Evol. 2000;210:231–42.
Marine D. The metamorphosis of the endostyle (thyroid gland) of ammocoetes branchialis (larval land-locked Petromyzon marinus (Jordan) or Petromyzon dorsatus (Wilder)). J Exp Med. 1913;17:379–95.
Stockard CR. The development of the thyroid gland in Bdellostoma stoutii. Anat Anz. 1906;29:91–9.
Mallatt J, Sullivan J. 28S and 18S rDNA sequences support the monophyly of lampreys and hagfishes. Mol Biol Evol. 1998;15:1706–18.
Oisi Y, Ota KG, Kuraku S, Fujimoto S, Kuratani S. Craniofacial development of hagfishes and the evolution of vertebrates. Nature. 2013;493:175–80.
Ota KG, Kuratani S. Cyclostome embryology and early evolutionary history of vertebrates. Integr Comp Biol. 2007;47:329–37.
Nilsson M, Fagman H. Development of the thyroid gland. Development. 2017;144:2123–40.
Parlato R, Rosica A, Rodriguez-Mallon A, Affuso A, Postiglione MP, Arra C, et al. An integrated regulatory network controlling survival and migration in thyroid organogenesis. Dev Biol. 2004;276:464–75.
Kimura S, Hara Y, Pineau T, Fernandez-Salguero P, Fox CH, Ward JM, et al. The T/ebp null mouse: thyroid-specific enhancer-binding protein is essential for the organogenesis of the thyroid, lung, ventral forebrain, and pituitary. Genes Dev. 1996;10:60–9.
Nakada C, Iida A, Tabata Y, Watanabe S. Forkhead transcription factor foxe1 regulates chondrogenesis in zebrafish. J Exp Zool B Mol Dev Evol. 2009;312:827–40.
Dean B. On the embryology of Bdellostoma stouti. A general account of myxinoid development from the egg and segmentation to hatching. Jena: Gustav Fischer; 1899. p. 220–76.
Ota KG, Kuraku S, Kuratani S. Hagfish embryology with reference to the evolution of the neural crest. Nature. 2007;446:672–5.
Sugahara F, Pascual-Anaya J, Oisi Y, Kuraku S, Aota S-I, Adachi N, et al. Evidence from cyclostomes for complex regionalization of the ancestral vertebrate brain. Nature. 2016;531:97–100.
Serra M, Alysandratos KD, Hawkins F, McCauley KB, Jacob A, Choi J, et al. Pluripotent stem cell differentiation reveals distinct developmental pathways regulating lung-versus thyroid-lineage specification. Development. 2017;144:3879–93.
Tahara Y. Normal stages of development in the lamprey, Lampetra reissneri (Dybowski). Zool Sci. 1988;5:109–18.
Simakov O, Kawashima T, Marlétaz F, Jenkins J, Koyanagi R, Mitros T, et al. Hemichordate genomes and deuterostome origins. Nature. 2015;527:459–65.
Fagman H, Amendola E, Parrillo L, Zoppoli P, Marotta P, Scarfò M, et al. Gene expression profiling at early organogenesis reveals both common and diverse mechanisms in foregut patterning. Dev Biol. 2011;359:163–75.
Sakiyama J, Yamagishi A, Kuroiwa A. Tbx4-Fgf10 system controls lung bud formation during chicken embryonic development. Development. 2003;130:1225–34.
Murakami Y, Ogasawara M, Sugahara F, Hirano S, Satoh N, Kuratani S. Identification and expression of the lamprey Pax6 gene: evolutionary origin of the segmented brain of vertebrates. Development. 2001;128:3521–31.
Onai T, Lin HC, Schubert M, Koop D, Osborne PW, Alvarez S, et al. Retinoic acid and Wnt/β-catenin have complementary roles in anterior/posterior patterning embryos of the basal chordate amphioxus. Dev Biol. 2009;332:223–33.
Miccadei S, De Leo R, Zammarchi E, Natali PG, Civitareale D. The synergistic activity of thyroid transcription factor 1 and Pax 8 relies on the promoter/enhancer interplay. Mol Endocrinol. 2002;16:837–46.
Evans TM, Janvier P, Docker MF. The evolution of lamprey (Petromyzontida) life history and the origin of metamorphosis. Rev Fish Biol Fisheries. 2018;28:825–38.
Miyashita T, Gess RW, Tietjen K, Coates MI. Non-ammocoete larvae of Palaeozoic stem lampreys. Nature. 2021;591:408–12.
Northcutt RG, Gans C. The genesis of neural crest and epidermal placodes: a reinterpretation of vertebrate origins. Q Rev Biol. 1983;58:1–28.
Mallatt J. Feeding ecology of the earliest vertebrates. Zool J Linn Soc. 1984;82:261–72.
Shu DG, Morris SC, Han J, Zhang ZF, Yasui K, Janvier P, et al. Head and backbone of the Early Cambrian vertebrate Haikouichthys. Nature. 2003;421:526–9.
Chang MM, Wu F, Miao D, Zhang J. Discovery of fossil lamprey larva from the Lower Cretaceous reveals its three-phased life cycle. Proc Natl Acad Sci U S A. 2014;111:15486–90.
Oisi Y, Kakitani O, Kuratani S, Ota KG. Analysis of embryonic gene expression patterns in the hagfish. In: Hauptmann G, editor. In situ hybridization methods. New York: Springer; 2015. p. 249–62.
Oisi Y, Fujimoto S, Ota KG, Kuratani S. On the peculiar morphology and development of the hypoglossal, glossopharyngeal and vagus nerves and hypobranchial muscles in the hagfish. Zool Lett. 2015;1:6.
Ballard WW, Mellinger J, Lechenault H. A series of normal stages for development of Scyliorhinus canicula, the lesser spotted dogfish (Chondrichthyes: Scyliorhinidae). J Exp Zool. 1993;267:318–36.
Higuchi S, Sugahara F, Pascual-Anaya J, Takagi W, Oisi Y, Kuratani S. Inner ear development in cyclostomes and evolution of the vertebrate semicircular canals. Nature. 2019;565:347–50.
Adachi N, Pascual-Anaya J, Hirai T, Higuchi S, Kuratani S. Development of hypobranchial muscles with special reference to the evolution of the vertebrate neck. Zool Lett. 2018;4:5.
Sugahara F, Murakami Y, Adachi N, Kuratani S. Evolution of the regionalization and patterning of the vertebrate telencephalon: what can we learn from cyclostomes? Curr Opin Genet Dev. 2013;23:475–83.
Pascual-Anaya J, Sato I, Sugahara F, Higuchi S, Paps J, Ren Y, et al. Hagfish and lamprey Hox genes reveal conservation of temporal colinearity in vertebrates. Nat Ecol Evol. 2018;2:859–66.
Hara Y, Yamaguchi K, Onimaru K, Kadota M, Koyanagi M, Keeley SD, et al. Shark genomes provide insights into elasmobranch evolution and the origin of vertebrates. Nat Ecol Evol. 2018;2:1761–71.
Kusakabe R, Higuchi S, Tanaka M, Kadota M, Nishimura O, Kuratani S. Novel developmental bases for the evolution of hypobranchial muscles in vertebrates. BMC Biol. 2020;18:120.
Sugahara F, Murakami Y, Kuratani S. Gene expression analysis of lamprey embryos. In: Hauptmann G, editor. In situ hybridization methods. New York: Springer; 2015. p. 263–78.
Acknowledgements
We thank K.G. Ota and O. Kakitani for the sample collection of hagfish; K. Shirato for the sample collection of catshark; H. Nagashima, N. Sato, and S. Yanai for the sample collection of lamprey; T. Onai for the sample collection of Amphioxus; T. Hirasawa for technical advice; M.K.S. Wong for critical reading of the manuscript; S. Shibuya and K. Yamamoto for maintenance of aquarium tanks; S. Kuraku, Y. Hara, M. Kadota, K. Itomi, C. Tanegashima, K. Tatsumi, and O. Nishimura from the Phyloinformatics Team, RIKEN BDR, for gene sequence analyses.
Funding
This work was supported by Grant-in-Aid for Scientific Research (A) 15H02416, Grant-in-Aid for Scientific Research on Innovative Areas (Research in a Proposed Research Area) 17H06385, and a Naito Grant for the Promotion of Focused Research (The Naito Foundation) to SK; Grant-in-Aid for JSPS Fellows 15J08671 and a Special Postdoctoral Researcher Program of RIKEN to WT; Grant-in-Aid for Scientific Research (C) 19K06794 to FS
Author information
Authors and Affiliations
Contributions
WT and SK conceived the project, designed the experiments, and wrote the paper. WT, FS, SH1, and JP-A obtained the hagfish embryos. WT, FS, SH1, RK, JP-A, IS, YO, NO, HM, and NA performed experiments. WT, FS, SH1, RK, JP-A, IS, YO, NO, HM, NA, SH2, and SK discussed the results and contributed to the final version of the manuscript. The authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All animal experiments were conducted in accordance with the institutional and national guidelines for animal ethics and approved by the committees of RIKEN (Kobe Branch) and the University of Tokyo.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Fig. S1.
Endostyle and thyroid glands of chordates. Fig. S2. Hagfish thyroid development described by Charles Stockard and hagfish thyroid gland development during late developmental stages. Fig. S3. Thyroid development in catshark. Fig. S4. Expression patterns of cyclostome- and lamprey-specific paralogues. Fig. S5. Molecular phylogenetic analysis of Pax2/5/8 and Hhex.
Additional file 2: Table S1.
Staging of cyclostome and catshark embryos used in this study.
Additional file 3: Table S2.
Primer sets used for PCR.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Takagi, W., Sugahara, F., Higuchi, S. et al. Thyroid and endostyle development in cyclostomes provides new insights into the evolutionary history of vertebrates. BMC Biol 20, 76 (2022). https://doi.org/10.1186/s12915-022-01282-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12915-022-01282-7