Abstract
Purpose
The prevention of recurrence for patients with hepatocellular carcinoma after curative resection is still a great challenge in clinical practice. There are numerous studies that trying to search for favorable strategies to decrease the recurrence and prolong life span for these patients, whereas no consensus is reached till now. Herein, we aim to compare the efficacy between different reported treatments by network meta-analysis(NMA).
Methods
We searched Pubmed, Web of Science and Cochrane Library for abstracts and full-text articles published from database inception through February 2023. All of the random controlled trials(RCTs) were evaluated and collected as eligible studies. The primary outcome was the prevention of recurrence between different procedures. The second outcomes were one-year survival, three-year survival and five-year survival.
Results
Thirty-two RCTs including 5783 patients were selected, and 12 treatments were classified. Most of the studies were high quality with low bias. Thirty-one studies including 5629 patients were recruited for recurrence analysis. The network meta-analysis showed benefits from transarterial chemoembolization(TACE) + portal vein chemotherapy(PVC)[OR, 2.84 (1.15,6.99)] and internal radiotherapy(IRT) [OR, 2.63 (1.41,4.91)] compared to non-adjuvant(NA) treatment when considering prevention of recurrence. Seventeen studies including 2047 patients were collected for one-year survival analysis. The network meta-analysis showed benefit from TACE[OR, 0.33 (0.14,0.75)] when considering one-year survival. Twenty-one studies including 2463 patients were collected for three-year survival analysis. The network meta-analysis showed TACE [OR, 0.51 (0.30,0.86)], IRT[OR, 0.41 (0.20,0.83)] and dendritic cell(DC) [OR, 0.09 (0.01,0.98)] were better than NA when considering three-year survival. Sixteen studies including 1915 patients were collected for five-year survival analysis. The network meta-analysis didn’t show any benefit from different treatments when considering five-year survival. Other strategies including external radiotherapy(ERT), branched-chain amino acids(BCAA), hepatic artery infusion chemotherapy(HAIC), cytokine-induced killer(CIK), adoptive immunotherapy(AIT), Huaier, interferon(IFN), oral chemotherapy(OCT) and sorafenib(SOR) didn’t show significant benefit regardless of prevention of recurrence or short-, long- time survival.
Conclusion
This NMA found that TACE + PVC and IRT were considered as the procedures to decrease HCC recurrence rate. TACE, IRT and DC were preferred when considering the extending of life span for post-operative patients with HCC. Large scale of RCTs are needed to verify it.
Similar content being viewed by others
Introduction
Resection is still one of the main strategies for the patients with early stage of hepatocellular carcinoma(HCC), and the expected survival of these patients could reach up to more than 5 years(Reig et al. 2022). However, nearly 50% of the patients will develop recurrent HCC within 5 year after surgical resection(Tabrizian et al. 2015; Yao et al. 2022), which decreases the overall survival(OS) in these patients and patients with unresectable or advanced stage indicate poor OS (Sun et al. 2019; Wang et al. 2019, 2022) A study based on large scale of investigation and follow-up demonstrated that preoperative alpha-fetoprotein(AFP) level higher than 400 ug/L, tumor size greater than 5 cm, multiple tumors, satellites, microvascular invasion, cirrhosis and intraoperative blood transfusion were considered as risk factors of tumor recurrences(Yao et al. 2022).
To decrease the recurrence rate, numerous strategies were applied to be adjuvant therapy for the patients after tumor resection, including adoptive immunotherapy(AIT), external radiotherapy(ERT), hepatic artery infusion chemotherapy(HAIC), interferon(IFN), internal radiotherapy(IRT), oral chemotherapy(OCT), transarterial chemoembolization(TACE)(Chen et al. 2021; Huo et al. 2020; Liu et al. 2021). However, there is still no consensus on the adjuvant treatment for patients with HCC after resection. Sorafenib(SOR) was not an effective adjuvant therapy for HCC following resection or ablation based on a large random controlled trial(RCT)(Bruix et al. 2015), and a post-hoc study identified that no mutation, gene amplification or proposed gene signatures predicted adjuvant sorafenib benefit(Pinyol et al. 2019). A meta-analysis recruited 7 studies, although most of them were retrospective studies, demonstrated that adjuvant TACE is superior for the patients with microvascular invasion (MVI)(Shen et al. 2020). However, another meta-analysis concluded that TACE, radiotherapy and sorafenib were listed as the beneficial treatment to prevent recurrence, although the studies included were mostly retrospective studies(Yang et al. 2021). HAIC was also identified as an option for adjuvant therapy, which had favorable prognosis on OS and disease-free survival (DFS) when compared with control group(Ke et al. 2021; Li et al. 2021). Other attempts such as OCT including 5-fluorouracil or uracil-tegafur(Hasegawa et al. 2006; Yamamoto et al. 1996), adoptive immunotherapy with lymphocyte infusions or cytokine-induced killer cells(Hui et al. 2009; Takayama et al. 2000), IFN(Nishiguchi et al. 2005), oral Huaier granule(Chen et al. 2018), et al. were identified as effective therapy for postoperative treatment.
However, there are still limitations for us to get a better conclusion of adjuvant therapy for these patients. One is the limited RCTs to verify the effectiveness of treatments, which needs more high quality studies to be carried out, another is deficiency of comparison between different options, which could be the best choice for the prevention of recurrence for the patients with HCC after curative resection. Herein, we try to collect all of the RCTs about adjuvant therapy for HCC after curative resection, and conclude the different effectiveness of variable adjuvant treatments by network meta-analysis(NMA).
Methods
This study followed the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) statement. As current meta-analysis was not based on individual patient-level, informed consent was waived.
Search strategies and selection criteria
We searched Pubmed, Web of Science and Cochrane Library from database inception up through February 2023 for abstracts and full-text articles published about the comparison of different adjuvant procedures for patients with HCC after curative resection. Key words for the data search included liver cancer, hepatocellular carcinoma, adjuvant, post-operative. Consensus-based discussions were taken to solve the disagreements between authors(YYY and YW).
Studies including random controlled trials that compared the efficacy of different treatments for prevention of recurrence and acquisition of survival were selected. We excluded single arm studies or non-RCTs. We chose the most recent or complete study when duplicate publications or studies published in the same center with patients overlapped.
Two reviewers(YW and FMY) of us independently evaluated and extracted data from each study. The basic information of studies included: author/publication year, country, group, patients number, sex distribution, age, tumor size, tumor number, liver cirrhosis status, hepatitis status, Child-pugh score, Eastern Cooperative Oncology(ECOG), pre-operative AFP, macrovascular invasive, microvascular invasive and surgical margin. The primary outcome was the recurrence rate between different procedures. The secondary outcome was the survival of different treatments, including one-year survival, three-year survival and five-year survival.
Risk of bias evaluation
The Cochrane risk of bias tool was used to evaluate the quality, which included the following domains: random sequence generation, allocation concealment, blinding, incomplete outcome data, and selective outcome reporting(Higgins et al. 2011). Two authors(HQX and FMY) evaluated the studies independently and made a consensus after discussion.
Statistical analysis
The statistical analysis was conducted using Stata software (version 16, Stata Corp. LP, College station, TX, USA). Review Manager 5.3 software (Cochrane Collaboration, Oxford, UK) was used to evaluate the risk bias. The heterogeneity of direct evidence and in-direct evidence was according to inconsistency factor and the value of heterogeneity. The assessment of heterogeneity was according to the I2 test, and cut-off values of less than 25%, 25–75%, and greater than 75% represented low, moderate, and high heterogeneity, respectively. Network meta-analyses(NMA) of different treatments were using a random-effects models. League tables were generated for back-transformed network estimates. Odds ratios (ORs) and 95% confidence intervals were used to compare different treatments.
Results
Study selection and characteristics
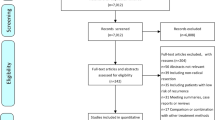
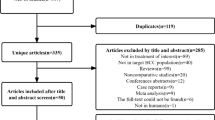
Thirty-two studies(Bruix et al. 2015; Chen et al. 2012, 2013, 2018; Chung et al. 2013; Hachiya et al. 2020; Hasegawa et al. 2006; Hirokawa et al. 2020; Huang et al. 2015; Hui et al. 2009; Izumi et al. 1994; Lai et al. 1998; Lau et al. 1999; Li et al. 1995, 2006, 2020a, b; Lo et al. 2007; Matsui et al. 2021; Mazzaferro et al. 2006; Nishiguchi et al. 2005; Peng et al. 2009; Shi et al. 2022; Sun et al. 2006; Takayama et al. 2000; Wang et al. 2018; Wei et al. 2018; Xia et al. 2010; Xu et al. 2016; Yamamoto et al. 1996; Yu et al. 2014; Zhong et al. 2009) including 5783 patients were selected from a total of 5846 records(Fig. 1). The comparison of the studies included: ERT vs. non-adjuvant(NA)(n = 2); branched-chain amino acids(BCAA) vs. NA(n = 1); IRT vs. NA(n = 4); HAIC vs. NA(n = 3); dendritic cell(DC) vs. NA(n = 1); cytokine-induced killer(CIK) vs. NA(n = 2); AIT vs. NA(n = 1); Huaier vs. NA(n = 1); IFN vs. NA(n = 5); OCT vs. NA(n = 3); TACE vs. NA(n = 7); TACE vs. TACE + portal vein chemotherapy(PVC) vs. NA(n = 1); SOR vs. NA(n = 1). All of the studies included were random controlled trials, with matched number of patients, sex distribution, tumor size, liver cirrhosis status, Child-pugh score, ECOG, pre-operative alpha-fetoprotein(AFP) in each trial. However, there were some selection bias between different trials, such as macro-vascular invasive, micro-vascular invasive and surgical margin, which might be the most important factors that impact the tumor recurrence. The details of the studies were presented in Table 1.
When we considered the risk of bias, most of the studies have perfect random sequence generation, allocation concealment, complete outcome data and low reporting bias, whereas the different treatments correlated with different procedures or adverse reactions that were easy to distinguish with non-adjuvant therapy cause the impossible for blinding of participants and personnel for nearly all trials. Moreover, there were a small number of studies reported blinding of outcome assessment. The risk of bias was presented in Fig. 2.
Network meta-analysis
Thirty-one studies including 5629 patients were collected for preventing recurrence between different treatments. The network plot showed that most of the studies were compared different treatments with NA, whereas there was a loop between TACE, TACE + PVC and NA(Fig. 3A). The funnel plot also demonstrated uniform distribution of different comparisons(Fig. 3B). The network analysis showed benefit from TACE + PVC[OR, 2.84 (1.15,6.99)] and IRT[OR, 2.63 (1.41,4.91)] when considering recurrence. SOR[OR, 0.57 (0.24,1.38)] had the trend of increasing recurrence rate, although there were no significant differences between them. Other treatments including TACE, OCT, IFN, Huaier, HAIC, ERT, CIK, BCAA and AIT didn’t present significant difference with NA, although all of them had the trend of preventing recurrence(Table 2). The ranking of different treatments according to surface under the cumulative ranking curve (SUCRA) was as follows: TACE + PVC, 88.0%; IRT, 82.3%; ERT, 72.8%; AIT, 71.8%; Huaier, 59.4%; HAIC, 58.7%; TACE, 51.0%; IFN, 48.0%; OCT, 39.0%; CIK, 36.7%; DC, 30.7%; BCAA, 30.5%; NA, 23.4%; SOR, 7.8%(Table 3).
Network plot and funnel plot of studies included in the analysis of recurrence. A Network plot of studies included in the analysis of recurrence. B funnel plot of studies included in the analysis of recurrence. ERT, external radiotherapy; BCAA, branched-chain amino acids; IRT, internal radiotherapy; HAIC, hepatic artery infusion chemotherapy; DC, dendritic cell; CIK, cytokine-induced killer; AIT, adoptive immunotherapy; IFN, interferon; OCT, oral chemotherapy; TACE, transcatheter arterial chemoembolization; PVC, portal vein chemotherapy; SOR, sorafenib; NA, non-adjuvant
When we did network meta-analysis for survival, we used the endpoints including one-year survival, three-year survival and five-year survival. For one-year survival, seventeen studies including 2047 patients were recruited. The network plot showed that most of the studies were compared different treatments with NA, and no loop was presented in different treatments(Fig. 4A). The funnel plot also demonstrated uniform distribution of different comparisons(Fig. 4B). The network analysis showed benefit from TACE [OR, 0.33 (0.14,0.75)] when considering one-year survival. CIK demonstrated the trend of decreasing one-year survival[OR, 2.24 (0.64,7.85)], although no significant difference between CIK and NA. Other treatments including OCT, IRT, IFN, HAIC and ERT didn’t present significant difference between them, although all of them had the trend of increasing one-year survival(Table 4). The ranking of different treatments according to SUCRA was as follows: IFN, 77.0%; IRT 67.8%; TACE, 66.8%; HAIC, 63.1%; ERT, 55.6%; OCT, 38.6%; NA, 24.4%; CIK, 6.7%(Table 5).
Network plot and funnel plot of studies included in the analysis of one-year survival. A Network plot of studies included in the analysis of one-year survival. B funnel plot of studies included in the analysis of one-year survival. ERT, external radiotherapy; IRT, internal radiotherapy; HAIC, hepatic artery infusion chemotherapy; CIK, cytokine-induced killer; IFN, interferon; OCT, oral chemotherapy; TACE, transcatheter arterial chemoembolization; NA, non-adjuvant
For three-year survival, twenty-one studies including 2463 patients were collected. The network plot showed that most of the studies were compared different treatments with NA, and no loop was presented in different treatments(Fig. 5A). The funnel plot also demonstrated uniform distribution of different comparisons(Fig. 5B). The network analysis showed benefit from TACE [OR, 0.51 (0.30,0.86)], IRT[OR, 0.41 (0.20,0.83)] and DC[OR, 0.09 (0.01,0.98)] when considering three-year survival. CIK demonstrated the trend of decreasing three-year survival[OR, 1.59 (0.66,3.84)], although no significant difference between CIK and NA. Other treatments including OCT, IFN, HAIC, ERT and IRT didn’t present significant difference between them, although all of them had the trend of increasing three-year survival(Table 6). The ranking of different treatments according to SUCRA was as follows: DC, 92.3%; IRT, 67.9%; AIT, 66.5%; HAIC, 59.7%; TACE, 57.6%; ERT, 47.0%; IFN, 46.6%; OCT, 35.4%; NA, 19.8%; CIK, 7.1%(Table 7).
Network plot and funnel plot of studies included in the analysis of three-yea survival. A Network plot of studies included in the analysis of three-yea survival. B funnel plot of studies included in the analysis of three-yea survival. ERT, external radiotherapy; IRT, internal radiotherapy; HAIC, hepatic artery infusion chemotherapy; DC, dendritic cell; CIK, cytokine-induced killer; AIT, adoptive immunotherapy; IFN, interferon; OCT, oral chemotherapy; TACE, transcatheter arterial chemoembolization; NA, non-adjuvant
For five-year survival, Sixteen studies including 1915 patients were collected. The network plot showed that most of the studies were compared different treatments with NA, and no loop was presented in different treatments(Fig. 6A). The funnel plot also demonstrated uniform distribution of different comparisons(Fig. 6B). The network analysis didn’t show any benefit for different treatments when compared to non-adjuvant therapy. TACE[OR, 0.52 (0.25,1.05)], IRT[OR, 0.49(0.23,1.07)], IFN[OR, 0.42(0.10,1.79)], ERT[OR, 0.51(0.19,1.34)] and AIT[OR, 0.77(0.22,2.73)] had the trend of increasing five-year survival, but with no significant difference(Table 6). Other treatments including OCT[OR, 1.01(0.38,2.66)], HAIC[OR, 1.00(0.26,3.77)], and CIK[OR, 1.10(0.44,2.74) didn’t show any benefit(Table 8). The ranking of different treatments according to SUCRA was as follows: IFN, 74.1%; IRT, 71.8%; TACE, 69.5%; ERT, 69.1%; AIT, 47.1%; HAIC, 33.6%; OCT, 31.4%; NA, 27.3%; CIK, 26.1%(Table 9).
Network plot and funnel plot of studies included in the analysis of five-yea survival. A Network plot of studies included in the analysis of five-yea survival. B funnel plot of studies included in the analysis of five-yea survival. ERT, external radiotherapy; IRT, internal radiotherapy; HAIC, hepatic artery infusion chemotherapy; CIK, cytokine-induced killer; AIT, adoptive immunotherapy; IFN, interferon; OCT, oral chemotherapy; TACE, transcatheter arterial chemoembolization; NA, non-adjuvant
Discussion
Most of the patients with early-stage HCC would undergo curative resection, whereas 50% patients might have disease recurrence within 5 years(Llovet et al. 2016). Studies have identified risk factors that associated with recurrence after curative resection, including macrovascular/microvascular invasion, tumor size greater than 5 centimeters in diameter, multiple nodes, positive resection margin or resection margin less than 1 centimeter, hepatitis B virus infection and AFP greater than 400ng/L(Imamura et al. 2003; Wang et al. 2020; Zeng et al. 2022). Numerous procedures were explored to decrease the recurrence rate and prolong life span. A network meta-analysis included 23 RCTs showed that IRT and HAIC were ranked as the best strategies for preventing recurrence and providing survival benefit(Liu et al. 2021). However, A recent evidence-based management of hepatocellular carcinoma identified 7 RCTs and concluded that adjuvant treatments didn’t improve recurrence-free survival (RFS)(Haber et al. 2021). There needs to be more studies to get more confident conclusions.
In present NMA, we collected thirty-two studies including 5783 patients from a total of 5846 records, which was the largest data for the analysis of adjuvant therapy for post-operative patients with HCC. Furthermore, all the trials included were RCTs with high quality and low bias according to the Cochrane evaluation. When considering the treatments that preventing post-operative recurrence, TACE + PVC[OR, 2.84 (1.15,6.99)] and IRT[OR, 2.63 (1.41,4.91)] were showed to be beneficial for these groups of patients. PVC was considered as an effective strategy in preventing recurrence in patients with portal vein tumor thrombosis(Fan et al. 2005). However, the surgical skill requirement and high risk limited the popularization of the treatment. TACE was confirmed to be disease-free survival benefit in MVI-positive patients, but weekly supports in patients without portal venous tumor thrombus(PVTT) in meta-analysis(Huo et al. 2020; Shen et al. 2020; Yang et al. 2021). Alpha fetoprotein level, systemic inflammation response index, alanine aminotransferase, tumour diameter and portal vein tumour thrombus were also confirmed to be independent prognostic factors of HCC early recurrence in patients with MVI who underwent TACE(Mao et al. 2022). However, PVC promoted median time to recurrence and OS in HCC patients with PVTT(Gao et al. 2019). The combination of TACE and PVC provided favorable recurrence free in our study, which might be related with the mixed baseline in studies recruited, and TACE plus PVC would decrease the recurrence rate in all patients with HCC after curative resection. The IRT included I131-mAb, I131-lipiodol and I125 seeds, obviously three strong RCTs provided the benefits for all the internal radiotherapy(Chen et al. 2013; Lau et al. 1999; Li et al. 2020a). However, some studies didn’t show any recurrence-free or OS benefit(Chung et al. 2013; Furtado et al. 2015).The implantation of internal radioactive source is also technique requirement which might be restriction of the application. The ranking of different treatments according to SUCRA was as follows: TACE + PVC, 88.0%; IRT, 82.3%; ERT, 72.8%; AIT, 71.8%; Huaier, 59.4%; HAIC, 58.7%; TACE, 51.0%, IFN, 48.0%; OCT, 39.0%; CIK, 36.7%; DC, 30.7%; BCAA, 30.5%; NA, 23.4%; SOR, 7.8%. From this data, we found out that adjuvant sorafenib was not a good choice for the patients to prevent recurrence according to the STORM trial(Bruix et al. 2015). Apart from the treatments collected in this study, immune checkpoint inhibitors(Kudo et al. 2022), antiviral therapy(Yin et al. 2013), Lenvatinib(Bai et al. 2022), and traditional herbal medicine(Zhai et al. 2018) also indicated recurrence free or OS benefit in single arm or prospective studies.
As for comparison of one-year survival, three-year survival and five-year survival in different treatments, TACE was considered to be favorable prognosis for one-year survival [OR, 0.33 (0.14,0.75)] and three-year survival[OR, 0.51 (0.30,0.86)], which was also confirmed by the meta-analysis before(Huo et al. 2020; Shen et al. 2020; Yang et al. 2021). IRT was considered as the good choice when evaluating three-year survival[OR, 0.41 (0.20,0.83)]. As for DC in the adjuvant therapy, the small size of the trial which might be selection bias for this result and more confident evidence needs to be performed(Matsui et al. 2021). The primary outcome of studies included in this analysis is the prevention of recurrence. However, the second outcomes in different studies are diverse. For example, the comparison of one-year survival just includes IFN, IRT, TACE, HAIC, ERT, OCT and CIK, which is fewer than the primary outcome. Moreover, the basic characteristics in IFN groups are better than other comparison, which might cause the difference and bias.
However, there are limitations in this NMA. Firstly, the basic characteristic for each trial is not paired enough. For example, some trials include patients with vascular invasive, surgical margins are diverse for different trials. Microvascular invasion and/or positive/narrow surgical margins are independent risk factors for HCC patients under hepatic resection(Hwang et al. 2023; Liu et al. 2023; Wang et al. 2023). The different patients included in trials might cause the bias of the comparison. For example, most of the patients recruited in ERT are positive surgical margins or Margin ≤ 1 cm, and the treatment of ERT might bring significant benefit for these patients(Shi et al. 2022; Yu et al. 2014). The benefit for IRT is the same as ERT(Chen et al. 2013; Chung et al. 2013; Li et al. 2020a). Most of the patients recruited in HAIC are MVI positive, however, nearly none of the patients collected in CIK are MVI negative, which might cause the significant different effect when comparing with observation(Li et al. 2020b; Xu et al. 2016). Apart from the diverse basic characteristic between different comparison, the patients included in the same comparison are different(Hirokawa et al. 2020; Li et al. 2020b). Secondly, the number of patients collected in each group are enormous diversity, which might bring bias when analyzing the data. Thirdly, some treatment such as IRT including different internal radioactive source which maybe different effectiveness. Furthermore, some treatment is single trial with limited number of patients recruited, which might restrict the application of the treatment.
In conclusion, the effectiveness for different adjuvant treatments in post-operative patients with HCC varies in trials. However, the adjuvant treatments have the trend of preventing recurrence and increase overall survival, although most of the procedures are no significant difference when compared to non-adjuvant therapy. This NMA found that TACE + PVC and IRT were considered as the best way to decrease recurrence rate. TACE, IRT and DC were preferred when considering the extend for life span. There needs to be more large scale of studies to confirm the results.
Availability of data and materials
The original contributions presented in the study are included in the article, further inquiries can be.
directed to the corresponding author.
References
Bai S, Hu L, Liu J, Sun M, Sun Y, Xue F. Prognostic Nomograms Combined Adjuvant Lenvatinib for Hepatitis B Virus-related Hepatocellular Carcinoma with Microvascular Invasion after Radical Resection. Front Oncol. 2022;12:919824. https://doi.org/10.3389/fonc.2022.919824.
Bruix J, et al. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015;16:1344–54. https://doi.org/10.1016/s1470-2045(15)00198-9.
Chen LT, et al. Long-term results of a randomized, observation-controlled, phase III trial of adjuvant interferon Alfa-2b in hepatocellular carcinoma after curative resection. Ann Surg. 2012;255:8–17. https://doi.org/10.1097/SLA.0b013e3182363ff9.
Chen K, Xia Y, Wang H, Xiao F, Xiang G, Shen F. Adjuvant iodine-125 brachytherapy for hepatocellular carcinoma after complete hepatectomy: a randomized controlled trial. PLoS ONE. 2013;8:e57397. https://doi.org/10.1371/journal.pone.0057397.
Chen Q, et al. Effect of Huaier granule on recurrence after curative resection of HCC: a multicentre, randomised clinical trial. Gut. 2018;67:2006–16. https://doi.org/10.1136/gutjnl-2018-315983.
Chen A, et al. Adjuvant transarterial chemoembolization to sorafenib in unresectable hepatocellular carcinoma: a meta-analysis. J Gastroenterol Hepatol. 2021;36:302–10. https://doi.org/10.1111/jgh.15180.
Chung AY, et al. Adjuvant hepatic intra-arterial iodine-131-lipiodol following curative resection of hepatocellular carcinoma: a prospective randomized trial. World J Surg. 2013;37:1356–61. https://doi.org/10.1007/s00268-013-1970-4.
Fan J, Zhou J, Wu ZQ, Qiu SJ, Wang XY, Shi YH, Tang ZY. Efficacy of different treatment strategies for hepatocellular carcinoma with portal vein tumor thrombosis. World J Gastroenterol. 2005;11:1215–9. https://doi.org/10.3748/wjg.v11.i8.1215.
Furtado RV, Ha L, Clarke S, Sandroussi C. Adjuvant iodine (131) Lipiodol after Resection of Hepatocellular Carcinoma. J Oncol. 2015;2015:746917. https://doi.org/10.1155/2015/746917.
Gao Y, et al. Chemotherapeutic perfusion of portal vein after tumor thrombectomy and hepatectomy benefits patients with advanced hepatocellular carcinoma: a propensity score-matched survival analysis. Cancer Med. 2019;8:6933–44. https://doi.org/10.1002/cam4.2556.
Haber PK, et al. Evidence-based management of Hepatocellular Carcinoma: systematic review and Meta-analysis of Randomized controlled trials (2002–2020). Gastroenterology. 2021;161:879–98. https://doi.org/10.1053/j.gastro.2021.06.008.
Hachiya H, et al. Effects of branched-chain amino acids on postoperative tumor recurrence in patients undergoing curative resection for hepatocellular carcinoma: a randomized clinical trial. J Hepatobiliary Pancreat Sci. 2020;27:819–29. https://doi.org/10.1002/jhbp.830.
Hasegawa K, et al. Uracil-tegafur as an adjuvant for hepatocellular carcinoma: a randomized trial. Hepatology. 2006;44:891–5. https://doi.org/10.1002/hep.21341.
Higgins JP, et al. The Cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. https://doi.org/10.1136/bmj.d5928.
Hirokawa F, et al. Is postoperative adjuvant transcatheter arterial infusion Therapy Effective for patients with Hepatocellular Carcinoma who underwent Hepatectomy? A prospective Randomized Controlled Trial. Ann Surg Oncol. 2020;27:4143–52. https://doi.org/10.1245/s10434-020-08699-w.
Huang SX, Wu YL, Tang CW, Feng WM, Xu YQ, Bao Y, Zheng YY. Prophylactic hepatic artery infusion chemotherapy improved survival after curative resection in patients with hepatocellular carcinoma. Hepatogastroenterology. 2015;62:122–5.
Hui D, Qiang L, Jian W, Ti Z, Da-Lu K. A randomized, controlled trial of postoperative adjuvant cytokine-induced killer cells immunotherapy after radical resection of hepatocellular carcinoma. Dig Liver Dis. 2009;41:36–41. https://doi.org/10.1016/j.dld.2008.04.007.
Huo YR, Chan MV, Chan C. Resection Plus post-operative adjuvant transcatheter arterial chemoembolization (TACE) compared with resection alone for Hepatocellular Carcinoma: a systematic review and Meta-analysis. Cardiovasc Intervent Radiol. 2020;43:572–86. https://doi.org/10.1007/s00270-019-02392-6.
Hwang YJ, Bae JS, Lee Y, Hur BY, Lee DH, Kim H. Classification of microvascular invasion of hepatocellular carcinoma: correlation with prognosis and magnetic resonance imaging. Clin Mol Hepatol. 2023;29:733–46. https://doi.org/10.3350/cmh.2023.0034.
Imamura H, et al. Risk factors contributing to early and late phase intrahepatic recurrence of hepatocellular carcinoma after hepatectomy. J Hepatol. 2003;38:200–7. https://doi.org/10.1016/s0168-8278(02)00360-4.
Izumi R, et al. Postoperative adjuvant hepatic arterial infusion of Lipiodol containing anticancer drugs in patients with hepatocellular carcinoma. Hepatology. 1994;20:295–301.
Ke Q, Wang L, Wu W, Huang X, Li L, Liu J, Guo W. Meta-analysis of postoperative adjuvant hepatic artery infusion Chemotherapy Versus Surgical resection alone for Hepatocellular Carcinoma. Front Oncol. 2021;11:720079. https://doi.org/10.3389/fonc.2021.720079.
Kudo M, et al. Final results of adjuvant nivolumab for hepatocellular carcinoma (HCC) after surgical resection (SR) or radiofrequency ablation (RFA) (NIVOLVE): a phase 2 prospective multicenter single-arm trial and exploratory biomarker analysis. J Clin Oncol. 2022;40:416–6. https://doi.org/10.1200/JCO.2022.40.4_suppl.416.
Lai EC, Lo CM, Fan ST, Liu CL, Wong J. Postoperative adjuvant chemotherapy after curative resection of hepatocellular carcinoma: a randomized controlled trial. Arch Surg. 1998;133:183–8. https://doi.org/10.1001/archsurg.133.2.183.
Lau WY, et al. Adjuvant intra-arterial iodine-131-labelled lipiodol for resectable hepatocellular carcinoma: a prospective randomised trial. Lancet. 1999;353:797–801. https://doi.org/10.1016/s0140-6736(98)06475-7.
Li JQ, Zhang YQ, Zhang WZ, Yuan YF, Li GH. Randomized study of chemoembolization as an adjuvant therapy for primary liver carcinoma after hepatectomy. J Cancer Res Clin Oncol. 1995;121:364–6. https://doi.org/10.1007/bf01225689.
Li Q, Wang J, Sun Y, Cui YL, Juzi JT, Qian BY, Hao XS. Postoperative transhepatic arterial chemoembolization and portal vein chemotherapy for patients with hepatocellular carcinoma: a randomized study with 131 cases. Dig Surg. 2006;23:235–40. https://doi.org/10.1159/000095396.
Li J, et al. Adjuvant (131)I-metuximab for hepatocellular carcinoma after liver resection: a randomised, controlled, multicentre, open-label, phase 2 trial. Lancet Gastroenterol Hepatol. 2020a;5:548–60. https://doi.org/10.1016/s2468-1253(19)30422-4.
Li S, et al. Postoperative adjuvant transarterial infusion chemotherapy with FOLFOX could improve outcomes of Hepatocellular Carcinoma Patients with Microvascular Invasion: a preliminary Report of a phase III, Randomized Controlled Clinical Trial. Ann Surg Oncol. 2020b;27:5183–90. https://doi.org/10.1245/s10434-020-08601-8.
Li S, et al. The role of hepatic arterial infusion Chemotherapy in the treatment of Hepatocellular Carcinoma: a systematic review and Meta-analysis. Chemotherapy. 2021;66:124–33. https://doi.org/10.1159/000518257.
Liu Y, et al. Comparative effectiveness of Adjuvant Treatment for Resected Hepatocellular Carcinoma: a systematic review and network Meta-analysis. Front Oncol. 2021;11:709278. https://doi.org/10.3389/fonc.2021.709278.
Liu J et al. (2023) The comparison of Surgical margins and type of hepatic resection for Hepatocellular Carcinoma with Microvascular Invasion. Oncologist. https://doi.org/10.1093/oncolo/oyad124.
Llovet JM, Zucman-Rossi J, Pikarsky E, Sangro B, Schwartz M, Sherman M, Gores G. Hepatocellular carcinoma. Nat Rev Dis Primers. 2016;2:16018. https://doi.org/10.1038/nrdp.2016.18.
Lo CM, et al. A randomized, controlled trial of postoperative adjuvant interferon therapy after resection of hepatocellular carcinoma. Ann Surg. 2007;245:831–42. https://doi.org/10.1097/01.sla.0000245829.00977.45.
Mao S, et al. A new prognostic model predicting hepatocellular carcinoma early recurrence in patients with microvascular invasion who received postoperative adjuvant transcatheter arterial chemoembolization. Eur J Surg Oncol. 2022. https://doi.org/10.1016/j.ejso.2022.08.013.
Matsui HM, et al. Novel adjuvant dendritic cell therapy with transfection of heat-shock protein 70 messenger RNA for patients with hepatocellular carcinoma: a phase I/II prospective randomized controlled clinical trial. Cancer Immunol Immunother. 2021;70:945–57. https://doi.org/10.1007/s00262-020-02737-y.
Mazzaferro V, et al. Prevention of hepatocellular carcinoma recurrence with alpha-interferon after liver resection in HCV cirrhosis. Hepatology. 2006;44:1543–54. https://doi.org/10.1002/hep.21415.
Nishiguchi S, Tamori A, Kubo S. Effect of long-term postoperative interferon therapy on intrahepatic recurrence and survival rate after resection of hepatitis C virus-related hepatocellular carcinoma. Intervirology. 2005;48:71–5. https://doi.org/10.1159/000082098.
Peng BG, He Q, Li JP, Zhou F. Adjuvant transcatheter arterial chemoembolization improves efficacy of hepatectomy for patients with hepatocellular carcinoma and portal vein tumor thrombus. Am J Surg. 2009;198:313–8. https://doi.org/10.1016/j.amjsurg.2008.09.026.
Pinyol R, et al. Molecular predictors of prevention of recurrence in HCC with sorafenib as adjuvant treatment and prognostic factors in the phase 3 STORM trial. Gut. 2019;68:1065–75. https://doi.org/10.1136/gutjnl-2018-316408.
Reig M, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol. 2022;76:681–93. https://doi.org/10.1016/j.jhep.2021.11.018.
Shen A, Liu M, Zheng D, Chen Q, Wu Z. Adjuvant transarterial chemoembolization after curative hepatectomy for hepatocellular carcinoma with microvascular invasion: a systematic review and meta-analysis. Clin Res Hepatol Gastroenterol. 2020;44:142–54. https://doi.org/10.1016/j.clinre.2019.06.012.
Shi C, et al. Adjuvant stereotactic body radiotherapy after marginal resection for hepatocellular carcinoma with microvascular invasion: a randomised controlled trial. Eur J Cancer. 2022;166:176–84. https://doi.org/10.1016/j.ejca.2022.02.012.
Sun HC, et al. Postoperative interferon alpha treatment postponed recurrence and improved overall survival in patients after curative resection of HBV-related hepatocellular carcinoma: a randomized clinical trial. J Cancer Res Clin Oncol. 2006;132:458–65. https://doi.org/10.1007/s00432-006-0091-y.
Sun JY, Yin T, Zhang XY, Lu XJ. Therapeutic advances for patients with intermediate hepatocellular carcinoma. J Cell Physiol. 2019;234:12116–21. https://doi.org/10.1002/jcp.28019.
Tabrizian P, Jibara G, Shrager B, Schwartz M, Roayaie S. Recurrence of hepatocellular cancer after resection: patterns, treatments, and prognosis. Ann Surg. 2015;261:947–55. https://doi.org/10.1097/sla.0000000000000710.
Takayama T, et al. Adoptive immunotherapy to lower postsurgical recurrence rates of hepatocellular carcinoma: a randomised trial. Lancet. 2000;356:802–7. https://doi.org/10.1016/s0140-6736(00)02654-4.
Wang Z, et al. Adjuvant Transarterial Chemoembolization for HBV-Related Hepatocellular Carcinoma after Resection: a randomized controlled study. Clin Cancer Res. 2018;24:2074–81. https://doi.org/10.1158/1078-0432.Ccr-17-2899.
Wang JC, Xia AL, Xu Y, Lu XJ. Comprehensive treatments for hepatocellular carcinoma with portal vein tumor thrombosis. J Cell Physiol. 2019;234:1062–70. https://doi.org/10.1002/jcp.27324.
Wang MD, et al. Early and late recurrence of Hepatitis B Virus-Associated Hepatocellular Carcinoma. Oncologist. 2020;25:e1541–51. https://doi.org/10.1634/theoncologist.2019-0944.
Wang J, Wu R, Sun JY, Lei F, Tan H, Lu X. An overview: management of patients with advanced hepatocellular carcinoma. Biosci Trends. 2022;16:405–25. https://doi.org/10.5582/bst.2022.01109.
Wang H, Liu R, Mo H, Li R, Lian J, Liu Q, Han S. A novel nomogram predicting the early recurrence of hepatocellular carcinoma patients after R0 resection. Front Oncol. 2023;13:1133807. https://doi.org/10.3389/fonc.2023.1133807.
Wei W, et al. Adjuvant transcatheter arterial chemoembolization after curative resection for hepatocellular carcinoma patients with solitary tumor and microvascular invasion: a randomized clinical trial of efficacy and safety. Cancer Commun (Lond). 2018;38:61. https://doi.org/10.1186/s40880-018-0331-y.
Xia Y, et al. Adjuvant therapy with capecitabine postpones recurrence of hepatocellular carcinoma after curative resection: a randomized controlled trial. Ann Surg Oncol. 2010;17:3137–44. https://doi.org/10.1245/s10434-010-1148-3.
Xu L, et al. A randomized controlled trial on patients with or without adjuvant autologous cytokine-induced killer cells after curative resection for hepatocellular carcinoma. Oncoimmunology. 2016;5:e1083671. https://doi.org/10.1080/2162402x.2015.1083671.
Yamamoto M, Arii S, Sugahara K, Tobe T. Adjuvant oral chemotherapy to prevent recurrence after curative resection for hepatocellular carcinoma. Br J Surg. 1996;83:336–40. https://doi.org/10.1002/bjs.1800830313.
Yang J, et al. The effects of several postoperative adjuvant therapies for hepatocellular carcinoma patients with microvascular invasion after curative resection: a systematic review and meta-analysis. Cancer Cell Int. 2021;21:92. https://doi.org/10.1186/s12935-021-01790-6.
Yao LQ et al. (2022) Clinical features of recurrence after hepatic resection for early-stage Hepatocellular Carcinoma and Long-Term Survival Outcomes of patients with recurrence: a multi-institutional analysis. Ann Surg Oncol. https://doi.org/10.1245/s10434-022-11454-y.
Yin J, et al. Effect of antiviral treatment with nucleotide/nucleoside analogs on postoperative prognosis of hepatitis B virus-related hepatocellular carcinoma: a two-stage longitudinal clinical study. J Clin Oncol. 2013;31:3647–55. https://doi.org/10.1200/jco.2012.48.5896.
Yu W, et al. Adjuvant radiotherapy in centrally located hepatocellular carcinomas after hepatectomy with narrow margin (< 1 cm): a prospective randomized study. J Am Coll Surg. 2014;218:381–92. https://doi.org/10.1016/j.jamcollsurg.2013.11.030.
Zeng ZM, et al. Advances in postoperative adjuvant therapy for primary liver cancer. World J Gastrointest Oncol. 2022;14:1604–21. https://doi.org/10.4251/wjgo.v14.i9.1604.
Zhai XF, Liu XL, Shen F, Fan J, Ling CQ. Traditional herbal medicine prevents postoperative recurrence of small hepatocellular carcinoma: a randomized controlled study. Cancer. 2018;124:2161–8. https://doi.org/10.1002/cncr.30915.
Zhong C, Guo RP, Li JQ, Shi M, Wei W, Chen MS, Zhang YQ. A randomized controlled trial of hepatectomy with adjuvant transcatheter arterial chemoembolization versus hepatectomy alone for stage III a hepatocellular carcinoma. J Cancer Res Clin Oncol. 2009;135:1437–45. https://doi.org/10.1007/s00432-009-0588-2.
Acknowledgements
We acknowledged the authors of the studies included in this study.
Funding
The study was supported by Jiangxi provincial department of science and technology (grant number 20203BBGL73144), Health Commission of Jiangxi Province (grant number 202110059), and National Natural Science Foundation of China(grant number 82260130).
Author information
Authors and Affiliations
Contributions
All authors contributed to the article and approved the submitted version. FMY designed the study, YYY and YW searched the literatures, YW and FMY evaluated and extracted data from each study. HQX and FMY evaluated the bias of studies. YYY and FMY drafting of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This is a meta-analysis. The Second affiliated hospital of Nanchang University Research Ethics Committee has confirmed that no ethical approval is required.
This is a meta-analysis. Informed consent is not required.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Ye, Y., Wang, Y., Xu, H. et al. Network meta-analysis of adjuvant treatments for patients with hepatocellular carcinoma after curative resection. BMC Gastroenterol 23, 320 (2023). https://doi.org/10.1186/s12876-023-02955-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12876-023-02955-5