Abstract
Background
The rs1520220 polymorphism in the insulin-like growth factor 1 (IGF1) gene has been reported to affect cancer susceptibly in several studies. However, the results of the relevant studies are inconsistent. We conduct a current meta-analysis to investigate the association between rs1520220 and cancer susceptibly.
Methods
Three databases (PubMed, Embase, and Web of Science) were searched for studies regarding the relationship between rs1520220 and cancer susceptibly. Odds ratios (ORs) and the related 95% confidence intervals (CIs) were employed to assess the strength of the associations. A stratified analysis was performed according to cancer type, ethnicity, and quality score, and when results were obtained from no fewer than two studies, these results were pooled.
Results
There was no positive association between rs1520220 and overall cancer risk. However, the analysis stratified by ethnicity revealed that rs1520220 significantly increased cancer susceptibility in Asian populations (allele model OR = 1.10, 95%Cl = 1.00–1.21, p = 0.040; homozygote model OR = 1.22, 95%Cl = 1.01–1.47, p = 0.040; dominant model OR = 1.19, 95%Cl = 1.01–1.39, p = 0.033). No significantly association was detected in Caucasian populations. The analysis stratified by cancer type suggested that rs1520220 was not associated with susceptibility to breast cancer.
Conclusions
The results of our meta-analysis demonstrate that the role of IGF1 rs1520220 in cancer susceptibility varies by ethnicity and cancer type and that rs1520220 increases cancer susceptibility in Asian populations.
Similar content being viewed by others
Background
The occurrence of cancer depends on both genetic and environmental factors [1, 2]. Relevant environmental factors include pollution, tobacco and alcohol intake, overweight, and infection [3]. Studies based on twins have found that genetic factors are also an important risk factor for cancer [2, 4]. Recently, the role of SNPs in the occurrence and development of cancer has attracted increasing attention [5]. The SNPs that are associated with cancer risk may act as biomarkers for cancer diagnosis [6, 7].
IGF1 is a growth factor that involves in many important biological and pathological processes [8, 9]. The important functions of IGF1 are promoting cell proliferation and inhibiting apoptosis [10]. IGF1 has also been reported to be involved in cancer development [11]. Plasma IGF1 levels depend on many factors, such as BMI, but gene is also an important factor [12, 13]. Many studies have reported that several IGF1 SNPs affect plasma IGF1 levels and thus influence the risk of cancer [14, 15].
rs1520220 is located in intron 3 of IGF1 gene which might lead to alternative splicing and a subsequent change in protein function [16]. It has been reported that rs1520220 G to C substitution leads to increased plasma IGF1 level, increasing cancer risk as a result [13, 17]. However, the studies regarding the relationship between rs1520220 and cancer susceptibility are inconsistent [18,19,20,21,22,23,24,25]. For instance, Al-Zahrani et al. reported that rs1520220 increased susceptibility to breast cancer [18], but Li et al. suggested that rs1520220 was not related to susceptibility to breast cancer [25]. Considering the disagreement between these studies, we performed a meta-analysis of the associations between rs1520220 and cancer susceptibility to review these results and draw a more accurate conclusion.
Methods
Search strategy
We searched for relevant studies in three databases: PubMed, Embase, and Web of Science. The search conditions limited the language to English and the data of publication prior to February 28, 2018. The following keywords were used: “IGF1 or IGF-1 or insulin-like growth factor 1 or rs1520220,” “cancer or tumor or carcinoma,” and “SNP or polymorphism or variant or mutation.” We also checked the references of the identified articles to ensure that we obtained all potentially relevant studies.
Inclusion and exclusion criteria
The inclusion criteria of this meta-analysis are as follows: studies must (1) concern the relationship between rs1520220 and cancer susceptibility, (2) be case-control or cohort study, and (3) contain sufficient genotyping data to allow for the pooling of the results (the GG, GC, and CC genotype frequencies in the case and control groups were provided directly or could be calculated from the provided data). The exclusion criteria are as follows: (1) when subjects of two studies overlap, the one containing fewer subjects was excluded, and (2) reviews and meta-analyses are excluded.
Data extraction
The following information was extracted from the included studies by two authors independently: first author’s name, year of publication, country, cancer type, ethnicity, genotyping methods, control source, genotype distributions of cases and controls, and Hardy-Weinberg equilibrium (HWE) for controls. Disagreements were resolved via discussion.
Quality score
We assessed the quality of the included studies based on the following five factors [26]: case source, control source, specimens used for determining genotypes, HWE in controls, and total sample size (Additional file 1: Table S1). A perfect score was 15.
Statistical analysis
We estimated the strengths of the associations using pooled ORs with corresponding 95% CIs. Five genetic models are employed: the allele model (C vs. G), the homozygote model (CC vs. GG), the heterozygote model (GC vs. GG), the dominant model (CC + GC vs. GG), and the recessive model (CC vs. GC + GG). The heterogeneity was evaluated using a Q test and quantified by I2 [27]. When heterogeneity not exists (P > 0.1), the fixed-effects model was used [28]. Otherwise, the random-effects model was applied [29]. Hardy-Weinberg equilibrium (HWE) for controls was assessed using a chi-squared test. P values less than 0.05 were considered to indicate significant disequilibrium. Stratified analyses were conducted by ethnicity, cancer type, and quality score. Only results synthesized from no fewer than two studies are shown. Sensitivity analyses were performed via metainf command which investigates the influence of each individual study on the overall meta-analysis summary estimate by omitting each study in turn [30]. Publication bias was assessed using Begg’s test and Egger’s test [31, 32]. All statistical analyses were performed using the STATA software (Version 12.0; Stata Corporation, College Station, TX, USA).
Results
Characteristics of the studies
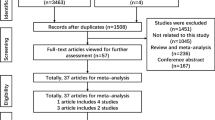
We obtained 2086 relevant articles through database searching after removing duplicates. Then, by screening the titles and abstracts, we excluded 1953 articles, and 133 articles remained. We read the full texts of these 133 articles and ultimately identified eight articles that meet the inclusion criteria (Fig. 1), which involved 12,884 cases and 58,304 controls. The characteristics of the included studies are shown in Table 1. Among these eight studies, four were carried out in Asian populations, two were carried out in Caucasian populations, and two were carried out in mix populations. Four of them concerned breast cancer, and four concerned other cancers including testicular germ cell tumors (TGCT), stomach cancer, pancreatic cancer, and colorectal cancer. Seven of the included studies had quality scores of no less than 12. The distributions of the genotypes and allele frequencies in the cases and controls are shown in Table 2.
Meta-analysis
We investigate the role of rs1520220 polymorphisms in cancer susceptibility via pooled OR and 95%Cl. Only results synthesized from no fewer than two studies are shown. In the overall analysis, we did not find positive associations between rs1520220 and cancer susceptibility (Table 3).
In the analysis stratified by ethnicity, we found that rs1520220 was significantly associated with increased cancer susceptibility in Asian populations (Table 3 and Fig. 2, allele model OR = 1.10, 95%Cl = 1.00–1.21, p = 0.040; homozygote model OR = 1.22, 95%Cl = 1.01–1.47, p = 0.040; dominant model OR = 1.19, 95%Cl = 1.01–1.39, p = 0.033). Thus, no significant association was detected in Caucasian populations (Table 3 and Fig. 2).
Stratification analyses by ethnicity between IGF1 rs1520220 polymorphism and cancer susceptibility. a Allele model. b Homozygous model, c Heterozygous model. d Dominant model. e Recessive model. The squares and horizontal lines correspond to the study specific OR and 95% CI. The area of the squares reflects the weight. The diamond represents the summary OR and 95% CI. The fixed-effects model was used
In the analysis stratified by cancer type, the results show that rs1520220 was not associated with susceptibility to breast cancer (Table 3). Also, the results synthesized from studies that scored no less than 12 did not exhibited any differences from the results of the overall analysis (Table 3).
Sensitivity analysis
We performed sensitive analysis and found there was not an individual study that affected the results of the overall analysis (Fig. 3 and Additional file 1: Table S2), indicating that in this meta-analysis, our results are relatively stable.
Publication bias
Begg’s test and Egger’s test were performed to assess the publication bias among the included studies. No publication bias was detected in the present meta-analysis (Fig. 4 and Table 4).
Discussion
Many SNPs have been reported to be associated with cancer susceptibility and thus may have the potentiality as biomarkers for clinical diagnosis [5,6,7]. Thus far, with the improvement of living standards in more and more developing countries, obesity and lifestyle-related cancers have been on the increase [33, 34]. IGF1 has been reported to relate to be associated with the cancer susceptibility, especially cancers caused by obesity, due to its important role in cell proliferation [35].
Several IGF1 SNPs have been reported to be associated with cancer susceptibility [36,37,38,39]. These SNPs include rs1520220, rs6214, rs6220, rs35767, and rs5742612. Of these, rs6214 and rs6220 are located in the 3′-UTR region of the IGF1 gene. It has been reported that rs6214 is associated with increased esophageal adenocarcinoma (EAC) and head and neck cancer (HNC) susceptibility in women [39] and that rs6220 is associated with increased prostate cancer susceptibility [40]. The rs35767 and rs5742612 SNPs are located in the promoter region of the IGF1 gene. It has been reported that rs35767 is significantly associated with increased susceptibility of childhood acute lymphoblastic leukemia (ALL) [41] and that rs5742612 is associated with increased susceptibility to prostate cancer [42].
rs1520220 is an SNP that is located in the intron of the IGF1 gene, and it has a minor allele frequency (MAF) about 10~ 40% in the populations included in the human 1000 Genomic Project phase 3 (Additional file 1: Table S3). We paid special attention to rs1520220 because it has been reported to be associated with plasma IGF1 levels in many studies and thus associated with cancer susceptibility [13, 17, 18].
In this meta-analysis, we systematically searched for literature on IGF1 SNPs and cancer in three important databases (PubMed, Embase, and Web of Science). After removing duplicate documents, 2086 related articles were initially obtained, which ensured the maximum possible recall rate. Through meta-analysis, we found that rs1520220 was not related to cancer susceptibility in the overall analysis based on the present epidemiology studies. Thus, in the analysis stratified by ethnicity, we revealed that rs1520220 increased cancer susceptibility in Asian populations.
The present studies regarding the effect of IGF1 rs1520220 polymorphism on serum IGF1 are inconsistent [13, 18]. In brief, rs1520220 may influence circulating IGF1 expression by altering the secondary structure of the RNA or DNA [16], and this effect may be enhanced by dietary factors [43]. Therefore, we infer that rs1520220 affects cancer susceptibility in Asians but not other populations due to the combined effects of genetic and environmental factors. The mechanism via which rs1520220 affects serum levels must be investigated in the future.
Our meta-analysis has several limitations. Firstly, we found that rs1520220 increased cancer susceptibility in Asians. The molecular mechanism via which the rs1520220 C allele increases plasma IGF1 levels and thus cancer risk remains unclear. Secondly, we did not consider potential external factors, such as gender, age, diet, and tobacco and alcohol intake habits or gene-gene interactions. Thirdly, the number of studies included in the meta-analysis is limited. We only included studies written in English, and important-related studies in other languages may have been overlooked.
Conclusion
The present meta-analysis showed that IGF1 rs1520220 is not significantly associated with overall cancer susceptibility. However, we did find that rs1520220 significantly increased cancer susceptibility in Asian populations. We also suggest that rs1520220 was not associated with susceptibility to breast cancer. There is a need for additional well-designed epidemiology and molecular biology studies to verify these conclusions and provide new insights into the role of SNPs in the etiology of cancer.
Abbreviations
- 3′-UTR:
-
three prime untranslated region
- ALL:
-
Acute lymphoblastic leukemia
- CI:
-
Confidence interval
- EAC:
-
Esophageal adenocarcinoma
- HB:
-
Hospital-based
- HNC:
-
Head and neck cancer
- HWE:
-
Hardy-Weinberg equilibrium
- IGF1:
-
Insulin-like growth factor 1
- MAF:
-
Minor allele frequency
- OR:
-
Odds ratio
- PB:
-
Population-based
- SNP:
-
Single nucleotide polymorphism
- TGCT:
-
Testicular germ cell tumors
References
Ekman P. Genetic and environmental factors in prostate cancer genesis: identifying high-risk cohorts. Eur Urol. 1999;35:362–9. https://doi.org/10.1159/000019910.
Lichtenstein P, Holm NV, Verkasalo PK, Iliadou A, Kaprio J, Koskenvuo M, Pukkala E, Skytthe A, Hemminki K. Environmental and heritable factors in the causation of cancer––analyses of cohorts of twins from Sweden, Denmark, and Finland. N Engl J Med. 2000;343:78–85. https://doi.org/10.1056/NEJM200007133430201.
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. https://doi.org/10.3322/caac.21262.
Sturm RA. Skin colour and skin cancer––MC1R, the genetic link. Melanoma Res. 2002;12:405–16.
Lei H, Hemminki K, Johansson R, Altieri A, Enquist K, Henriksson R, Lenner P, Forsti A. PAI-1-675 4G/5G polymorphism as a prognostic biomarker in breast cancer. Breast Cancer Res Treat. 2008;109:165–75. https://doi.org/10.1007/s10549-007-9635-3.
Zhu DQ, Zou Q, Hu CH, Su JL, Zhou GH, Liu P. XRCC1 genetic polymorphism acts a potential biomarker for lung cancer. Tumour Biol. 2015;36:3745–50. https://doi.org/10.1007/s13277-014-3014-6.
Matsuoka H, Makimura C, Koyama A, Fujita Y, Tsurutani J, Sakai K, Sakamoto R, Nishio K, Nakagawa K. Prospective replication study implicates the catechol-O-methyltransferase Val(158)Met polymorphism as a biomarker for the response to morphine in patients with cancer. Biomed Rep. 2017;7:380–4. https://doi.org/10.3892/br.2017.963.
Tao Y, Pinzi V, Bourhis J, Deutsch E. Mechanisms of disease: signaling of the insulin-like growth factor 1 receptor pathway––therapeutic perspectives in cancer. Nat Clin Pract Oncol. 2007;4:591–602. https://doi.org/10.1038/ncponc0934.
Pollak M. Insulin and insulin-like growth factor signalling in neoplasia. Nat Rev Cancer. 2008;8:915–28. https://doi.org/10.1038/nrc2536.
Pollak MN, Schernhammer ES, Hankinson SE. Insulin-like growth factors and neoplasia. Nat Rev Cancer. 2004;4:505–18. https://doi.org/10.1038/nrc1387.
Samani AA, Yakar S, LeRoith D, Brodt P. The role of the IGF system in cancer growth and metastasis: overview and recent insights. Endocr Rev. 2007;28:20–47. https://doi.org/10.1210/er.2006-0001.
Chen WF, Wong MS. Genistein enhances insulin-like growth factor signaling pathway in human breast cancer (MCF-7) cells. J Clin Endocrinol Metab. 2004;89:2351–9. https://doi.org/10.1210/jc.2003-032065.
Wang Q, Liu L, Li H, Tao P, Qi Y, Li J. Effects of high-order interactions among IGFBP-3 genetic polymorphisms, body mass index and soy isoflavone intake on breast cancer susceptibility. PLoS One. 2016;11:e0162970. https://doi.org/10.1371/journal.pone.0162970.
Tempfer CB, Hefler LA, Schneeberger C, Huber JC. How valid is single nucleotide polymorphism (SNP) diagnosis for the individual risk assessment of breast cancer? Gynecol Endocrinol. 2006;22:155–9. https://doi.org/10.1080/09513590600629175.
Poole EM, Tworoger SS, Hankinson SE, Baer HJ. Genetic variability in IGF-1 and IGFBP-3 and body size in early life. BMC Public Health. 2012;12:659. https://doi.org/10.1186/1471-2458-12-659.
Lu L, Risch E, Deng Q, Biglia N, Picardo E, Katsaros D, Yu H. An insulin-like growth factor-II intronic variant affects local DNA conformation and ovarian cancer survival. Carcinogenesis. 2013;34:2024–30. https://doi.org/10.1093/carcin/bgt168.
Gu F, Schumacher FR, Canzian F, Allen NE, Albanes D, Berg CD, Berndt SI, Boeing H, Bueno-de-Mesquita HB, Buring JE, et al. Eighteen insulin-like growth factor pathway genes, circulating levels of IGF-I and its binding protein, and risk of prostate and breast cancer. Cancer Epidemiol Biomark Prev. 2010;19:2877–87. https://doi.org/10.1158/1055-9965.EPI-10-0507.
Al-Zahrani A, Sandhu MS, Luben RN, Thompson D, Baynes C, Pooley KA, Luccarini C, Munday H, Perkins B, Smith P, et al. IGF1 and IGFBP3 tagging polymorphisms are associated with circulating levels of IGF1, IGFBP3 and risk of breast cancer. Hum Mol Genet. 2006;15:1–10. https://doi.org/10.1093/hmg/ddi398.
Chia VM, Sakoda LC, Graubard BI, Rubertone MV, Chanock SJ, Erickson RL, McGlynn KA. Risk of testicular germ cell tumors and polymorphisms in the insulin-like growth factor genes. Cancer Epidemiol Biomark Prev. 2008;17:721–6. https://doi.org/10.1158/1055-9965.EPI-07-0768.
Patel AV, Cheng I, Canzian F, Le Marchand L, Thun MJ, Berg CD, Buring J, Calle EE, Chanock S, Clavel-Chapelon F, et al. IGF-1, IGFBP-1, and IGFBP-3 polymorphisms predict circulating IGF levels but not breast cancer risk: findings from the breast and prostate cancer cohort consortium (BPC3). PLoS One. 2008;3:e2578. https://doi.org/10.1371/journal.pone.0002578.
Ennishi D, Shitara K, Ito H, Hosono S, Watanabe M, Ito S, Sawaki A, Yatabe Y, Yamao K, Tajima K, et al. Association between insulin-like growth factor-1 polymorphisms and stomach cancer risk in a Japanese population. Cancer Sci. 2011;102:2231–5. https://doi.org/10.1111/j.1349-7006.2011.02062.x.
Nakao M, Hosono S, Ito H, Watanabe M, Mizuno N, Yatabe Y, Yamao K, Ueda R, Tajima K, Tanaka H, et al. Interaction between IGF-1 polymorphisms and overweight for the risk of pancreatic cancer in Japanese. Int J Mol Epidemiol Genet. 2011;2:354–66.
Qian B, Zheng H, Yu H, Chen K. Genotypes and phenotypes of IGF-I and IGFBP-3 in breast tumors among Chinese women. Breast Cancer Res Treat. 2011;130:217–26. https://doi.org/10.1007/s10549-011-1552-9.
Simons CC, Schouten LJ, Godschalk RW, van Engeland M, van den Brandt PA, van Schooten FJ, Weijenberg MP. Genetic variants in the insulin-like growth factor pathway and colorectal cancer risk in the Netherlands cohort study. Sci Rep. 2015;5:14126. https://doi.org/10.1038/srep14126.
Li H, Zhao M, Wang Q, Liu L, Qi YN, Li JY. Genetic polymorphisms of insulin-like growth factor 1 and insulin-like growth factor binding protein 3, xenoestrogen, phytoestrogen, and premenopausal breast cancer. Curr Oncol. 2016;23:e17–23. https://doi.org/10.3747/co.23.2835.
Tian X, Dai S, Sun J, Jiang S, Jiang Y. Association between TP53 Arg72Pro polymorphism and leukemia risk: a meta-analysis of 14 case-control studies. Sci Rep. 2016;6 https://doi.org/10.1038/srep24097.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58. https://doi.org/10.1002/sim.1186.
Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959;22:719–48.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Steichen T. METANINF: Stata module to evaluate influence of a single study in meta-analysis estimation, Statistical Software Components S419201, Boston College Department of Economics. 2001. https://ideas.repec.org/c/boc/bocode/s419201.html.
Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–101.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Hendriks SH, Schrijnders D, van Hateren KJ, Groenier KH, Siesling S, Maas A, Landman GWD, Bilo HJG, Kleefstra N. Association between body mass index and obesity-related cancer risk in men and women with type 2 diabetes in primary care in the Netherlands: a cohort study (ZODIAC-56). BMJ Open. 2018;8:e018859. https://doi.org/10.1136/bmjopen-2017-018859.
Tarasiuk A, Mosinska P, Fichna J. The mechanisms linking obesity to colon cancer: an overview. Obes Res Clin Pract. 2018; https://doi.org/10.1016/j.orcp.2018.01.005.
LeRoy EC, Moore JH, Hu C, Martinez ME, Lance P, Duggan D, Thompson PA. Genes in the insulin and insulin-like growth factor pathway and odds of metachronous colorectal neoplasia. Hum Genet. 2011;129:503–12. https://doi.org/10.1007/s00439-010-0942-0.
Birmann BM, Tamimi RM, Giovannucci E, Rosner B, Hunter DJ, Kraft P, Mitsiades C, Anderson KC, Colditz GA. Insulin-like growth factor-1- and interleukin-6-related gene variation and risk of multiple myeloma. Cancer Epidemiol Biomark Prev. 2009;18:282–8. https://doi.org/10.1158/1055-9965.EPI-08-0778.
Feik E, Baierl A, Hieger B, Fuhrlinger G, Pentz A, Stattner S, Weiss W, Pulgram T, Leeb G, Mach K, et al. Association of IGF1 and IGFBP3 polymorphisms with colorectal polyps and colorectal cancer risk. Cancer Causes Control. 2010;21:91–7. https://doi.org/10.1007/s10552-009-9438-4.
Dong X, Li Y, Tang H, Chang P, Hess KR, Abbruzzese JL, Li D. Insulin-like growth factor axis gene polymorphisms modify risk of pancreatic cancer. Cancer Epidemiol. 2012;36:206–11. https://doi.org/10.1016/j.canep.2011.05.013.
Ong J, Salomon J, te Morsche RH, Roelofs HM, Witteman BJ, Dura P, Lacko M, Peters WH. Polymorphisms in the insulin-like growth factor axis are associated with gastrointestinal cancer. PLoS One. 2014;9:e90916. https://doi.org/10.1371/journal.pone.0090916.
Johansson M, McKay JD, Wiklund F, Rinaldi S, Verheus M, van Gils CH, Hallmans G, Balter K, Adami HO, Gronberg H, et al. Implications for prostate cancer of insulin-like growth factor-I (IGF-I) genetic variation and circulating IGF-I levels. J Clin Endocrinol Metab. 2007;92:4820–6. https://doi.org/10.1210/jc.2007-0887.
Chokkalingam AP, Metayer C, Scelo G, Chang JS, Schiffman J, Urayama KY, Ma X, Hansen HM, Feusner JH, Barcellos LF, et al. Fetal growth and body size genes and risk of childhood acute lymphoblastic leukemia. Cancer Causes Control. 2012;23:1577–85. https://doi.org/10.1007/s10552-012-0035-6.
Qian J, Zhou H, Chen J, Ding Q, Cao Q, Qin C, Shao P, Li P, Cai H, Meng X, et al. Genetic polymorphisms in IGF-I and IGFBP-3 are associated with prostate cancer in the Chinese population. PLoS One. 2014;9:e85609. https://doi.org/10.1371/journal.pone.0085609.
Wang Q, Liu L, Li H, McCullough LE, Qi YN, Li JY, Zhang J, Miller E, Yang CX, Smith JS. Genetic and dietary determinants of insulin-like growth factor (IGF)-1 and IGF binding protein (BP)-3 levels among Chinese women. PLoS One. 2014;9:e108934. https://doi.org/10.1371/journal.pone.0108934.
Funding
This work was supported by grants from the National Natural Science Foundation of China (grant no. 81601826).
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Author information
Authors and Affiliations
Contributions
GPX and LFW conceived and designed the experiments. GPX and LFW performed the experiments. GPX, LFW, and WYX analyzed the data. LFW and WXC contributed the reagents/materials/analysis tools. LFW and GPX wrote the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Additional file
Additional file 1:
Table S1. Quality score assessment. Table S2. Sensitivity analyses for IGF1 rs1520220 polymorphism and cancer susceptibility. Table S3. MAFs of IGF1 rs1520220 polymorphism in the populations from the 1000 Genomes Project Phase 3. (DOCX 30 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Xu, GP., Chen, WX., Xie, WY. et al. The association between IGF1 gene rs1520220 polymorphism and cancer susceptibility: a meta-analysis based on 12,884 cases and 58,304 controls. Environ Health Prev Med 23, 38 (2018). https://doi.org/10.1186/s12199-018-0727-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12199-018-0727-y