Abstract
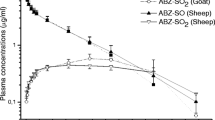
Proprietary formulations of sulfaphenazole were administered intravenously and orally to sheep. After intravenous injection the disposition of sulfaphenazole was described by an open two compartment model, and the elimination half-time was on average 5.58 h. The apparent volume of distribution was 0.273 1/kg and total body clearance 34.1 ml/kg/h. Judged from the area under the curves, the oral dose was completely absorbed, Drug plasma concentrations versus time fitted an open one compartment model, the half-time of absorption and elimination being 2.66 and 7.12 h, respectively. The binding to plasma proteins was high i.e. 93–96 % at therapeutic concentrations, and concentration dependent. The results demonstrate that the doses indicated by the manufacturer appear to be low and more appropriate for drugs with a longer elimination half-time. Consequently, considerable adjustments in the dosage regimen are recommended.
Sammendrag
Registrerte formuleringer av sulfafenazol (Eftolon®) ble gitt intravenøst og oralt til sau. Etter intravenøs injeksjon kunne konsentrasjonene av sulfonamid i plasma tilpasses en to-kompartment modell. Halveringstiden for eliminasjonen var i gjennomsnitt 5,58 t, tilsynelatende distribusjonsvolum 0,273 l/kg, og total clearance 34,1 ml/kg/t. Bedømt etter arealet under kurvene, ble sulfafenazol fullstendig absorbert etter oral dosering. Konsenstrasjoner i plasma ble beskrevet av en en-kompartmenit modell med halveringstider for absorpsjon og eliminasjon på henholdsvis 2,66 og 7,12 t. Binding til plasmaproteiner var konsentrasjonsavhengig, og varierte ved terapeutiske konsentrasjoner av sulfafenazol fra 93 til 96 %.
Resultatene viser at de doser som angis av produsenten er lave, foruten at doseintervallene passer bedre for farmaka med lengre halveringstid. Det er følgelig anbefalt vesentlige endringer i doseringsregime.
Article PDF
Similar content being viewed by others
References
Baggot, J. D.: Principles of Drug Disposition in Farm Animals: The Basis of Veterinary Clinical Pharmacology. W. B. Saunders Company, Philadelphia, USA 1977.
Gibaldi, M. & D. Perrier: Pharmacokinetics. In J. Swarbrick (ed.), Drugs and the Pharmaceutical Sciences. Vol. 1. Marcel Dekker, New York 1975.
Indrebø, A. & S. A. Ødegaard: Sulfametoxypyridaizin (Longamid®) og sulfanilamid til søye. En farmakokinetisk studie. (Sulfamethoxypyridazine (Longamid®) and sulfanilamide in the ewe. A pharmacokinetic study). Noirsk Vet.-tidsskr. 1986, 98, 183–188.
Liithmann, J. & S.-O. Jacobsson: Senimkoncentrationer av suifonamider efter oral och parenteral tillførsel. (Sulfonamide concentrations in blood serum after oral and parenteral administrar tion). Svensk Vet.-Tidn. 1979, 31, 783–787.
Nielsen, P. & F. Rasmussen: Half-life, apparent volume of distribution and protein-binding for some sulphonamides in cows. Res. Vet. Sci. 1977, 22, 205–208.
Pike, E. & B. Skuterud: Methodological aspects on the determination of plasma binding of amitriptyline and nortriptyline. Norv. pharm, acta 1983, 45, 33–42.
Sobach, S. & U. Lamminsiuu: The effect of protein-binding on the excretion of three sulphonamide preparations in the milk of dairy cows, examined by chemical and microbiological methods. Nord. Vet.-Med. 1979, 31, 309–315.
Ødegaard, S. A. & A. Råstad: Pharmacokinetics of sulfaphenazole in catte. J. vet. Pharmacol. Therap. 1986, 9, in press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ødegaard, S.A. Pharmacokinetics of Sulfaphenazole in Sheep. Acta Vet Scand 27, 243–249 (1986). https://doi.org/10.1186/BF03548168
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1186/BF03548168