Abstract
Background
Large cell neuroendocrine carcinoma is a very rare ovarian neoplasm that has a poor clinical outcome even in the early stage, and there is as yet no established treatment. Diagnostic laparoscopy has been used to determine the possibility of primary optimal cytoreductive surgery or neoadjuvant chemotherapy in patients with advanced epithelial ovarian cancer. However, the role of diagnostic laparoscopy is still unclear in large cell neuroendocrine carcinoma due to its rarity.
Case presentation
A 31-year-old woman with abdominal distention was referred to our hospital. She was strongly suspected of having advanced ovarian cancer because of a huge pelvic mass, massive ascites, and their appearance on medical imaging. However, cytological examinations from ascitic fluid by abdominal paracentesis did not show any malignant cells. She underwent diagnostic laparoscopy to evaluate the possibility of primary optimal cytoreductive surgery, and only tissue sampling was performed for pathological diagnosis because of the countless disseminated lesions of various sizes in the intraperitoneal organs. The patient had no postoperative complications, leading to the early start of postoperative chemotherapy.
Conclusions
To date, there have been no systematic reviews that focused on determining the treatment strategy using laparoscopy. Diagnostic laparoscopy can be helpful to determine the optimal treatment, including primary debulking surgery, neoadjuvant chemotherapy, or best supportive care, assisting in decision-making particularly for patients with advanced large cell neuroendocrine carcinoma with carcinomatous peritonitis.
Similar content being viewed by others
Introduction
Neuroendocrine carcinomas, particularly large-cell neuroendocrine carcinoma (LCNEC) of the ovary, are extremely rare but aggressive neoplasms. The other common subtypes of ovarian cancer such as high-grade serous, mucinous, or endometrioid carcinoma are often associated with LCNEC. LCNEC can follow a different clinical course with more adverse outcomes. Early and accurate diagnosis of LCNEC can be the best way to proceed to the optimal treatment and improve the prognosis of these patients. However, there are no optimal diagnostic and treatment strategies because of the paucity of evidence about the clinical or imaging features.
Currently, diagnostic laparoscopy has been used to determine the treatment strategy, including primary optimal cytoreductive surgery or neoadjuvant chemotherapy, in patients with advanced epithelial ovarian cancer, although the role of diagnostic laparoscopy is still unclear in LCNEC. A case of diagnostic laparoscopy with no perioperative complications resulting in the early start of postoperative chemotherapy is presented, along with a review of the literature to explain the role of diagnostic laparoscopy in selecting the optimal treatment for LCNEC patients.
Case presentation
A 31-year-old woman (gravida 0, para 0) visited the hospital due to abdominal distension. She had been in good health. Physical examination showed a markedly distended abdomen. Abdominal ultrasound (US) showed a pelvic mass and gross ascites. She was referred to our hospital for further examination and subsequent treatment.
Transvaginal US showed the presence of marked ascites and a large solid and cystic mass with a diameter of 20 cm around the uterus in the pelvic cavity. CT of the abdomen and pelvis showed gross ascites that extended under the diaphragm, a strongly enhanced, heterogeneous, huge mass in the pelvic cavity, and multiple peritoneal nodule lesions. There was no lymphadenopathy. On pelvic MRI, the pelvic mass showed homogeneous low intensity on T1-weighted MRI and heterogeneous low and high intensities on T2-weighted MRI, suggesting the presence of cystic and solid lesions. There were no fatty components. The patient’s serum CA 125 level was 165.7 U/ml (normal value < 35 U/ml). Serum CEA and CA 19–9 levels were within normal ranges. The pelvic cyst with solid components, the high CA 125 level, massive ascites, and multiple peritoneal nodule lesions strongly indicated the presence of an advanced ovarian cancer.
The patient underwent whole-body FDG PET/MRI to confirm malignancy and the presence of lymph node or distant metastases. The huge mass in the pelvic cavity and multiple peritoneal nodule lesions showed strong FDG uptake (Fig. 1a and b). She underwent abdominal paracentesis several times to confirm malignant cells in the ascitic fluid and reduce the abdominal discomfort caused by the massive ascites. However, cytological examinations showed only mesothelial cells without any malignant cells despite the imaging appearance of suspected malignancy on CT, MRI, and FDG-PET.
Whole-body FDG PET shows strong FDG uptake in multiple peritoneal nodule lesions (a). Integrated FDG-PET/T2-weighted MRI shows strong FDG uptake in the huge mass in the pelvic cavity (arrows) (b). Laparoscopic findings show the huge mass with a diameter greater than 20 cm in the pelvic cavity and strongly adhered to the adjacent organs (arrows) (c) and the countless disseminated lesions of various sizes in the intraperitoneal organs (arrows) (d)
Therefore, exploratory laparoscopy was performed for diagnostic purposes, and 6900 ml of bloody serous ascites were evacuated and obtained for cytology. A huge mass with a diameter of more than 20 cm occupied the pelvic cavity and adhered strongly to adjacent organs including the uterus, adnexa, and rectum (Fig. 1c). It was decided that the optimal surgery could not be performed because there were countless disseminated lesions of various sizes in the intraperitoneal organs, including the omentum, mesentery, and peritoneum (Fig. 1d). Therefore, one nodule of the omentum was resected by a harmonic device for frozen section examination, and it was diagnosed as adenocarcinoma. Additional resection of the omental nodule was performed for permanent fixation and further pathological examination. The pathological examination showed that the tumor consisted of small cells and large cells with hyperchromatic nuclei and a high mitotic rate, showing nested, trabecular, and pseudoglandular growth patterns (Fig. 2a). There was no other histologic subtype. Immunohistochemical analysis showed that these cells were positive for neuroendocrine markers such as synaptophysin and CD56, and the Ki-67 index was greater than 20% (Fig. 2b, c and d). Cytological examination of the obtained ascitic fluid showed no malignant cells. There were no other suspicious malignant lesions in the lung or digestive system on CT or FDG-PET. Therefore, the final pathological diagnosis was large cell neuroendocrine carcinoma of the ovaries with FIGO stage IIIC.
Hematoxylin and eosin-stained paraffin section of the tumor at × 40 magnification shows that the tumor consists of small cells and large cells with hyperchromatic nuclei and a high mitotic rate, with nested, trabecular, and pseudoglandular growth patterns (a). Immunohistochemical analysis at × 40 magnification shows that these cells are positive for the neuroendocrine markers, synaptophysin (b) and CD56 (c). The Ki-67 index is greater than 20% (d)
The patient’s postoperative course was uneventful. On the 11th day after surgery, she received the first course of chemotherapy with etoposide and cisplatin. After 2 courses of the chemotherapy, the symptoms of abdominal distension had improved, although there was residual tumor in the abdominal cavity. However, the residual tumor then increased rapidly, and the patient died 2 months after surgery.
Discussion and conclusions
Neuroendocrine carcinomas (NECs) are rare but aggressive types of neoplasms that are generally seen in the lungs or gastrointestinal tract, whereas they are very rarely seen at sites in the female genital tract including the ovaries. Family history of cancer, body mass index, diabetes mellitus, cigarette smoking and alcohol consumption have been reported as the potential risk factors for NECs of the pancreas, small intestine, and rectum, whereas the risk factors for NECs in the ovaries remain unknown [1]. The 2014 WHO classification of ovarian tumors included carcinoid and small-cell carcinoma, pulmonary type, but no separate category of NEC. Moreover, large-cell NEC arises very rarely in the ovary, although the WHO classification does not include it [2]. NECs account for only 0.1% of all ovarian cancers and are often associated with other epithelial carcinomas, such as high-grade serous, mucinous, or endometrioid carcinoma. Unlike carcinoid, small-cell and large-cell NECs of the ovaries often occur in young women, with a median age of 23.9 years, and are associated with a much poorer prognosis [3]. Surgery with the aim of diagnosis and complete resection and adjuvant chemotherapy with platinum-based chemotherapy have been used regardless of the paucity of data. Pathological examination shows large cells with significant pleomorphism, large nuclei with coarse and granular chromatin, prominent nucleoli, significant mitotic activity, and palisading with rosette formation. Immunohistochemical analysis shows positivity for one or more of the neuroendocrine markers such as synaptophysin, CD56, or chromogranin in at least 10% of tumor cells. In many cases, the early and accurate diagnosis of NEC can be the best way to improve the prognosis of NEC of the ovaries.
The clinicopathological features of 31 cases with FIGO stage I/II (Table 1) [4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24] and 25 cases with FIGO stage III/IV (Table 2) [7, 8, 14, 17, 18, 20, 25,26,27,28,29,30,31,32,33,34,35] were reviewed. A Kaplan-Meier survival curve (SPSS Statistics version 24; IBM, Armonk, NY) of all 53 cases excluding 3 cases without clinical outcome data showed total 5-year survival of 34.6% and median survival time of 17 months. There were no significant differences between the cases with FIGO stage I/II (n = 28) and III/IV (n = 25), with total 5-year survival of 38.8 and 29.2% and median survival time of 19 and 9 months, respectively (p = 0.458). These results were almost similar to the previous report by Oshita et al. [18], suggesting that cases with LCNEC showed much worse clinical outcomes than the other subtypes, such as epithelial ovarian cancer even in early disease, regardless of FIGO stage. In particular, cases with carcinomatous peritonitis (n = 11) showed significantly much poorer clinical outcomes than cases without carcinomatous peritonitis (n = 42), with median survival time of 7 and 20 months, respectively (p = 0.036), suggesting that a different therapeutic strategy should be considered in these cases (Fig. 3).
Kaplan-Meier survival curves for overall survival of LCNEC patients. The total 5-year survival is 34.6%, and median survival time is 17 months in all LCNEC patients (n = 53) (a). There are no significant differences between the cases with FIGO stage I/II (n = 28) and III/IV (n = 25), with total 5-year survival of 38.8 and 29.2% and median survival time of 19 and 9 months, respectively (p = 0.458) (b). The cases with carcinomatous peritonitis (n = 11) show significantly much worse clinical outcomes than cases without carcinomatous peritonitis (n = 42), with median survival time of 20 and 7 months, respectively (p = 0.036) (c)
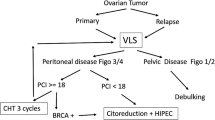
In advanced epithelial ovarian cancer patients with cancerous peritonitis and ascites, ascitic fluid cytology by abdominal paracentesis before surgery can be helpful and less invasive to confirm malignant cells in the ascitic fluid to proceed to neoadjuvant chemotherapy followed by interval debulking surgery with improvement of the prognosis [36]. In the present case, the patient was strongly suspected of having advanced ovarian cancer because of the huge pelvic mass, massive ascites, and their appearance on medical imaging. However, cytological examinations from ascitic fluid by abdominal paracentesis did not show any malignant cells. The previous study had shown that 96.7% of patients with carcinomatous peritonitis had positive ascitic fluid cytology [37]. In our review, only 2 of 5 cases with stage I/II and 2 of 4 cases with stage III/IV had positive ascitic fluid cytology. Only in 2 cases with carcinomatous peritonitis, including the present case, ascitic fluid cytology has been reported. The present case did not show any malignant cells in the ascitic fluid, whereas another one showed malignant cells consistent with papillary serous adenocarcinoma, which was the associated component in this case [29]. A previous study showed that the adenocarcinoma components were predominately located at the surface of the tumors, and most stromal and vascular invasion and lymph node metastases involved neuroendocrine components in the mixed adenoneuroendocrine carcinomas of hepatobiliary organs [38], suggesting that it might be difficult to identify malignant cells derived from pure LCNEC in ascitic fluid. Therefore, pathological examination has to be performed to differentiate non-epithelial ovarian cancer or other diseases including inflammation in these LCNEC cases. In the previous cases with stage III/IV [7, 8, 14, 17, 18, 20, 25,26,27,28,29,30,31,32,33,34], most patients underwent laparotomy for diagnosis and treatment. However, complete surgery could not be performed in many cases, leading to the deterioration of the patients’ general condition and much poorer outcomes. Therefore, a non-invasive method for differential diagnosis is needed.
The role of diagnostic laparoscopy to determine the possibility of primary optimal cytoreductive surgery in patients with advanced epithelial ovarian cancer has been reported. In a randomized, controlled trial involving patients with suspected advanced ovarian cancer, diagnostic laparoscopy was reported to reduce the number of futile laparotomies and be reasonable to proceed with primary cytoreductive surgery if cytoreduction to less than 1 cm of residual disease seems feasible [39]. Moreover, the same group has reported that diagnostic laparoscopy did not increase total direct medical health care costs or adversely affect complications or quality of life [40], suggesting that laparoscopy might be a potential diagnostic procedure in advanced epithelial ovarian cancer, although port-site metastasis occurs in 16–47% of cases, and the prognostic impact is still controversial [41].
In the previous cases with LCNEC, Herold et al. reported that diagnostic laparoscopy can be useful to achieve complete primary debulking surgery leading to better outcomes [35]. Oshita et al. reported the usefulness of neoadjuvant chemotherapy, leading to complete interval debulking surgery and long survival [18]. In the present case, the patient underwent diagnostic laparoscopy, and it was decided that the optimal surgery could not be performed because of the countless peritoneal lesions. Therefore, tissue sampling was performed without any intraoperative complications. The patient also had no postoperative complications, leading to the early start of postoperative chemotherapy. In terms of the chemotherapeutic regimen, the regimen against the epithelial component including paclitaxel and carboplatin could be considered in cases of mixed epithelial and LCNEC ovarian tumors, whereas the regimen against the neuroendocrine component including platinum-etoposide could be considered in cases of pure LCNEC [3, 42]. Therefore, the patient received the chemotherapy with etoposide and cisplatin, although postoperative chemotherapy did not improve the clinical outcome in the present case with carcinomatous peritonitis. Previous reports have shown the possible efficacy of paclitaxel and carboplatin, which can be less toxic than cisplatin in cases of even pure LCNEC of the ovary [9, 15, 20, 27, 33, 35], as well as in cases of NEC of the uterine cervix [43], suggesting that these chemotherapeutic regimens could be considered in patients with poor performance status and prognosis because of unresectable carcinomatous peritonitis. Taken together, in LCNEC, diagnostic laparoscopy followed by primary debulking surgery or neoadjuvant chemotherapy might be useful in cases without carcinomatous peritonitis, whereas it might also be useful for deciding whether patients should receive less invasive chemotherapy or best supportive care in cases with carcinomatous peritonitis with much poorer outcomes.
In summary, diagnostic laparoscopy could facilitate determination of subsequent treatment, including primary debulking surgery or neoadjuvant chemotherapy, in LCNEC patients. Moreover, it can also be useful for deciding whether to give adjuvant treatment or best supportive care to LCNEC patients with carcinomatous peritonitis who show much worse clinical outcomes.
Availability of data and materials
The data supporting the findings of this study are available within the article.
Abbreviations
- CT:
-
Computed tomography
- FDG:
-
Fluorodeoxyglucose
- FIGO:
-
International Federation of Gynecology and Obstetrics
- LCNEC:
-
Large cell neuroendocrine carcinoma
- MRI:
-
Magnetic resonance imaging
- PET:
-
Positron emission tomography
- US:
-
Ultrasonography
- WHO:
-
World Health Organization
References
Leoncini E, Carioli G, La Vecchia C, Boccia S, Rindi G. Risk factors for neuroendocrine neoplasms: a systematic review and meta-analysis. Ann Oncol. 2016;27(1):68–81.
Howitt BE, Kelly P, McCluggage WG. Pathology of neuroendocrine Tumours of the female genital tract. Curr Oncol Rep. 2017;19(9):59.
Gardner GJ, Reidy-Lagunes D, Gehrig PA. Neuroendocrine tumors of the gynecologic tract: a Society of Gynecologic Oncology (SGO) clinical document. Gynecol Oncol. 2011;122(1):190–8.
Collins RJ, Cheung A, Ngan HY, Wong LC, Chan SY, Ma HK. Primary mixed neuroendocrine and mucinous carcinoma of the ovary. Arch Gynecol Obstet. 1991;248(3):139–43.
Khurana KK, Tornos C, Silva EG. Ovarian neuroendocrine carcinoma associated with a mucinous neoplasm. Arch Pathol Lab Med. 1994;118(10):1032–4.
Jones K, Diaz JA, Donner LR. Neuroendocrine carcinoma arising in an ovarian mucinous cystadenoma. Int J Gynecol Pathol. 1996;15(2):167–70.
Eichhorn JH, Lawrence WD, Young RH, Scully RE. Ovarian neuroendocrine carcinomas of non-small-cell type associated with surface epithelial adenocarcinomas. A study of five cases and review of the literature. Int J Gynecol Pathol. 1996;15(4):303–14.
Chen KT. Composite large-cell neuroendocrine carcinoma and surface epithelial-stromal neoplasm of the ovary. Int J Surg Pathol. 2000;8(2):169–74.
Behnam K, Kabus D, Behnam M. Primary ovarian undifferentiated non-small cell carcinoma, neuroendocrine type. Gynecol Oncol. 2004;92(1):372–5.
Hirasawa T. Ovarian neuroendocrine carcinoma associated with mucinous carcinoma and teratoma. Nihon Rinsho. 2004;62(5):973–8.
Ohira S, Itoh K, Shiozawa T, Horiuchi A, Ono K, Takeuchi H, et al. Ovarian non-small cell neuroendocrine carcinoma with paraneoplastic parathyroid hormone-related hypercalcemia. Int J Gynecol Pathol. 2004;23(4):393–7.
Ahmed Z, Aftab K, Kayani N. Ovarian primary neuroendocrine carcinoma of non-small cell type: report of an extremely rare neoplasm. J Pak Med Assoc. 2005;55(2):82–4.
Lindboe CF. Large cell neuroendocrine carcinoma of the ovary. APMIS. 2007;115(2):169–76.
Veras E, Deavers MT, Silva EG, Malpica A. Ovarian nonsmall cell neuroendocrine carcinoma: a clinicopathologic and immunohistochemical study of 11 cases. Am J Surg Pathol. 2007;31(5):774–82.
Tartaglia E, Di Serio C, Rotondi M, Di Serio M, Scaffa C, Tolino A. Endometrial metastasis of a primitive neuroendocrine ovarian carcinoma: management and treatment of a case. Eur J Gynaecol Oncol. 2008;29(1):101–4.
Aslam MF, Choi C, Khulpateea N. Neuroendocrine tumour of the ovary. J Obstet Gynaecol. 2009;29(5):449–51.
Chenevert J, Bessette P, Plante M, Tetu B, Dube V. Mixed ovarian large cell neuroendocrine carcinoma, mucinous adenocarcinoma, and teratoma: a report of two cases and review of the literature. Pathol Res Pract. 2009;205(9):657–61.
Oshita T, Yamazaki T, Akimoto Y, Tanimoto H, Nagai N, Mitao M, et al. Clinical features of ovarian large-cell neuroendocrine carcinoma: four case reports and review of the literature. Exp Ther Med. 2011;2(6):1083–90.
Lee JJLJ, Lee DH, Kim MJ, Lee DJ. A case of ovarian large cell neuroendocrine carcinoma. Korean J Obstet Gynecol. 2012;55(1):43–6.
Ki EY, Park JS, Lee KH, Bae SN, Hur SY. Large cell neuroendocrine carcinoma of the ovary: a case report and a brief review of the literature. World J Surg Oncol. 2014;12:314.
Asada K, Kawana K, Teshima S, Saito A, Kawabata M, Fujii T. Poor prognosis of ovarian cancer with large cell neuroendocrine carcinoma: case report and review of published works. J Obstet Gynaecol Res. 2014;40(3):869–72.
Ding DC, Chu TY, Hsu YH. Large-cell neuro-endocrine carcinoma associated with borderline mucinous tumor of the ovary. Taiwan J Obstet Gynecol. 2014;53(2):270–2.
Sehouli J, Woopen H, Pavel M, Richter R, Lauterbach LK, Taube E, et al. Neuroendocrine neoplasms of the ovary: a retrospective study of the north eastern German Society of Gynecologic Oncology (NOGGO). Anticancer Res. 2016;36(3):1003–9.
Doganay M, Cengaver N, Kizilkanat KT, Oz M, Akbay S, Kokanali MK. Pure large cell neuroendocrine carcinoma of ovary: a rare clinical entity. J Exp Ther Oncol. 2019;13(1):55–8.
Choi YD, Lee JS, Choi C, Park CS, Nam JH. Ovarian neuroendocrine carcinoma, non-small cell type, associated with serous carcinoma. Gynecol Oncol. 2007;104(3):747–52.
Tsuji T, Togami S, Shintomo N, Fukamachi N, Douchi T, Taguchi S. Ovarian large cell neuroendocrine carcinoma. J Obstet Gynaecol Res. 2008;34(4 Pt 2):726–30.
Dundr P, Fischerova D, Povysil C, Cibula D. Primary pure large-cell neuroendocrine carcinoma of the ovary. Pathol Res Pract. 2008;204(2):133–7.
Yasuoka H, Tsujimoto M, Fujita S, Kunishige I, Nishio Y, Kodama R, et al. Monoclonality of composite large cell neuroendocrine carcinoma and mucinous epithelial tumor of the ovary: a case study. Int J Gynecol Pathol. 2009;28(1):55–8.
Draganova-Tacheva RA, Khurana JS, Huang Y, Hernandez E, Zhang X. Large cell neuroendocrine carcinoma of the ovary associated with serous carcinoma with mucin production: a case report and literature review. Int J Clin Exp Pathol. 2009;2(3):304–9.
Miyamoto M, Takano M, Goto T, Kato M, Sasaki N, Furuya K. Large cell neuroendocrine carcinoma arising in mature cystic teratoma: a case report and review of the literature. Eur J Gynaecol Oncol. 2012;33(4):414–8.
Shakuntala PN, Uma Devi K, Shobha K, Bafna UD, Geetashree M. Pure large cell neuroendocrine carcinoma of ovary: a rare clinical entity and review of literature. Case Rep Oncol Med. 2012;2012:120727.
Cokmert S, Demir L, Doganay L, Demir N, Kocacelebi K, Unek IT, et al. Large cell neuroendocrine carcinoma of the ovary and its skin metastases: a case report and review of the literature. West Indian Med J. 2014;63(6):667–72.
Lin CH, Lin YC, Yu MH, Su HY. Primary pure large cell neuroendocrine carcinoma of the ovary. Taiwan J Obstet Gynecol. 2014;53(3):413–6.
Agarwal L, Gupta B, Jain A. Pure large cell neuroendocrine carcinoma of the ovary with metastasis to cervix: a rare case report and review of literature. J Clin Diagn Res. 2016;10(9):ED01–ED3.
Herold N, Wappenschmidt B, Markiefka B, Keupp K, Krober S, Hahnen E, et al. Non-small cell neuroendocrine carcinoma of the ovary in a BRCA2-germline mutation carrier: a case report and brief review of the literature. Oncol Lett. 2018;15(4):4093–6.
Vergote I, Trope CG, Amant F, Kristensen GB, Ehlen T, Johnson N, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363(10):943–53.
Runyon BA, Hoefs JC, Morgan TR. Ascitic fluid analysis in malignancy-related ascites. Hepatology. 1988;8(5):1104–9.
Harada K, Sato Y, Ikeda H, Maylee H, Igarashi S, Okamura A, et al. Clinicopathologic study of mixed adenoneuroendocrine carcinomas of hepatobiliary organs. Virchows Arch. 2012;460(3):281–9.
Rutten MJ, van Meurs HS, van de Vrie R, Gaarenstroom KN, Naaktgeboren CA, van Gorp T, et al. Laparoscopy to predict the result of primary Cytoreductive surgery in patients with advanced ovarian Cancer: a randomized controlled trial. J Clin Oncol. 2017;35(6):613–21.
van de Vrie R, van Meurs HS, Rutten MJ, Naaktgeboren CA, Opmeer BC, Gaarenstroom KN, et al. Cost-effectiveness of laparoscopy as diagnostic tool before primary cytoreductive surgery in ovarian cancer. Gynecol Oncol. 2017;146(3):449–56.
Ataseven B, Grimm C, Harter P, Heikaus S, Heitz F, Traut A, et al. Prognostic impact of port-site metastasis after diagnostic laparoscopy for epithelial ovarian Cancer. Ann Surg Oncol. 2016;23(Suppl 5):834–40.
Voutsadakis IA. Large cell neuroendocrine carcinoma of the ovary: a pathologic entity in search of clinical identity. World J Clin Oncol. 2014;5(2):36–8.
Tempfer CB, Tischoff I, Dogan A, Hilal Z, Schultheis B, Kern P, et al. Neuroendocrine carcinoma of the cervix: a systematic review of the literature. BMC Cancer. 2018;18(1):530.
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
HT, KY, and TK performed the operation. HT, SY, TO, and MY were involved in acquisition of data and preparing the figures. HT wrote the manuscript. YY proofread and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This report was approved by the Hospital Ethics Committee of the University of Fukui.
Consent for publication
Written, informed consent was obtained from the patient for publication of this case report and any accompanying images.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Tsuyoshi, H., Yashiro, K., Yamada, S. et al. Role of diagnostic laparoscopy in patients with large cell neuroendocrine carcinoma of the ovary with cancerous peritonitis: case report and review of the literature. J Ovarian Res 12, 95 (2019). https://doi.org/10.1186/s13048-019-0571-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13048-019-0571-8