Abstract
Objectives
Limited aortic annulus exposure during minimal invasive aortic valve replacement (mini-AVR) proves to be challenging and contributes to procedure complexity, resulting in longer procedure times. New innovations like sutureless valves have been introduced to reduce procedure complexity. Additionally, preoperative imaging could also contribute to reducing procedure times. Therefore, we hypothesize that Computed Tomography (CT)-image based measurements are associated with mini-AVR complexity.
Methods
One hundred patients who underwent a mini-sternotomy and had a preoperative CT scan were included. With a CT-based mini-AVR planning tool, we measured access distance, access angle, annulus dimensions, and calcium volume. The associations of these measurements with cardiopulmonary bypass (CPB) time and aortic cross-clamp (AoX) time were assessed using univariable and multivariable regression models. In the multivariable models, these measurements were adjusted for age and suture technique.
Results
In the univariable regression models, calcium volume and annulus dimensions were associated with longer CPB and AoX time. After adjusting for age and suture technique, increasing calcium volume was still associated with longer CPB (adjusted β-coefficient 0.002, 95%-CI (0.005, 0.019), p-value = 0.002) and AoX time (adjusted β-coefficient 0.010, 95%-CI (0.004, 0.016), p-value = 0.002). However, after adjusting for these confounders, the association between annulus dimensions and procedure times lost statistical significance.
Conclusion
Increase in calcium volume are associated with longer CPB and AoX times, with age and sutureless valve implantation as independent confounders. In contrast to previous studies, access angle was not associated with procedure complexity.
Similar content being viewed by others
Introduction
Surgical valve replacement is standard treatment of severe aortic stenosis (AS). Minimally invasive aortic valve replacement (mini-AVR) is an alternative to conventional open-heart surgery to treat severe AS. Mini-AVR aims to minimize the degree of surgical intrusiveness by operating through a smaller incision (also known as ‘minimal access AVR’). Nevertheless, this procedure can still be considered an invasive surgical procedure. Because the term “Mini-AVR” has achieved common acceptance, we opted to use this as well in the remainder of this manuscript. Compared to conventional aortic valve replacement (CAVR), mini-AVR has shown to reduce transfusion incidences, postoperative pain, ICU stay, hospitalization, renal failure, and wound infection [1,2,3]. On the other hand, the limited exposure to the heart during mini-AVR makes myocardial protection and de-airing more challenging. Recent randomized controlled trials have shown that mini-sternotomy did not result in shorter hospital stay, faster recovery, improved survival, or less transfusion of blood compared to CAVR [4, 5]. In addition, mini-AVR has also been associated with increased cardiopulmonary bypass (CPB) time, aortic cross-clamp (AoX) time, overall operating time, and higher costs [6]. Several studies have demonstrated that prolonged CPB time and AoX time is associated with post-operative morbidity and mortality in both low- and high-risk cardiac patients [7,8,9,10]. An important advancement of mini-AVR procedures was the introduction of sutureless valves. Multiple studies have demonstrated that the use of sutureless valves reduces CPB and AoX time and are therefore associated with less post-operative complications [11,12,13,14].
To further improve mini-AVR procedures, preoperative imaging can be used to support surgical planning to decrease procedure complexity. 3D reconstructed Computer Tomography (CT)-images are commonly used for this goal, either for visual evaluation or for measuring distances between the incision and aortic valve [15,16,17,18,19].
In a recent study, it was found that the access angle, which is the angle between the aortic root and the incision at the annulus, was significantly associated with annulus access difficulty, CPB time, and AoX time in patients treated through a mini-sternotomy [20]. Other radiological characteristics that have been suggested to be associated with procedure complexity are the diameter of the aortic valve annulus, the distance from the ascending aorta to the sternum, and the extent of calcifications in the aortic valve and the ascending aorta [21, 22]. The reported association between access angle and procedural complexity was based on a relatively small patient population, which limited reliable statistical analysis for additional CT-based measures.
The primary purpose of this study is to determine whether CT-image based measurements are associated with mini-AVR surgical times. Additionally, we evaluate whether this hypothesized association of CT-image based parameters with the outcomes differ between sutured and sutureless valve replacement surgeries.
Methods
Study population
All patients that underwent mini-AVR at two institutes (Amsterdam UMC, location Academic Medical Center, The Netherlands; Onze Lieve Vrouwe Gasthuis, The Netherlands) and had a preoperative CT-scan between December 2014 and March 2018 were included in this study. The CT-imaging was not part of the treatment selection. Baseline characteristics, intraoperative data and postoperative outcomes were collected. Imaging kilovoltage ranged from 70 to 120 kV. The chest and abdomen were scanned using 1 bolus of contrast Iomeron 400 (Bracco Imaging SpA, Milan, Italy) ranging from 80 to 120 ml intravenously infused at a rate of 3.5–5.0 ml/s. In case patients had both CT and CT Angiography (CTA) acquisitions, the latter was used because of the higher contrast between the aortic root and surrounding structures. In case of dynamic CTA’s, the images acquired at 70% of the cardiac cycle (mid-diastole) were selected because at this phase the aortic valve is closed [20]. The image volumes contained approximately 400–900 slices, and each slice in a volume contained 512 × 512 isotropic pixels with a 16-bit depth. The slice thickness for the data sets ranged from 0.45–3.0 mm. The institutional review board approved the study design and waived informed consent since solely data obtained in the context of clinical care was utilized.
Surgical technique
All patients in this study underwent a mini-sternotomy performed by one of three operators (A.K., V.H. and R.C.). This approach was executed through a J-shaped incision starting from the sternal angle (manubriosternal joint) moving caudally for 4 cm in the sternum body to the 3rd intercostal space. After establishing cardiopulmonary bypass, the left ventricle was vented, and the aorta was cross-clamped to subsequently apply cardioplegia to stop the heart. The diseased valve was sharply excised and surrounding calcium was removed, after which a prosthetic aortic valve was implanted. The implanted valves were either manually sutured or mechanically fastened using the Cor-Knot device (LSI Solutions, Victor, NY, USA). Additionally, sutureless Perceval aortic bioprostheses (Sorin, Saluggia, Italy) were implanted.
CT-based mini-AVR planning tool
The mini-AVR planning tool is based on the 3mensio structural heart software (Pie Medical Imaging, Maastricht, The Netherlands) with additional options implemented for mini-AVR procedures. The tool allows the measurements of distances, angles, aortic valve dimensions, and calcifications. Furthermore, the tool automatically detects the aortic root, aortic annulus, and sinotubular junction and segments the ribcage in the CT scan. Additionally, the user can use a 3D probe to select an incision location on the 3D rendered chest cage for local measurements [20].
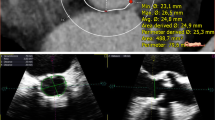
We measured the access distance, which is the distance between the incision location on the chest and the aortic sinotubular junction. We also measured the access angle, which is the angle between the aortic root axis and incision-annulus axis (Fig. 1). The aortic root centerline axis is the centerline that runs from the annulus towards the sinotubular junction. The incision-annulus centerline is the centerline under which the surgeon views the exposed aortic valve. In addition, the tool also provides the access distance and access angle as isocontour maps rendered on the 3D rendered CT volumes (Fig. 2). To mark the location of the planned incision, a 3D probe can be placed at the center of the manubriosternal joint. The hinge points of the aortic valve leaflets are automatically detected allowing the determination of the annulus dimensions such as minimum, maximum diameters, area, and perimeter. Annulus measurements were only measured in CTA. We measured the calcium volume quantitatively in mm3 after thresholding the Hounsfield Unit intensities to separate the calcifications from the enhanced blood and aortic wall. The calcium volume is calculated after setting a volume of interest including aortic annulus and leaflet. Calcifications in the left ventricular outflow tract, coronary arteries and ascending aorta are excluded [23].
Screenshot of the mini-AVR planning tool. The graphical user interface (GUI) shows the isocontours of the access distance and access angle. The access angle contours are colorized ranging from dark green (small angle) to white (large angle). The access distance contours are colorized ranging from dark blue (short distance) to white (long distance). The quantitative measures are displayed in the legend on the right: the left column of bars for angles and right column for distance
Outcome parameters
Surgical complexity was assessed by the CPB time and AoX time as proxies for procedure complexity.
Statistical analysis
Patients’ demographic and intra-operative data were expressed as mean (SD: standard deviation) for normally distributed values and as medians [interquartile range, IQR: 25th, 75th percentile] otherwise. Categorical data were presented as prevalence percentages.
Association of access distance, access angle, annular dimensions, and calcium volume with outcome parameters were tested with univariable linear regression.
Age and suture technique are known to also be associated with procedure complexity. Therefore, we adjusted for these confounders in multivariable analyses. The non-adjusted β1-coefficients and adjusted β1-coefficients with 95% Confidence Intervals (95%CI) of the linear regression models were presented.
To analyze whether there was a difference in the associations between sutured and sutureless valve replacement surgeries, an interaction term of the implantation technique and the CT-image based parameters that were significantly associated with the outcomes. The IBM SPSS Statistics, version 24 (IBM, Armonk, NY, USA) was used for statistical analysis.
Results
One hundred patients that underwent mini-AVR and had a pre-operative CT scan were included in this study. The median age was 71 [IQR: 63, 75] years and 34 patients were female. Sixteen patients presented New York Health Association (NYHA) functional class III or IV symptoms. The median predicted operative risk by means of logistic EuroSCORE I was 4.3 [IQR: 2.9, 6.3]. Ten patients had mild left ventricular dysfunction, while the rest had normal left ventricular function. Eighty-three patients had severe aortic stenosis and 30 patients had a bicuspid aortic valve. Baseline characteristics, cardiovascular comorbidities and hemodynamic data are listed in Table 1.
Fifty-five patients only underwent a CT and forty-five patients underwent both a CT and CTA. Ten scans were excluded for the access angle and access distance measurements: in these scan acquisitions the manubriosternal joint was not scanned, therefore it could not be used as a landmark for the incision location of our measurements. In total, 90 access distance and angle measurements were performed. For all patients the calcium volume was determined. Mean access angle was 42 (SD: 11) °, mean access distance was 86 (SD: 13) mm and the mean calcium volume was 689 (SD: 509) mm3. Forty-five annulus measurements were performed. The CT measurements are reported in Table 2.
CPB time and AoX time were 88 (SD: 24) and 63 (SD: 21) minutes, respectively. Ninety-nine patients had an isolated aortic valve replacement, with one mini-AVR procedure that was converted to a full sternotomy because the annulus could not be fully exposed to effectively perform the procedure. There was one patient that had a non-isolated aortic valve replacement, in which the patient required additional repair of the ascending aorta. Intra-operative data is summarized in Table 3.
In the univariable regression models, calcium volume, all annulus measures, sutureless valves, and age were significantly associated with CPB and AoX time (p < 0.05). Access angle and access distance were not associated with CPB and AoX time. Increasing levels of calcium volume and annulus measures showed an association with an increase in the CPB and AoX time, whereas advancing age and sutureless valves showed an association with a decrease in CPB and AoX time. After adjustment for confounding variables age and sutureless valves, only calcium volume was significantly associated with CPB (adjusted β-coefficient 0.002, 95%-CI (0.005, 0.019), p-value = 0.002) and AoX time (adjusted β-coefficient 0.010, 95%-CI (0.004, 0.016), p-value = 0.002) (Table 4).
There was no significant interaction found between suture technique and any of the annulus dimensions. Both suture technique and calcium were significant in the CPB and AoX models (p < 0.001), but their interaction coefficients were not (p = 0.24 for the CPB model and p = 0.15 for the AoX model).
Discussion
This study shows that annulus dimensions and calcium volume are associated with CPB and AoX time, however, after adjusting for age and suture technique, the association between annulus dimensions and procedure times lost statistical significance. With increasing size in annulus dimensions and calcium volumes, CPB and AoX time increases. The use of sutureless valves and increasing age showed an association with a decrease in surgical time. In contrast to previous findings, in our population, access angle and distance were not associated with the procedure complexity and outcome.
In the study of Ellatar et al. [20], access angle was significantly associated with AoX time. We did not find such an association of access angle with neither AoX time nor CPB time. Our population is an extension of that same population, which could suggest that this previous finding was due to the limited experience in the procedure. In another study of Gilmanov et al. [2], access angle and distance were suggested to be associated with outcome parameters, which also contradicts with our findings. In their study, access angle and access distance were defined as the distance from ascending aorta to sternum and the angle between the sternum midline and position of the ascending aorta at the level of the main pulmonary artery. The discrepancy between these findings and the present study could be due to the differences in treatment selection. In our study, only patients that were treated through mini-sternotomy were included in this study, whereas the study of Gilmanov included mini-sternotomy and mini-thoracotomy procedures. Another reason why access angle and distance might not be associated with outcome parameters, is that the surgeons who performed the surgery have increased in skill over the years, and thus have become less dependent on the aortic anatomy. In Martella et al. [24], preoperative CT scans were used to plan the incision for right thoracotomies. Their results showed that if the incision location is perpendicular to the plane of the aortic valve, the surgeon has a better view on the exposed valve. It was shown that for this procedure, anterior and medially positioned aortas are more challenging because the angle towards the valve becomes more difficult, especially for right thoracotomies. Our study indicates that procedure complexity decreases with age. This may be explained by the fact that the elasticity of the aortic annulus and ascending aorta diminishes with advancing age. In contrast to our study, a previous research showed that increased annulus size was associated with a shorter procedural time, which was explained by the fact that the increased size in aortic annulus diameter and the elongation of the aorta gives the surgeon more working space [25]. This relation was not found in our study.
Our findings suggest that with each increment 100 mm3 of calcium surgical procedure time increases approximately by 1 min. So, a severely calcified aortic valve with an approximate volume of 500 mm3 would increase the CPB and AoX time by 5 min, which is a considerable amount of time since the average of our CPB and AoX time was 88 and 63 min respectively.
Surgical times are reduced by using sutureless valves, which has been confirmed by multiple studies [25,26,27,28]. This suggests that the choice of sutureless valves over sutured valves should always to be considered to decrease AoX time, in order to diminish the chance of post-operative morbidity and mortality [8, 10, 29]. In our study, the use of sutureless valves showed an association with a decrease of AoX time approximately by 26 min on average, which is a significant amount on an average of 63 min. Our study also suggests that there is no interaction between the annular dimensions and the choice of suture technique, indicating that the relation of annulus measures and calcium volume on surgical time is similar for both techniques.
Mini-AVR and CAVR are not the only choice of intervention for AS. The patient can also be treated through the less invasive transcatheter aortic valve implantation (TAVI). TAVI is favoured for high-risk and intermediate-risk patients who clinically are frail and old and have an increased risk for surgery. Mini-AVR and CAVR are favoured for low-risk and intermediate-risk patients, who might have endocarditis and might require additional interventions like revascularization of the coronaries. Surgery is considered when the aortic valve annulus is out of range for TAVI, the aortic root morphology is unfavourable for TAVI, and when the morphology of the valve (bicuspid valves, degree of calcification) is unfavourable for TAVI [30]. For the therapeutic choice between TAVI, mini-AVR and CAVR, a dedicated heart team assesses each patient based on previous cardiologic history and baseline characteristics, calculate the risk of surgery, evaluate the feasibility of TAVI or surgery, and local experience. TAVI is associated with increased pacemaker implantation, vascular complications and paravalvular leakage [31]. For mini-AVR and CAVR these complications are less common. However bleeding complications, acute kidney injury and new-onset atrial fibrillation happen more frequent when compared with TAVI [32]. When an institute is able to perform both surgical as percutaneous valve replacement, the heart team can evaluate technical suitability and risk-benefit ratio and decide the best course of action.
Our study suffers from a number of limitations: Only a single rater performed the CT measurements. The measures used to assess procedure complexity (CPB and AoX time) is only a derivative of the complexity and may under- or overestimate the surgical complexity. Although various steps of the measurements have been automated, which should reduce interobserver variation, the robustness of these measures and their association with the outcome parameters could be validated by more raters. The surgeries have been performed by multiple surgeons, which might have influenced the variability in surgical time. The image data was not uniform over the whole dataset. Non-contrast CT, CTA and multi-phase CTA were used based on what was available, because it is not a standard procedure to perform a CT before mini-AVR surgery. The number of bicuspid aortic valves was larger in the sutured group of patients, which might indicate bias to the selection of type of valve.
Conclusion
We have shown that an increase in annulus size and calcium volume is associated with increased aortic cross-clamp and cardiopulmonary bypass time in patients with a mini-AVR. However, after adjusting for age and suture technique, the association between annulus dimensions and procedure times lost statistical significance.
Additionally, the study confirms that the implantation of sutureless valves and advancing age is associated with decreased aortic cross-clamp and cardiopulmonary bypass time. In contrast to previous studies, access angle was not associated with procedure complexity.
Availability of data and materials
The datasets used are available from the corresponding author on reasonable request.
Abbreviations
- AoX:
-
Aortic cross-clamp
- AS:
-
Aortic stenosis
- CAVR:
-
Conventional aortic valve replacement
- CC:
-
Creatine clearance
- CI:
-
Confidence Interval
- CPB:
-
Cardiopulmonary bypass
- CT:
-
Computed tomography
- CTA:
-
Computed tomography angiography
- EuroSCORE:
-
European System for Cardiac Operative Risk Evaluation
- GUI:
-
Graphical user interface
- ICU:
-
Intensive care unit
- IQR:
-
Interquartile range
- LVEF:
-
Left ventricular ejection fraction
- Mini-AVR:
-
Minimal invasive aortic valve replacement
- NYHA:
-
New York Heart Association
- SD:
-
Standard deviation
- TAVI:
-
Transcatheter aortic valve implantation
References
Bustamante J, Canovas S, Fernández ÁL. Minimally Invasive Aortic Valve Surgery - New Solutions to Old Problems. In: Aortic Stenosis - Etiology, Pathophysiology and Treatment: InTech; 2011. http://www.intechopen.com/books/aortic-stenosis-etiology-pathophysiology-and-treatment/minimally-invasive-aortic-valve-surgery-new-solutions-to-old-problems.
Gilmanov D, et al. Minimally invasive aortic valve replacement: 12-year single center experience. Ann Cardiothorac Surg. 2015;4(2):160–9.
Castrovinci S, et al. Minimally invasive aortic valve surgery. J Geriatr Cardiol. 2016;13(6):499–503.
Akowuah E, et al. Manubrium-limited ministernotomy versus conventional sternotomy for aortic valve replacement (MAVRIC): study protocol for a randomised controlled trial. Trials. 2017;18(1):46.
Nair SK, et al. Mini-Stern Trial: A randomized trial comparing mini-sternotomy to full median sternotomy for aortic valve replacement. J Thorac Cardiovasc Surg. 2018;156(6):2124–2132.e31.
Cooley DA. Antagonist’s view of minimally invasive heart valve surgery. J Card Surg. 2000;15(1):3–5.
Safi HJ, et al. Effect of extended cross-clamp time during thoracoabdominal aortic aneurysm repair. Ann Thorac Surg. 1998;66(4):1204–9.
Nissinen J, et al. Safe time limits of aortic cross-clamping and cardiopulmonary bypass in adult cardiac surgery. Perfusion. 2009;24(5):297–305.
Durandy YD, Younes M, Mahut B. Pediatric warm open heart surgery and prolonged cross-clamp time. Ann Thorac Surg. 2008;86(6):1941–7.
Al-Sarraf N, et al. Cross-clamp time is an independent predictor of mortality and morbidity in low- and high-risk cardiac patients. Int J Surg. 2011;9(1):104–9.
Di Eusanio M, Phan K. Sutureless aortic valve replacement. Ann Cardiothorac Surg. 2015;4(2):123–30.
Durdu MS, et al. Sutureless valve replacement through a right anterior mini-thoracotomy in elderly patients with Stenotic bicuspid aortic valve. Semin Thorac Cardiovasc Surg. 2018;31(3):458–64.
Fischlein T, Pfeiffer S, Pollari F, Sirch J, Vogt F, Santarpino G. Sutureless valve implantation via mini J-Sternotomy: a single center experience with 2 years mean follow-up. Thorac Cardiovasc Surg. 2015;63(06):467–71.
Gersak B, et al. Sutureless, rapid deployment valves and stented bioprosthesis in aortic valve replacement: recommendations of an international expert consensus panel. Eur J Cardio-Thoracic Surg. 2016;49(3):709–18.
Loor G, Desai MY, Roselli EE. Pre-operative 3D CT imaging for virtual planning of minimally invasive aortic valve surgery. JACC Cardiovasc Imaging. 2013;6(2):269–71.
Abu Saleh WK, et al. Direct aortic access Transcatheter aortic valve replacement: three-dimensional computed tomography planning and real-time fluoroscopic image guidance. J Heart Valve Dis. 2015;24(4):420–5.
Heuts S, Maessen JG, Nia PS. Preoperative planning of left-sided valve surgery with 3D computed tomography reconstruction models: Sternotomy or a minimally invasive approach? Interact Cardiovasc Thorac Surg. 2016;22(5):587–93.
Tops LF, et al. Noninvasive evaluation of the aortic root with multislice computed tomography. Implications for Transcatheter aortic valve replacement. JACC Cardiovasc Imaging. 2008;1(3):321–30.
Plass A, et al. Aortic valve replacement through a minimally invasive approach: preoperative planning, surgical technique, and outcome. Ann Thorac Surg. 2009;88(6):1851–6.
Elattar MA, et al. A computed tomography-based planning tool for predicting difficulty of minimally invasive aortic valve replacement. Interact Cardiovasc Thorac Surg. 2018;27(4):505–11.
Loor G, Roselli EE. Imaging and minimally invasive aortic valve replacement. Ann Cardiothorac Surg. 2015;4(1):62–6.
Glauber M, Ferrarini M, Miceli A. Minimally invasive aortic valve surgery: state of the art and future directions. Ann Cardiothorac Surg. 2015;4(1):26–32.
Elattar M, et al. Automatic aortic root landmark detection in CTA images for preprocedural planning of transcatheter aortic valve implantation. Int J Cardiovasc Imaging. 2016;32(3):501–11.
Martella A, Kernis S, Curiale S, Ndubizu U, Cichelli J. Right Thoracotomy Minimally Invasive AVR: Use of Preoperative CT Scan to Plan Incision. J Cardiothorac Surg. 2015;10(S1):A28.
Sugawara J, Hayashi K, Yokoi T, Tanaka H. Age-associated elongation of the ascending aorta in adults. JACC Cardiovasc Imaging. 2008;1(6):739–48.
Merk DR, et al. Minimal invasive aortic valve replacement surgery is associated with improved survival: a propensity-matched comparison†. Eur J Cardio-Thoracic Surg. 2015;47(1):11–7.
Miceli A, et al. Minimally invasive aortic valve replacement using right minithoracotomy is associated with better outcomes than ministernotomy. J Thorac Cardiovasc Surg. 2014;148(1):133–7.
Rosu C, Soltesz EG. Selection of valve prostheses. Semin Thorac Cardiovasc Surg. 2015;27(2):152–8.
Salis S, et al. Cardiopulmonary bypass duration is an independent predictor of morbidity and mortality after cardiac surgery. J Cardiothorac Vasc Anesth. 2008;22(6):814–22.
Baumgartner H, et al. 2017 ESC/EACTS guidelines for the management of valvular heart disease. Eur Heart J. 2017;38(36):2739–91.
Hoffmann CT, Heiner JA, Nguyen TC. Review of minimal access versus transcatheter aortic valve replacement for patients with severe aortic stenosis. Ann Cardiothorac Surg. 2017;6(5):498–503.
Siontis GCM, et al. Transcatheter aortic valve implantation vs. surgical aortic valve replacement for treatment of severe aortic stenosis: a meta-analysis of randomized trials. Eur Heart J. 2016;37(47):3503–12.
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
BB, VH, ES, AMK, ADW and EV contributed to the data collection of the manuscript. VH, BM, NP, AK and HA contributed to the study idea and conceptualization of the manuscript. VH, AK and RC performed the surgeries. All authors contributed to revisions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Amsterdam UMC Institutional Review Board and waived informed consent since solely data obtained in the context of clinical care was utilized.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Boti, B.R., Hindori, V.G., Schade, E.L. et al. Minimal invasive aortic valve replacement: associations of radiological assessments with procedure complexity. J Cardiothorac Surg 14, 173 (2019). https://doi.org/10.1186/s13019-019-0997-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13019-019-0997-5