Abstract
Background
Increasing evidence has suggested that the presence of remnant lipoproteins is a significant risk factor for atherosclerosis. Remnant lipoproteins are lipoproteins that are rich in triglycerides (TGs), and the main components include very-low-density lipoprotein (VLDL) in the fasting state. Diabetic patients often have hypertriglyceridemia with elevated levels of VLDL cholesterol but normal levels of low-density lipoprotein cholesterol (LDL-C). The aim of the present study was to elucidate the potential role of remnant lipoproteins-induced atherosclerosis in the occurrence and development of in-stent restenosis (ISR) in diabetic patients with coronary artery disease.
Methods
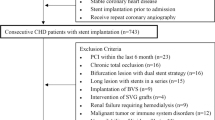
The present study enrolled 2312 patients with type 2 diabetes mellitus who underwent percutaneous coronary intervention from January 2013 to December 2014 and who were followed up by angiography. Patients were divided into two groups based on the presence or absence of ISR, and multivariate Cox’s proportional hazards regression modelling showed that remnant-like particle cholesterol (RLP-C) was an independent risk factor for ISR. According to the receiver operating characteristic curve, the optimal cutoff point of the RLP-C was identified, and the patients were further divided into 2 groups. Propensity score matching analysis was performed, and 762 pairs were successfully matched. Log-rank tests were used to compare Kaplan–Meier curves for overall follow-up to assess ISR.
Results
The multivariate Cox’s proportional hazards regression analysis showed that RLP-C was independently associated with ISR, and the baseline RLP-C level at 0.505 mmol/L was identified as the optimal cutoff point to predict ISR. Patients were divided into 2 groups by RLP levels. After propensity score matching analysis, a total of 762 pairs matched patients were generated. Kaplan–Meier curves showed that the estimated cumulative rate of ISR was significantly higher in patients with RLP-C levels ≥ 0.505 mmol/L (log-rank P < 0.001; HR equal to 4.175, 95% CI = 3.045–5.723, P < 0.001) compared to patients with RLP-C levels < 0.505 mmol/L.
Conclusions
The present study emphasized the importance of remnant-like particle cholesterol in cardiovascular pathology in diabetic patients. Physicians should take measures to control RLP-C below the level of 0.505 mmol/L to better prevent of in-stent restenosis in diabetic patients.
Similar content being viewed by others
Background
In-stent restenosis is an important factor for successful underwent percutaneous coronary intervention (PCI). Several large-scale clinical trials have confirmed that the occurrence rate of n-stent restenosis (ISR) ranges from 3 to 20% after coronary stenting implantation, even in the drug-eluting stents (DES) era [1]. Patients with diabetes mellitus (DM) have a 2–4 times higher risk of developing ISR after PCI compared with non-diabetic patients [2, 3]. The poor prognosis [4, 5] of this particular population deserves additional attention.
The high rate of ISR may be related to dyslipidaemia in DM. A recent multicentre study has shown that the prevalence of dyslipidaemia has reached up to 67.1% among diabetic patients in China [6] and is uniquely manifested by high levels of triglycerides (TGs) and very-low-density lipoprotein cholesterol (VLDL-C) but normal levels of low-density lipoprotein cholesterol (LDL-C) [7]. Remnant lipoproteins are lipoproteins that are rich in triglycerides (TGs), and the main components include VLDL in the fasting state [8]. Therefore, diabetic patients have abnormal lipid metabolism mainly due to increased remnant-like particle cholesterol (RLP-C). Recent studies have shown that high levels of remnant lipoproteins can predict coronary events in diabetic patients independent of the degree of coronary stenosis, age, gender, hypercholesterolaemia, low-density lipoprotein, hypertriglyceridaemia and other risk factors [9]. Therefore, it is of great significance to elucidate the role of RLP-induced atherosclerosis in diabetic patients.
Methods
Study patients
The present study is a retrospective cohort study of 2701 coronary artery disease (CAD) patients with Type 2 diabetes mellitus (T2DM) who underwent successful coronary second-generation drug-eluting stents (G2-DESs) implantation at Beijing Anzhen Hospital (Beijing, China) from January 2013 to December 2014 and were followed up by angiography. Patients who died in the hospital after baseline PCI or without sufficient clinical and angiographic data at baseline and follow up were excluded. Of these patients, 2312 patients who met the inclusion and exclusion criteria were analysed in the present study. Multivariate Cox’s proportional hazards regression modelling showed that RLP-C was an independent risk factor for ISR. According to the receiver operating characteristic (ROC), the optimal cutoff point of the RLP-C was identified, and the patients were divided into the following 2 groups: low RLP-C group (n = 1072) and high RLP-C group (n = 1240). Propensity score matching analysis was performed in the two groups with a proportion of 1:1, including baseline data (age, gender, BMI, duration of diabetes mellitus, symptom-driven hospitalization and SYNTAX score). Finally, 762 pairs of DM patients were successfully matched. Log-rank tests were used to compare Kaplan–Meier curves for overall follow-up to assess ISR between the two groups.
Stent implantation
All enrolled patients received G2-DESs implantations in the catheterization centre. The type of G2-DESs included zotarolimus-eluting stents (Endeavor and Endeavor Resolute; Medtronic Vascular, USA), domestic sirolimus-eluting stents (Firebird2; MicroPort Medical, China), everolimus-eluting stents (Xience V and Xience Prime; Abbott Vascular, USA, Promus and Promus Element; Boston Scientific, USA). Stent implantation was performed according to current practice guidelines, and stents were selected by experienced interventional cardiologists. During the procedure, patients received a bolus of 100 IU/kg heparin with a repeated bolus of 2000 IU heparin to maintain the activated clotting time of ≥ 300 s. All patients received aspirin (100 mg/day was administered) and clopidogrel (300 mg loading dose followed by 75 mg/day for at least 12 months). When ISR was diagnosed, patients were treated with re-DES implantation. Procedural success was defined as follows: reduction of stenosis to less than 10% residual narrowing; thrombolysis in myocardial infarction (TIMI) flow grade III; improvement in ischaemic symptoms; and no major procedure related complications [7].
Data collection
A standard case report form (CRF) was used to collect patients’ demographic and clinical characteristics, including age, gender, smoking, drinking, CAD risk factors, family history, life style, medical history and coronary angiographic information at baseline PCI and follow-up angiography. During a physical examination, anthropometric indices, such as weight, height and blood pressure (BP), were measured. Body mass index (BMI) was calculated as weight in kilograms divided by the square of the height in metres.
Coronary angiogram data, such as minimal stent diameter, average stent length and stenosis percent, were also recorded by two experienced investigators at baseline and follow-up for coronary angiography analysis.
Laboratory analysis
Venous blood samples were collected after an overnight fast for testing lipid profiles, HbA1c, fasting blood glucose (FBG), high-sensitivity C-reactive protein (hs-CRP) and uric acid (UA) levels using standard laboratory methods at baseline PCI and follow-up angiography.
The HbA1c was tested using ion exchange high-performance liquid chromatograph (HPLC) method. Blood samples for lipid profiles were collected from patients taking statin for more than 2 weeks. The total cholesterol (TC), TG, FBG and UA levels were determined according to enzymatic methods. LDL-C and high-density lipoprotein cholesterol (HDL-C) levels were measured by homogeneous assays. RLP-C levels were calculated as TC minus LDL-C and HDL-C according to the recommendation of dyslipidaemia guidelines [10, 11].
Disease definitions
The primary end point of the present study was the occurrence of ISR. ISR was defined as a diameter stenosis of ≥ 50% occurring in the segment inside the stent, 5 mm proximal to the stent or 5 mm distal to the stent at follow-up angiography [12]. The target lesion was considered as the most severe narrowing vessel identified by angiographic appearance with electrocardiograph (ECG) changes. Multivessel disease (MVD) was defined as a diameter stenosis of ≥ 50% occurring in 2 or more vessels.
Diabetes mellitus was defined as either a previous diagnosis of DM (treated with diet, oral agents or insulin) or a new diagnosis of DM (FBG ≥ 7.0 mmol/L on 2 occasions during hospitalization) [13]. Hypertension was defined by systolic blood pressure (SBP) ≥ 140 mmHg, diastolic blood pressure (DBP) ≥ 90 mmHg and/or the use of antihypertensive treatment in the past 2 weeks [14]. The severity of coronary artery lesions was quantified by the synergy between PCI with taxus and cardiac surgery (SYNTAX) score, which was calculated using the online calculator for SYNTAX score.
Statistical analysis
Continuous variables were expressed as the mean (\( {\bar{\text{X}}} \)) ± standard deviation (SD) in the case of normal distribution, and differences between two groups were determined by two-sided t-test. Data were expressed as medians (interquartile ranges, P25, and P75) in the case of skewed distribution and compared between two groups using the Mann–Whitney test. Categorical variables were presented as counts (percentages) and compared by Chi square test.
Univariate Cox’s proportional hazards regression modelling was performed to identify determinants of ISR in diabetic patients. Baseline variables were selected if they had either a clinically plausible relation with the ISR or appeared to be imbalanced between ISR and non-ISR patients with a P-value less than 0.2. The potential variables were entered into multivariate Cox’s proportional hazards regression modelling using the stepwise method (entry, 0.05; removal, 0.05) to determine their independent risk associated with ISR in diabetes. The hazard ratio (HR) and 95% confidence intervals (95% CIs) were calculated to estimate the adjusted risk of ISR in diabetic patients. The predictive value of the Cox’s regression model was evaluated using the area under the receiver operating characteristics curve (AUC).
According to the ROC, the optimal cutoff point of the RLP-C was identified, and patients were divided into 2 groups. Propensity score matching analysis was performed in the two groups with a proportion of 1:1. Log-rank tests were used to compare Kaplan–Meier curves for overall follow-up to assess ISR between the two groups.
Statistical analyses were performed using SPSS software for Windows (version 24.0, SPSS Inc., Chicago, Illinois, USA). A two-sided probability value of < 0.05 was considered statistically significant in all analyses.
Results
Baseline clinical and angiographic characteristics (Total population)
The baseline clinical and angiographic characteristics of the total population are shown in Table 1. Significant differences were observed between the ISR and non-ISR group in terms of smoking, medical history, TG, HDL-C and RLP-C. After adjusting for other confounding factors in the multivariate Cox’s proportional hazards regression, the RLP-C level was identified as one of the independent predictors associated with ISR in diabetic patients (Table 2). ROC curve analysis indicated that the AUC was 0.722 (95% CI = 0.693–0.751, P < 0.001), which showed a good predictive accuracy of RLP-C for the risk of ISR in diabetic patients after baseline PCI (Fig. 1). The baseline RLP-C level at 0.505 mmol/L (19.4 mg/dL) was identified as the optimal cutoff point to predict the risk of ISR with a sensitivity of 82.8% and a specificity of 52.0%.
Continuous variables were expressed as the mean (\( {\bar{\text{X}}} \)) ± standard deviation (SD) in the case of normal distribution and compared between two groups by the independent samples t-test. Data were expressed as medians (interquartile ranges) in the case of skewed distribution and compared using the Mann–Whitney U-test. Categorical variables are presented as counts (percentages) and compared by the Chi square test.
Baseline clinical characteristics, angiographic characteristics and Kaplan–Meier curves (propensity score matching population)
After propensity score matching analysis, a total of 762 pairs of matched patients were created. The C statistics for the propensity score model was 0.01. The baseline clinical characteristics before and after propensity score-matched analysis are shown in Fig. 2. No significant differences were observed between the low and high RLP groups after propensity score–matched analysis in terms of age, gender, BMI, medical history, duration of diabetes mellitus, HbA1c, fasting glycaemia, drug use, other biomarkers, angiographic characteristics and procedural characteristics.
Absolute standardized differences in unweighted and propensity score-weighted data sensitivity analyses. Given the difference in baseline variables between high RLP-C and low RLP-C groups, a propensity score–based method was performed to balance baselines of the two groups. Importantly, after propensity score matching, all between-group standardized differences were < 10. CAG coronary artery angiography, HDL-C high-density lipoprotein cholesterol, ARB angiotensin receptor blocker, LDL-C low-density lipoprotein cholesterol, FBG fasting blood glucose, ACEI angiotensin converting enzyme inhibitor, LAD left anterior descending, SYNTAX synergy between PCI with taxus and cardiac surgery, TG triglyceride, LVEF left ventricular ejection fraction, UA uric acid, DBP diastolic blood pressure
Log-rank tests were used to compare Kaplan–Meier curves for overall follow-up to assess ISR between the two groups (Fig. 3).
Discussion
Main findings
The present observational cohort study from a high volume cardiovascular centre in China revealed potential atherosclerosis resulting from remnant lipoproteins in the occurrence and development of in-stent restenosis in diabetic patients. The major findings were as follows: (1) the presence of remnant-like particle cholesterol is an independent risk factor for in-stent restenosis in diabetic patients; and (2) diabetic patients with high RLP-C levels (≥ 0.505 mmol/L) have greater risk for in-stent restenosis compared to patients with low RLP-C levels.
Abnormal lipid metabolism and atherosclerosis in DM
It is well known that LDL-C is the major risk factor for atherosclerosis and CVD [11, 15]. However, several recent meta-analyses have indicated that a high residual risk of CVD remains even in patients whose LDL-C levels reach the treatment target after statin treatment [16, 17]. Additionally, current dyslipidaemia guidelines recommend non-HDL-C as the primary target of lipid-lowering therapy [10], including VLDL-C, which is the major component of RLP-C during fasting.
However, diabetic patients have dyslipidaemia characterized by high levels of RLP-C [7] but normal levels of LDL-C. Existing research has shown that increased remnant lipoprotein level is a risk factor for ischaemic heart disease. With an empty stomach, an increase of 1 mmol/L residual lipoprotein increases ischaemic heart disease risk by 2.8 times [18]. Recently, prospective studies tracking coronary events in diabetic patients have shown that remnant-like particle cholesterol are the most important independent risk factors of coronary artery disease and can predict coronary events [19, 20].
Both in vitro and animal experiments have confirmed that the formation of atherosclerosis induced by increases in remnant lipoproteins is similar to the formation of atherosclerosis caused by the accumulation of lipid in the arterial wall induced by increases in low-density lipoprotein. Many studies have confirmed that the mechanism of atherosclerosis induced by remnant lipoproteins mainly manifests in the following aspects: (1) induction of proliferation of smooth muscle cells but no involvement in oxidative stress [21]; (2) induction of apoptosis in endothelial cells [22]; (3) induction of mononuclear/macrophage migration in endothelial cells [23]; (4) induction of McP-1 expression in umbilical venous blood endothelial cells as well as early growth response factor-1 (egr-1) mRNA and protein expression in vascular smooth muscle cells, which induces the occurrence of inflammation [24]; and (5) induction of elevated levels of other atherogenic lipoproteins.
Contrast and enlightenment
China has over 92.4 million diabetic patients (9.7% of the adult population), which ranks at the top with DM patient numbers and higher diabetes-related burden than other countries [25]. A single-centre study from Fuwai Hospital of China [26] reported that second-generation drug-eluting stents have reliable efficacy and safety in diabetic and non-diabetic patients. In the subgroup analysis of diabetic patients, the risk factors associated with target lesion revascularization included current smoker, a history of coronary heart disease, and old myocardial infarction. In the baseline data of this study, only the history of hyperlipidaemia was associated with blood lipids, and the specific content of each component of the relevant blood lipids was not included, especially remnant-like particle cholesterol (possibly due to the large difference in blood lipid spectrum between diabetic and non-diabetic people). All patients included in the present study were treated with second generation drug-eluting stents. The baseline contained the contents of various blood lipid components. Propensity score matching found that remnant lipoproteins played an important role in in-stent restenosis, which supplemented findings from previous studies. In the present study, the TG level was higher in the in-stent restenosis group, which was attributed to the TG-rich remnant lipoproteins [8]. Therefore, TG increased with the increase of remnant lipoproteins. Patients were divided into two groups according to the remnant cholesterol level, and the baseline data of the two groups were matched with propensity scores. After matching, there was no statistically significant difference in TG between the two groups, excluding the influence of TG on the results, which constituted a two-way verification process with the previous multi-factor analysis.
Previous studies [27] have proven that the association between disorders of TG metabolism and remnant-like particle cholesterol may account for the risk of CAD in diabetic patients. According to the relationship between the level of remnant-like particle cholesterol and the degree of in-stent restenosis confirmed by coronary angiography, the present study further confirmed that the remnant-like particle cholesterol rich in triglycerides play an important role in atherosclerosis in coronary heart disease patients with diabetes mellitus. Therefore, it is important to strengthen the management of remnant-like particle cholesterol in diabetic population in addition to the control of LDL-C levels required by the guidelines.
Statins play an irreplaceable role in secondary prevention of coronary heart disease. However, T2DM patients on statin therapy presenting increased levels of cholesterol remnants and triglycerides are prone to slight decreases in left ventricular systolic function [28], which severely affects the prognosis of diabetic patients with coronary heart disease. Therefore, in the secondary prevention of coronary heart disease in diabetic patients, it is important to use statins to reduce LDL-C while monitoring remnant-like particle cholesterol. Previous studies have confirmed that empagliflozin [29] and pemafibrate [30] lower remnant-like particle cholesterol, indicating that they have a good curative effect and are recommended to delay the progression of coronary atherosclerosis.
Limitations
Some limitations and strengths of the present study need to be acknowledged. First, the study was only a single-centre study, which may weaken the statistical power of the conclusions. Second, propensity score matching provides yields weaker evidence than randomized controlled trials.
Conclusions
In conclusion, the present study further emphasized the importance of atherogenic lipids and remnant particles in cardiovascular pathology (such as in-stent restenosis), especially in diabetic patients. Physicians should take measures to lower the level of remnant-like particle cholesterol to < 0.505 mmol/L to better prevent in-stent restenosis in diabetic patients.
Abbreviations
- ACEI:
-
angiotensin converting enzyme inhibitor
- ARB:
-
angiotensin receptor blocker
- BMI:
-
body mass index
- BP–DESs:
-
biodegradable polymer drug-eluting stent
- CAD:
-
coronary artery disease
- CRF:
-
case report form
- DBP:
-
diastolic blood pressure
- DM:
-
diabetes mellitus
- ECG:
-
electrocardiograph
- FBG:
-
fasting blood glucose
- G2-DESs:
-
second–generation drug-eluting stent
- GFR:
-
glomerular filtration rate
- HDL-C:
-
high density lipoprotein cholesterol
- HPLC:
-
high performance liquid chromatograph
- hs-CRP:
-
high-sensitivity C-reactive protein
- ISR:
-
in-stent restenosis
- LAD:
-
left anterior descending
- LCX:
-
left circumflex artery
- LDL-C:
-
low density lipoprotein cholesterol
- LM:
-
left main
- LVEF:
-
left ventricular ejection fraction
- MI:
-
myocardial infraction
- MVD:
-
multivessel disease
- Non-HDL-C:
-
non-high density lipoprotein cholesterol
- PCI:
-
percutaneous coronary intervention
- RCA:
-
right coronary artery
- ROC:
-
receiver operating characteristics
- RLP-C:
-
remnant-like particle cholesterol
- SBP:
-
systolic blood pressure
- SYNTAX:
-
synergy between PCI with taxus and cardiac surgery
- TC:
-
total cholesterol
- TG:
-
triglyceride
- UA:
-
uric acid
- VLDL-C:
-
very low density lipoprotein cholesterol
References
Dangas GD, Claessen BE, Caixeta A, Sanidas EA, Mintz GS, Mehran R. In-stent restenosis in the drug-eluting stent era. J Am Coll Cardiol. 2010;56:1897–907.
Daemen J, Wenaweser P, Tsuchida K, Abrecht L, Vaina S, Morger C, et al. Early and late coronary stent thrombosis of sirolimus-eluting and paclitaxel-eluting stents in routine clinical practice: data from a large two-institutional cohort study. Lancet. 2007;369:667–78.
Qin SY, Zhou Y, Jiang HX, Hu BL, Tao L, Xie MZ. The association of diabetes mellitus with clinical outcomes after coronary stenting: a meta-analysis. PLoS ONE. 2013;8:e72710.
De Miguel-Yanes JM, Jimenez-Garcia R, Hernandez-Barrera V, Mendez-Bailon M, de Miguel-Diez J, Lopez-de-Andres A. Impact of type 2 diabetes mellitus on in-hospital-mortality after major cardiovascular events in Spain (2002–2014). Cardiovasc Diabetol. 2017;16:126.
Noh M, Kwon H, Jung CH, Kwon SU, Kim MS, Lee WJ, et al. Impact of diabetes duration and degree of carotid artery stenosis on major adverse cardiovascular events: a single-center, retrospective, observational cohort study. Cardiovasc Diabetol. 2017;16:74.
Yan L, Xu MT, Yuan L, Chen B, Xu ZR, Guo QH, et al. Prevalence of dyslipidemia and its control in type 2 diabetes: a multicenter study in endocrinology clinics of China. J Clin Lipidol. 2016;10:150–60.
Zhao K, Li YJ, Gao S. Role of red blood cell distribution in predicting drug-eluting stent restenosis in patients with stable angina pectoris after coronary stenting. Coron Artery Dis. 2015;26:220–4.
Chapman MJ, Ginsberg HN, Amarenco P, Andreotti F, Boren J, Catapano AL, et al. Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: evidence and guidance for management. Eur Heart J. 2011;32:1345–61.
Kugiyama K, Doi H, Takazoe K, Kawano H, Soejima H, Mizuno Y, et al. Remnant lipoprotein levels in fasting serum predict coronary events in patients with coronary artery disease. Circulation. 1999;99:2858–60.
Jacobson TA, Ito MK, Maki KC, Orringer CE, Bays HE, Jones PH, et al. National lipid association recommendations for patient-centered management of dyslipidemia: part 1-full report. J Clin Lipidol. 2015;9:129–69.
Expert Dyslipidemia Panel of the International Atherosclerosis Society Panel Members. An International atherosclerosis society position paper: global recommendations for the management of dyslipidemia–full report. J Clin Lipidol. 2014;8:29–60.
Alfonso F, Byrne RA, Rivero F, Kastrati A. Current treatment of in-stent restenosis. J Am Coll Cardiol. 2014;63:2659–73.
Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2003;26(Suppl 1):S5–20.
Mansia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G, et al. 2007 ESH-ESC guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European society of hypertension (ESH) and of the European society of cardiology (ESC). Blood Press. 2007;16:135–232.
Board JB. Joint British societies’ consensus recommendations for the prevention of cardiovascular disease (JBS3). Heart. 2014;100(Suppl 2):1–67.
Baigent C, Blackwell L, Emberson J, Holland LE, Reith C, Bhala N, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376:1670–81.
Mills EJ, Wu P, Chong G, Ghement I, Singh S, Akl EA, et al. Efficacy and safety of statin treatment for cardiovascular disease: a network meta-analysis of 170,255 patients from 76 randomized trials. QJM. 2011;104:109–24.
Varbo A, Benn M, Nordestgaard BG. Remnant cholesterol as a cause of ischemic heart disease: evidence, definition, measurement, atherogenicity, high risk patients, and present and future treatment. Pharmacol Ther. 2014;141:358–67.
Mitsutake R, Miura S, Zhang B, Saku K. HDL-associated factors provide additional prognostic information for coronary artery disease as determined by multi-detector row computed tomography. Int J Cardiol. 2010;143:72–8.
Fukushima H, Sugiyama S, Honda O, Koide S, Nakamura S, Sakamoto T, et al. Prognostic value of remnant-like lipoprotein particle levels in patients with coronary artery disease and type II diabetes mellitus. J Am Coll Cardiol. 2004;43:2219–24.
Zhao D, Letterman J, Schreiber BM. Beta-migrating very low density lipoprotein (beta VLDL) activates smooth muscle cell mitogen-activated protein (MAP) kinase via G protein-coupled receptor-mediated transactivation of the epidermal growth factor (EGF) receptor: effect of MAP kinase activation on beta VLDL plus EGF-induced cell proliferation. J Biol Chem. 2001;276:30579–88.
Shin HK, Kim YK, Kim KY, Lee JH, Hong KW. Remnant lipoprotein particles induce apoptosis in endothelial cells by NAD(P)H oxidase-mediated production of superoxide and cytokines via lectin-like oxidized low-density lipoprotein receptor-1 activation: prevention by cilostazol. Circulation. 2004;109:1022–8.
Aramaki Y, Mitsuoka H, Toyohara M, Jinnai T, Kanatani K, Nakajima K, et al. Lectin-like oxidized LDL receptor-1 (LOX-1) acts as a receptor for remnant-like lipoprotein particles (RLPs) and mediates RLP-induced migration of vascular smooth muscle cells. Atherosclerosis. 2008;198:272–9.
Park SY, Lee JH, Kim YK, Kim CD, Rhim BY, Lee WS, et al. Cilostazol prevents remnant lipoprotein particle-induced monocyte adhesion to endothelial cells by suppression of adhesion molecules and monocyte chemoattractant protein-1 expression via lectin-like receptor for oxidized low-density lipoprotein receptor activation. J Pharmacol Exp Ther. 2005;312:1241–8.
Yang W, Lu J, Weng J, Jia W, Ji L, Xiao J, et al. Prevalence of diabetes among men and women in China. N Engl J Med. 2010;362:1090–101.
Tang XF, Ma YL, Song Y, Xu JJ, Yao Y, He C, et al. Biodegradable polymer drug-eluting stents versus second-generation drug-eluting stents in patients with and without diabetes mellitus: a single-center study. Cardiovasc Diabetol. 2018;17:114.
Tani S, Yagi T, Atsumi W, Kawauchi K, Matsuo R, Hirayama A. Relation between low-density lipoprotein cholesterol/apolipoprotein B ratio and triglyceride-rich lipoproteins in patients with coronary artery disease and type 2 diabetes mellitus: a cross-sectional study. Cardiovasc Diabetol. 2017;16:123.
van Stee MF, de Graaf AA, Groen AK. Actions of metformin and statins on lipid and glucose metabolism and possible benefit of combination therapy. Cardiovasc Diabetol. 2018;17:94.
Hattori S. Empagliflozin decreases remnant-like particle cholesterol in type 2 diabetes patients with insulin resistance. J Diabetes Investig. 2018;9:870–4.
Fruchart J-C. Pemafibrate (K-877), a novel selective peroxisome proliferator-activated receptor alpha modulator for management of atherogenic dyslipidaemia. Cardiovasc Diabetol. 2017;16:124.
Authors’ contributions
ZQ (first author) made substantial contributions to study conception, study design, data acquisition, data analysis, data interpretation and manuscript draft. YJZ (corresponding author) made substantial contributions to study conception, study design and intellectual content. They contributed equally to this work. KZ, YPL, JLW, WJC, CS, CPH and HH made substantial contributions to data acquisition. All authors read and approved the final manuscript.
Authors’ information
Yujie Zhou M.D., Ph.D. is the Vice President and Chief Physician of Beijing Anzhen Hospital, Capital Medical University, Professor of Medicine and Doctoral Supervisor of Cardiology, Capital Medical University, and Deputy Director of Beijing Institute of Heart, Lung and Blood Vessel Diseases. Prof. Zhou is Head of the National Center for Clinical Research of Cardiovascular Diseases, the National Key Clinical Specialties for Cardiovascular and Geriatric Diseases, Key Laboratory for Precision Medicine of Coronary Artery Disease, Centre for Clinical Management of Coronary Artery Disease of Beijing Anzhen Hospital, “Dengfeng” Talent Development Program and “Shiming” Talent Planning Team. Prof. Zhou was rewarded with Leading Personnel in Beijing Health System, Excellent Health and Technology Talent, New Century Talents Project, National Outstanding Contribution Expert, State Council Expert for Special Allowance, Honoured Doctor and Beijing Scholar.
Prof. Zhou served as the President of the College of SINO-EURO Coronary Heart Disease, Fellowship Member of the Society for Cardiovascular Angiography and Interventions (SCAI), Heart Rhythm Society (HRS), American College of Cardiology (ACC) and European Society of Cardiology (ESC). Prof. Zhou was also the previous Chairperson of the Chinese Medical Association, the Association of Interventional Cardiology, China Medical and Health International Exchange Promotion Committee, Honorary Chairman of China Geriatric Health Association (a branch of the Cardiovascular Disease Committee), Chairman of China Council for Prevention and Treatment of Chronic Diseases of Geriatric Health Care Research, Chairman of the Chinese Society of Geriatrics, the Committee on Precise Medical Treatment of Cardiovascular Diseases, Vice Chairman of Chinese Medical Doctor’s Association, Vice Chairman of Geriatrics Committee of Chinese gerontology Society, Vice Chairman of Beijing Society of Cardiology, Chinese Medical Association, Vice Chairman of China Society of Geriatric Cardiology and Cerebrovascular Disease Specialized Committee, Secretary General of Chinese Society of Cardiology and Expert Consultation of Central Health Committee.
Prof. Zhou was the Editor of Angiology, Editor in Chief of Journal of Cardiovascular and Pulmonary Diseases, Associate Editor of Chinese Journal of Interventional Cardiology, and Associate Editor of European Heart Journal (Chinese). Prof. Zhou published more than 280 papers in peer-reviewed SCI journals with cumulative impact factor more than 500 and has undertaken National Keypoint Research and Interventional Programs of the Thirteenth “Precise Diagnosis and Treatment of Coronary Heart Disease and Atrial Fibrillation”. Dr. Zhou was the National Intervention Advisor for coronary artery disease and structural heart disease as a specialist in percutaneous coronary intervention for complex and higher-risk patients (total PCI volume > 20,000).
Acknowledgements
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
The datasets generated and analysed for this study are available from the corresponding author upon reasonable request.
Consent for publication
Not applicable.
Ethics approval and consent to participate
The present study was approved by the Clinical Research Ethics Committee of Beijing Anzhen Hospital, Capital Medical University, and all patients provided written informed consent for participation in the present study.
Funding
The present work was supported by the Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (Code: ZYLX201303, XMLX201601), a Grant from National Key Research and Development Program of China (2017YFC0908800), Beijing Municipal Administration of Hospitals’ Ascent Plan (Code: DFL20150601) and Mission plan (Code: SML20180601).
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Qin, Z., Zhou, K., Li, Yp. et al. Remnant lipoproteins play an important role of in-stent restenosis in type 2 diabetes undergoing percutaneous coronary intervention: a single-centre observational cohort study. Cardiovasc Diabetol 18, 11 (2019). https://doi.org/10.1186/s12933-019-0819-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12933-019-0819-z