Abstract
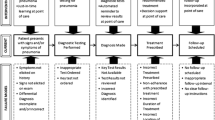
Clearly defined, documented, and managed processes form the foundation for how we effectively develop medicines for our patients. For this reason, process has been identified as a primary “element” of an effective quality management system (QMS) as described in the TransCelerate clinical quality management system (CQMS) conceptual framework. The importance of identifying and effectively managing processes is also emphasized in ICH GCP E6 (R2) in the new Section 5.0 Quality Management. An effective process management framework is fundamental to ensure the efficient and effective delivery of clinical development programs, enhance quality and productivity, and ultimately benefit our ability to deliver needed treatments to patients. The aim of this paper is to provide a conceptual process management framework to be used as guidance for effective process mapping, process documentation, implementation of optimal learning methods, and ensuring ongoing process performance evaluation and continuous improvement.
Article PDF
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
References
Meeker-O’Connell A, Borda MM, Little JA, Sam LM. Enhancing quality and efficiency in clinical development through a clinical QMS conceptual framework: concept paper vision and outline. Therapeutic Innovation & Regulatory Science. 2015;49(5):615–622.
Suprin, M, Chow A, Pillwein M, et al. Quality risk management framework: guidance for successful implementation of risk management in clinical development. Therapeutic Innovation & Regulatory Science. 2018; 1–9; DOI: https://doi.org/10.1177/2168479018817752.
Salzano KA, Maurer CA, Wyvratt JM, et al. A knowledge management framework and approach for clinical development. Therapeutic Innovation & Regulatory Science. 2016;50(5):536–545.
van Merrienboer JJG, Clark RE, de Croock MBM. Blueprints for complex learning: the 4C/ID-model. ETR&D. 2002;50(2):39–64.
Meeker-O’Connell A, Sam LM, Bergamo N, Little JA. TransCelerate’s clinical quality management system: from a vision to a conceptual framework. Therapeutic Innovation & Regulatory Science. 2016;50(4):397–413.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Husovich, M.E., Zadro, R., Zoller-Neuner, L.L. et al. Process Management Framework: Guidance to Successful Implementation of Processes in Clinical Development. Ther Innov Regul Sci 53, 25–35 (2019). https://doi.org/10.1177/2168479018817751
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1177/2168479018817751