Abstract
Background
Official drug prices have been set by the government and revised every other year in Japan. However, most drug prices are revised based on the price divergence rate of official and delivery prices every other year. This paper overviews how the drug price revision process works and reveals factors associated with price cutting, based on the price divergence rate.
Methods
Drugs approved as new molecular entities from 2005 to 2014 were selected for price cutting analysis, based on the price divergence rate. Logistic regression assessed the determinants of whether drugs were targeted for price cutting in 2016. Additionally, generic drugs approved from 2009 to 2013 and their off-patent drugs were also analyzed by multiple linear regression analysis to reveal determinants related to price divergence rates.
Results
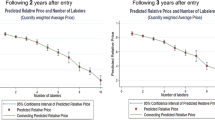
From the targeted on-patent drugs, 90% were targeted for price revision based on the price divergence rate. Of these drugs, 23% were targeted for price cutting. Drugs whose characteristics are larger sales, lower cost/patient, being followers, and being longer in the market were more likely to be targeted for price cutting. The number of generic manufacturers is directly proportional to the price cutting of both generic and off-patent branded drugs.
Conclusions
As drug price revision policy is under reconstruction in Japan, the characteristics of drugs targeted for price cutting shown in this study should be considered for establishing drug price revision policies that reflect market conditions adequately.
Similar content being viewed by others
References
Schoonveld E. The Price of Global Health: Drug Pricing Strategies to Balance Patient Access and the Funding of Innovation. New York, NY: Taylor & Francis; 2016.
Takayama A, Narukawa M. Pharmaceutical pricing and reimbursement in Japan: for faster, more complete access to new drugs. Therapeutic Innovation & Regulatory Science. 2016;50(3):361–367.
Shibata S, Uemura R, Suzuki T. Evaluating the effectiveness of repricing for market expansion in the Japanese drug pricing system. Therapeutic Innovation & Regulatory Science. 2016;50(6):751–758.
Shibata S, Uemura R, Suzuki T. Factors that affect the acquisition of reward premiums for promotion of innovative drug discovery in Japan. Therapeutic Innovation & Regulatory Science. 2016;50:56–65.
Shibata S, Uemura R, Suzuki T. Impact of premium rewards for the promotion of innovative drug discovery on the Japanese pharmaceutical market: an analysis by therapeutic area. Therapeutic Innovation & Regulatory Science. 2016;50:49–55.
Takayama A, Narukawa M. Investigation of factors affecting the degree of price gap between the National Health Insurance reimbursement price and the actual market price of new drugs [only abstract in English]. Regulatory Science of Medical Products. 2016;6(3):295–305.
Wakutsu N, Nakamura H. Analyzing the benefits from the new NHI drug pricing system in Japan: factor decomposition and simulation [only abstract in English]. Iryo to Syakai. 2015;25(2):205–220.
Central Social Insurance Medical Council. Minutes of Special Committee on Drug Prices, 2005-2014. Tokyo, Japan: Central Social Insurance Medical Council (Chuikyo). http://www.mhlw.go.jp/stf/shingi/shingi-chuo.html?tid=128154. Accessed August 9, 2016.
Japanese Ministry of Health, Labour and Welfare. http://www.mhlw.go.jp/topics/2016/04/tp20160401-01.html. Accessed August 9, 2016.
QuintilesIMS. IMS-Base JPM. Pharmaceutical Market Database. https://www.ims-japan.co.jp/japanese/s_navigator_domestic/data/. Accessed April 7, 2017.
EvaluatePharma Database. https://www.evaluategroup.com/public/Products-and-Solutions.aspx. Accessed April 7, 2017.
Joel L. The effect of generic competition on the price of brand-name drugs. Health Policy. 2004;68(1):47–54.
Grabowski H, Vernon J. Brand loyalty entry and price competition in pharmaceuticals after 1984 drug act. J Law Econ. 1992;35(2):331–350.
Frank RG, Salkever DS. Generic entry and the pricing of pharmaceuticals. J Econ Manage Strat. 1997;6(1):75–90.
Conti R, Berndt E. Specialty drug prices and utilization after loss of U.S. Patent exclusivity, 2001–2007. NBER Working Paper No. 20016. 2014.
Hudson J. Generic take-up in the pharmaceutical market following patent expiry: A multi-country study. Int Rev Law Econ. 2000;20:205–221.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukumoto, D., Tsuyuki, A. & Suzuki, T. Drugs Targeted for Price Cutting in Japan: The Case of Price Revisions Based on the Divergence of Official Versus Delivery Prices. Ther Innov Regul Sci 51, 597–603 (2017). https://doi.org/10.1177/2168479017705157
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1177/2168479017705157