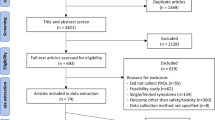
Abstract
Randomized controlled trials (RCTs) usually place less emphasis on the harmful effects than on the efficacy of interventions. The 10 CONSORT (Consolidated Standards of Reporting Trials) recommendations for harms reporting aim to improve harms data reporting of RCTs. The aim of this study was to assess the reporting of harms data in adjuvant trastuzumab studies in early-stage breast cancer. The resources PubMed, Cochrane Library, the American Society of Clinical Oncology, and the San Antonio Breast Cancer Symposium were searched for relevant RCTs that met the eligibility criteria. Each RCT was reviewed to determine whether the reporting of data complied with the 10 CONSORT recommendations for harms reporting, and the frequency of compliance with each CONSORT recommendation criterion was reviewed. Five RCTs met the eligibility criteria. Overall, selected RCTs failed to adhere to CONSORT recommendations in all sections of reporting. These results suggest that there is a need to standardize harms data reporting.
Similar content being viewed by others
References
Ethgen M, Boutron I, Baron G, Giraudeau B, Sibilia J, Ravaud P. Reporting of harm in randomized, controlled trials of nonpharmacologic treatment for rheumatic disease. Ann Intern Med. 2005;143(1):20–25.
Breau RH, Gaboury I, Scales CD, Fesperman SF, Watterson JD, Dahm P. Reporting of harm in randomized controlled trials published in the urological literature. J Urol. 2010;183:1693–1697.
Hayes DF, Picard MH. Heart of darkness: the downside of trastuzumab. J Clin Oncol. 2006;24(25):4056–4058.
Subar M, WenLong L, Chen W, Pittman DG. Lack of uniformity in cardiac assessment during trastuzumab therapy. Breast J. 2011;14(4):383–390.
National Cancer Institute. Phase III randomized study of two different regimens of adjuvant chemotherapy and trastuzumab (Herceptin) in women with HER2-postive stage I-IIIA breast cancer. https://cancer.gov/clinicaltrials/search/view?cdrid=584446&version=healthprofessional. Accessed November 10, 2011.
Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353(16):1659–1672.
Romond EH, Perez EA, Bryant J, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positve breast cancer. N Engl J Med. 2005;353(16):1673–1684.
Joensuu H., Bono P, Kataja V, et al. Fluorouracil, epirubicin, and cyclophosphamide with either docetaxel or vinorelbine, with or without trastuzumab, as adjuvant treatments of breast cancer: final results of FinHer Trial. J Clin Oncol. 2009;27(34):5685–5692.
Spielmann M, Roche H, Delozier T, et al. Trastuzumab for patients with axillary-node-positive breast cancer: result of FNCLCC-PACS 04 trial. J Clin Oncol. 2009;27(36):6129–6134.
Bordin P, Volpe C, Adami G, Moretti V, Ermacora P. Irreversible cardiotoxicity after adjuvant treatment with trastuzumab in a case of breast cancer. Tumori. 2008;95(5):777–778.
Ioannidis PA, Evans SJ, GØtzsche PC, et al. Better reporting of harms in randomized trials: an extension of the CONSORT statement. Ann Intern Med. 2004;141(10):781–788.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mahinbakht, A., Lavasani, S.M. & Guirguis, M. The Quality of Reporting Harms-Related Data in Clinical Trials of Adjuvant Trastuzumab in Early-Stage Breast Cancer Treatment. Ther Innov Regul Sci 48, 299–304 (2014). https://doi.org/10.1177/2168479013509383
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1177/2168479013509383