Abstract
Background
Young maternal age is associated with poorer birth outcomes, but the mechanisms are incompletely understood. Using data from a prospective cohort of pregnant women living in Mumbai slums, India, we tested whether lower maternal age was associated with adverse fetal growth.
Methods
Fetal crown-rump length (CRL) was recorded at a median (interquartile range, IQR) of 10 weeks’ gestation (9-10 weeks). Head circumference (HC), biparietal diameter (BPD), femur length (FL), and abdominal circumference (AC) were recorded at 19 (19-20) and 29 (28-30) weeks. Newborns were measured at a median (IQR) of 2 days (1-3 days) from delivery. Gestation was assessed using prospectively collected menstrual period dates.
Results
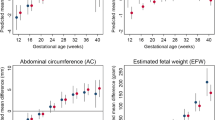
The sample comprised 1653 singleton fetuses without major congenital abnormalities, of whom 1360 had newborn measurements. Fetuses of younger mothers had smaller CRL (0.01 standard deviation [SD] per year of maternal age; 95% confidence interval CI: 0.00-0.021; P =.04), and smaller HC, FL, and AC at subsequent visits. Fetal growth of HC (0.04 cm; 95% CI: 0.02-0.05; P <.001), BPD (0.01 cm; 95% CI: 0.00-0.01; P =.009), FL (0.04 cm; 95% CI: 0.02-0.06; P <.001), and AC (0.01 cm; 95% CI: 0.00-0.01; P =.003) up to the third trimester increased with maternal age. Skinfolds, head, and mid-upper arm circumferences were smaller in newborns of younger mothers. Adjusting for maternal prepregnancy socioeconomic status, body mass index, height, and parity attenuated the associations between maternal age and newborn size but did not change those with fetal biometry.
Conclusion
Fetuses of younger mothers were smaller from the first trimester onward and grew slower, independently of known confounding factors.
Similar content being viewed by others
References
Bottomley C, Daemen A, Mukri F, et al. Assessing first trimester growth: the influence of ethnic background and maternal age. Hum Reprod. 2009;24(2):284–290.
Fall C, Sachdev H, Osmond C, et al. Association between maternal age at childbirth and child and adult outcomes in the offspring: a prospective study in five low-income and middle-income countries (COHORTS collaboration). Lancet Glob Health. 2015;3(7):e366–e377.
Kramer K, Lancaster J. Teen motherhood in cross-cultural perspective. Ann Hum Biol. 2010;37(5):613–628.
Bakker R, Steegers E, Biharie A, Mackenbach J, Hofman A, Jaddoe V. Explaining differences in birth outcomes in relation to maternal age: the Generation R Study. BJOG. 2011;118(4):500–509.
Mook-Kanamori D, Steegers E, Eilers P, Raat H, Hofman A, Jaddoe V. Risk factors and outcomes associated with first-trimester fetal growth restriction. JAMA. 2010;303(6):527–534.
Jolly M, Sebire N, Harris J, Robinson S, Regan L. The risks associated with pregnancy in women aged 35 years or older. Hum Reprod. 2000;15(11):2433–2437.
Kenny L, Lavender T, McNamee R, O’Neill S, Mills T, Khashan A. Advanced maternal age and adverse pregnancy outcome: evidence from a large contemporary cohort. PLos One. 2013;8(2):e56583.
Kozuki N, Lee A, Silveira M, et al. The associations of parity and maternal age with small-for-gestational-age, preterm, and neonatal and infant mortality: a meta-analysis. BMC Public Health. 2013;13(suppl 2):S2.
King J. The risk of maternal nutritional depletion on poor outcomes increases in early or closely spaced pregnancies. J Nutr. 2003;133(5 suppl 2):1732S–1736S.
Lamminpää R, Vehviläinen-Julkunen K, Gissler M, Heinonen S. Preeclampsia complicated by advanced maternal age: a registry-based study on primiparous women in Finland 1997–2008. BMC Pregnancy and Childbirth. 2012;12:47.
Potdar R, Sahariah S, Gandhi M, et al. Improving women’s diet quality preconceptionally and during gestation: effects on birth weight and prevalence of low birth weight—a randomized controlled efficacy trial in India (Mumbai Maternal Nutrition Project). Am J Clin Nutr. 2014;100(5):1257–1268.
Lawande A, Di Gravio C, Potdar R, et al. Effect of a micronutrient-rich snack taken preconceptionally and throughout pregnancy on ultrasound measures of fetal growth: the Mumbai Maternal Nutrition Project (MMNP). Matern Child Nutr. 2018;14(1):e12441.
International Institute for Population Sciences. National Family Health Survey (NFHS-2). India, 1998-99.Maharashtra, Mumbai: International Institute for Population Sciences; 2001.
Chopra H, Chheda P, Kehoe S, et al. Dietary habits of female urban slum-dwellers in Mumbai, India. Indian J Matern Child Health.2012;14(2):1–13.
Hadlock F. Sonographic estimation of fetal age and weight. Radiol Clin North Am. 1990;28(1):39–50.
Papageorghiou A, Ohuma E, Altman D, et al. International standards for fetal growth based on serial ultrasound measurements: the Fetal Growth Longitudinal Study of the INTERGROWTH-21st Project. Lancet. 2014;384(9946):869–879.
Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements—a prospective study. Am J Obstet Gynecol. 1985;151(3):333–337.
Villar J, Ismail LC, Victora CG, et al. International standards for newborn weight, length, and head circumference by gestational age and sex: the Newborn Cross-Sectional Study of the INTERGROWTH-21st Project. Lancet. 2014;384(9946):857–868.
Cole T, Green P. Smoothing reference centile curves; the LMS method and penalized likelihood. Stat Med. 1992;11(10):1305–1319.
Rigby R, Stasinopoulos D. Generalized additive models for location, scale and shape (with discussion). J R Stat Soc Ser C Appl Stat. 2005;54:507–554.
Gaillard R, Steegers E, de Jongste J, Hofman A, Jaddoe V. Tracking of fetal growth characteristics during different trimesters and the risk of adverse birth outcomes. Int J Epidemiol.2014;43(4):1140–1153.
Thame M, Hsu J, Gibson R, et al. Adaptation of in vivo amino acid kinetics facilitates increased amino acid availability for fetal growth in adolescent and adult pregnancies alike. Br J Nutr. 2014;112(11):1779–1786.
Hsu J, Thame M, Gibson R, et al. Unlike pregnant adult women, pregnant adolescent girls cannot maintain glycine flux during late pregnancy because of decreased synthesis from serine. Br J Nutr. 2016;115(5):759–763.
Barker D. Mothers, Babies and Disease in Later Life. Edinburgh, Scotland: Churchill Livingstone; 1995.
Jaquiery A, Oliver M, Honeyfield-Ross M, Harding J, Bloomfield F. Periconceptional undernutrition in sheep affects adult phenotype only in males. J Nutr Metab. 2012;2012:123610. doi: https://doi.org/10.1155/2012/123610
Rumball C, Bloomfield F, Oliver M, Harding J. Different periods of periconceptional undernutrition have different effects on growth, metabolic and endocrine status in fetal sheep. Pediatr Res. 2009;66(12):605–613.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Gravio, C., Lawande, A., Potdar, R.D. et al. The Association of Maternal Age With Fetal Growth and Newborn Measures: The Mumbai Maternal Nutrition Project (MMNP). Reprod. Sci. 26, 918–927 (2019). https://doi.org/10.1177/1933719118799202
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719118799202