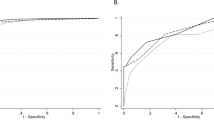
Abstract
This article presents an account of the research carried out so far in the use of metabolomics to find biomarkers of preterm birth (PTB) in fetal, maternal, and newborn biofluids. Metabolomic studies have employed mainly nuclear magnetic resonance spectroscopy or mass spectrometry–based methodologies to analyze, on one hand, prenatal biofluids (amniotic fluid, maternal urine/ maternal blood, cervicovaginal fluid) to identify predictive biomarkers of PTB, and on the other hand, biofluids collected at or after birth (amniotic fluid, umbilical cord blood, newborn urine, and newborn blood, maternal blood, or breast milk) to assess and follow up the health status of PTB babies. Besides advancing on the biochemical knowledge of PTB metabolism mainly during the in utero period and at birth, the work carried out has also helped to identify important requirements related to experimental design and analytical protocol that need to be addressed, if translation of these biomarkers to the clinic is to be envisaged. An outlook of possible future developments for the translation of laboratory results to the clinic is presented.
Similar content being viewed by others
References
Blencowe H, Cousens S, Chou D, et al; Born Too Soon Preterm Birth Action Group. Born too soon: the global epidemiology of 15 million preterm births. Reprod Health. 2013;10(suppl 1):S2.
Howson CP, Kinney MV, McDougall L, Lawn JE; Born Too Soon Preterm Birth Action Group. Born too soon: preterm birth matters. Reprod Health. 2013;10(suppl 1):S1.
Gracie S, Pennell C, Ekman-Ordeberg G, et al; PREBIC “-Omics” Research Group. An integrated systems biology approach to the study of preterm birth using “omic” technology—a guideline for research. BMC Pregnancy Childbirth. 2011;11(1):71.
Law K, Han TL, Tong C, Baker P. Mass spectrometry-based proteomics for pre-eclampsia and preterm birth. Int J Mol Sci. 2015;16(5):10952–10985.
Sheikh IA, Ahmad E, Jamal MS, et al. Spontaneous preterm birth and single nucleotide gene polymorphisms: a recent update. BMC Genomics. 2016;17(Suppl 9):759.
Johnson CH, Ivanisevic J, Siuzdak G. Metabolomics: beyond biomarkers and towards mechanisms. Nat Rev Mol Cell Biol. 2016;17(7):451–459.
Tolstikov V. Metabolomics: bridging the gap between pharmaceutical development and population health. Metabolites. 2016; 6(3):20.
Gowda GAN, Zhang S, Gu H, Asiago V, Shanaiah N, Raftery D. Metabolomics-based methods for early disease diagnostics. Expert Rev Mol Diagn. 2008;8(5):617–633.
Duarte IF, Diaz SO, Gil AM. NMR metabolomics of human blood and urine in disease research. J Pharm Biomed Anal. 2014;93: 17–26.
Bingol K, Bruschweiler-Li L, Li D, Zhang B, Xie M, Bru¨schweiler R. Emerging new strategies for successful metabolite identification in metabolomics. Bioanalysis. 2016;8(6):557–573.
Chen T, He P, Tan Y, Xu D. Biomarker identification and pathway analysis of preeclampsia based on serum metabolomics. Biochem Biophys Res Commun. 2017;485(1):119–125.
Virgiliou C, Gika HG, Witting M, et al. Amniotic fluid and maternal serum metabolic signatures in the second trimester associated with preterm delivery. J Proteome Res. 2017;16(2): 898–910.
Diaz SO, Pinto J, Barros AS, et al. Newborn urinary metabolic signatures of prematurity and other disorders: a case control study. J Proteome Res. 2016;15(1):311–325.
Law KP, Zhang H. The pathogenesis and pathophysiology of gestational diabetes mellitus: deductions from a three-part longitudinal metabolomics study in China. Clin Chim Acta. 2017;468: 60–70.
Dunn WB, Brown M, Worton SA, et al. Changes in the metabolic footprint of placental explant-conditioned culture medium identifies metabolic disturbances related to hypoxia and pre-eclampsia. Placenta. 2009;30(11):974–980.
Kenny LC, Broadhurst DI, Dunn W, et al; Screening for Pregnancy Endpoints Consortium. Robust early pregnancy prediction of later preeclampsia using metabolomic biomarkers. Hypertension. 2010;56(4):741–749.
Dunn WB, Brown M, Worton SA, et al. The metabolome of human placental tissue: investigation of first trimester tissue and changes related to preeclampsia in late pregnancy. Metabolomics. 2012;8(4):579–597.
Pinto J, Maciel E, Melo TS, et al. Maternal plasma phospholipids are altered in trisomy 21 cases and prior to preeclampsia and preterm outcomes. Rapid Commun Mass Spectrom. 2014; 28(14):1635–1638.
Austdal M, Skra°stad RB, Gundersen AS, Austgulen R, Iversen AC, Bathen TF. Metabolomic biomarkers in serum and urine in women with preeclampsia. PLoS One. 2014;9(3):e91923.
Graça G, Duarte IF, Barros AS, et al. Impact of prenatal disorders on the metabolic profile of second trimester amniotic fluid: a nuclear magnetic resonance metabonomic study. J Proteome Res. 2010;9(11):6016–6024.
Diaz SO, Barros AS, Goodfellow BJ, et al. Second trimester maternal urine for the diagnosis of trisomy 21 and prediction of poor pregnancy outcomes. J Proteome Res. 2013;12(6):2946–2957.
Graça G, Diaz SO, Pinto J, et al. Can biofluids metabolic profiling help to improve healthcare during pregnancy? Spectrosc Int J. 2012;27:515–523.
Sachse D, Sletner L, Mørkrid K, et al. Metabolic changes in urine during and after pregnancy in a large, multiethnic populationbased cohort study of gestational diabetes. PLoS One. 2012; 7(12):e52399.
Bentley-Lewis R, Huynh J, Xiong G, et al. Metabolomic profiling in the prediction of gestational diabetes mellitus. Diabetologia. 2015;58(6):1329–1332.
Pinto J, Almeida LM, Martins AS, et al. Prediction of gestational diabetes through NMR metabolomics of maternal blood. J Proteome Res. 2015;14(6):2696–2706.
Dessi` A, Atzori L, Noto A, et al. Metabolomics in newborns with intrauterine growth retardation (IUGR): urine reveals markers of metabolic syndrome. J Matern Neonatal Med. 2011;24(suppl 2): 35–39.
Sanz-Cortés M, Carbajo RJ, Crispi F, et al. Metabolomic profile of umbilical cord blood plasma from early and late intrauterine growth restricted (IUGR) neonates with and without signs of brain vasodilation. PLoS One. 2013;8(12):e80121.
Barberini L, Noto A, Fattuoni C, et al. Urinary metabolomics (GC-MS) reveals that low and high birth weight infants share elevated inositol concentrations at birth. J Matern Neonatal Med. 2014;27(suppl 2):20–26.
Dessi` A, Marincola FC, Pattumelli MG, et al. Investigation of the 1H-NMR based urine metabolomic profiles of IUGR, LGA and AGA newborns on the first day of life. J Matern Neonatal Med. 2014;27(suppl 2):13–19.
Maitre L, Villanueva CM, Lewis MR, et al. Maternal urinary metabolic signatures of fetal growth and associated clinical and environmental factors in the INMA study. BMC Med. 2016;14(1):177.
Constantinou M, Papakonstantinou E, Benaki D, et al. Application of nuclear magnetic resonance spectroscopy combined with principal component analysis in detecting inborn errors of metabolism using blood spots: a metabonomic approach. Anal Chim Acta. 2004;511(2):303–312.
Constantinou MA, Papakonstantinou E, Spraul M, et al. 1H NMR-based metabonomics for the diagnosis of inborn errors of metabolism in urine. Anal Chim Acta. 2005;542(2):169–177.
Dénes J, Szabo E, Robinette S, et al. Metabonomics of newborn screening dried blood spot samples: a novel approach in the screening and diagnostics of inborn errors of metabolism. Anal Chem. 2012;84(22):10113–10120.
Sahoo S, Franzson L, Jonsson JJ, Thiele I. A compendium of inborn errors of metabolism mapped onto the human metabolic network. Mol Biosyst. 2012;8(10):2545–2558.
Yang Q, Lin SS, Yang JT, Tang LJ, Yu RQ. Detection of inborn errors of metabolism utilizing GC-MS urinary metabolomics coupled with a modified orthogonal partial least squares discriminant analysis. Talanta. 2017;165:545–552.
Beckonert O, Keun HC, Ebbels TMD, et al. Metabolic profiling, metabolomic and metabonomic procedures for NMR spectroscopy of urine, plasma, serum and tissue extracts. Nat Protoc. 2007;2(11):2692–2703.
Yi L, Dong N, Yun Y, et al. Chemometric methods in data processing of mass spectrometry-based metabolomics: a review. Anal Chim Acta. 2016;914:17–34.
Romero R, Mazaki-Tovi S, Vaisbuch E, et al. Metabolomics in premature labor: a novel approach to identify patients at risk for preterm delivery. J Matern Neonatal Med. 2010;23(12): 1344–1359.
Graça G, Goodfellow BJ, Barros AS, et al. UPLC-MS metabolic profiling of second trimester amniotic fluid and maternal urine and comparison with NMR spectral profiling for the identification of pregnancy disorder biomarkers. Mol Biosyst. 2012;8(4):1243–1254.
Power KM, Sanchez-Galan JE, Luskey GW, Koski KG, Burns DH. Use of near-infrared spectroscopic analysis of second trimester amniotic fluid to assess preterm births. J Pregnancy. 2011; 2011:980985.
Graça G, Moreira AS, Correia AJV, et al. Mid-infrared (MIR) metabolic fingerprinting of amniotic fluid: a possible avenue for early diagnosis of prenatal disorders? Anal Chim Acta. 2013;764:24–31.
Thorp JM, Rice MM, Harper M, et al. Advanced lipoprotein measures and recurrent preterm birth. Am J Obstet Gynecol. 2013;209(4):342.e1-342.e7.
Diaz SO, Pinto J, Graça G, et al. Metabolic biomarkers of prenatal disorders: an exploratory NMR metabonomics study of second trimester maternal urine and blood plasma. J Proteome Res. 2011;10(8):3732–3742.
Maitre L, Fthenou E, Athersuch T, et al. Urinary metabolic profiles in early pregnancy are associated with preterm birth and fetal growth restriction in the Rhea mother-child cohort study. BMC Med. 2014;12:110.
Auray-Blais C, Raiche E, Gagnon R, Berthiaume M, Pasquier JC. Metabolomics and preterm birth: what biomarkers in cervicovaginal secretions are predictive of high-risk pregnant women? Int J Mass Spectrom. 2011;307(1-3):33–38.
Ghartey J, Bastek JA, Brown A, Anglim L, Elovitz M. Women with preterm birth have a distinct cervico-vaginal fluid. Am J Obstet Gynecol. 2015;212(6):775.e1-776.e12.
Thomas MM, Sulek K, McKenzie J, et al. Metabolite profile of cervicovaginal fluid from early pregnancy is not predictive of spontaneous preterm birth. Int J Mol Sci. 2015;16(11): 27741–27748.
Amabebe E, Reynolds S, Stern VL, et al. Identifying metabolite markers for preterm birth in cervicovaginal fluid by magnetic resonance spectroscopy. Metabolomics. 2016;12(4):67.
Amabebe E, Reynolds S, Stern V, Stafford G, Paley M, Anumba DOC. Cervicovaginal fluid acetate: a metabolite marker of preterm birth in symptomatic pregnant women. Front Med. 2016;3:48.
Stafford GP, Parker JL, Amabebe E, et al. Spontaneous preterm birth is associated with differential expression of vaginal metabolites by Lactobacilli-dominated microflora. Front Physiol. 2017;8:615.
Menon R, Jones J, Gunst PR, et al. Amniotic fluid metabolomic analysis in spontaneous preterm birth. Reprod Sci. 2014;21(6): 791–803.
Baraldi E, Giordano G, Stocchero M, et al. Untargeted metabolomic analysis of amniotic fluid in the prediction of preterm delivery and bronchopulmonary dysplasia. PLoS One. 2016; 11(10):e0164211.
Tea I, Le Gall G, Ku¨ster A, et al. 1H-NMR-based metabolic profiling of maternal and umbilical cord blood indicates altered materno-foetal nutrient exchange in preterm infants. PLoS One. 2012;7(1):e29947.
Alexandre-Gouabau MC, Courant F, Moyon T, et al. Maternal and cord blood LC-HRMS metabolomics reveal alterations in energy and polyamine metabolism, and oxidative stress in verylow birth weight infants. J Proteome Res. 2013;12(6):2764–2778.
Foxall PJ, Bewley S, Neild GH, Rodeck CH, Nicholson JK. Analysis of fetal and neonatal urine using proton nuclear magnetic resonance spectroscopy. Arch Dis Child. 1995;73(3):F153–F157.
Trump S, Laudi S, Unruh N, Goelz R, Leibfritz D. 1H-NMR metabolic profiling of human neonatal urine. Magn Reson Mater Phy. 2006;19(6):305–312.
Atzori L, Antonnucci R, Barberini L, et al. 1H NMR-based metabolomic analysis from preterm and term neonates. Front Biosci. 2011;3:1005–1012.
Moltu SJ, Sachse D, Blakstad EW, et al. Urinary metabolite profiles in premature infants show early postnatal metabolic adaptation and maturation. Nutrients. 2014;6(5):1913–30.
Wilson K, Hawken S, Ducharme R, et al. Metabolomics of prematurity: analysis of patterns of amino acids, enzymes, and endocrine markers by categories of gestational age. Pediatr Res. 2014; 75(2):367–373.
Wilson K, Hawken S, Murphy MSQ, et al. Postnatal prediction of gestational age using newborn fetal hemoglobin levels. EBioMedicine. 2016;15:203–209.
Wilson K, Hawken S, Potter BK, et al. Accurate prediction of gestational age using newborn screening analyte data. Am J Obstet Gynecol. 2016;214(4):513.e1-513.e9.
Liu J, Chen JJ, Li XW, Fu W, Zhang WQ. Metabolomic research on newborns infants with intrauterine growth restriction. Medicine( Baltimore). 2016;95(17):e3564.
Jelliffe-Pawlowski LL, Norton ME, Baer RJ, Santos N, Rutherford GW. Gestational dating by metabolic profile at birth: a California cohort study. Am J Obstet Gynecol. 2016;214(4):511.e1-511.e13.
Ryckman KK, Berberich SL, Dagle JM. Predicting gestational age using neonatal metabolic markers. Am J Obstet Gynecol. 2016;214(4):515.e1–515.e13.
Marincola FC, Noto A, Caboni P, et al. A metabolomic study of preterm human and formula milk by high resolution NMR and GC/MS analysis: preliminary results. J Matern Neonatal Med. 2012;25(suppl 5):62–67.
Longini M, Tataranno ML, Proietti F, et al. A metabolomic study of preterm and term human and formula milk by proton MRS analysis: preliminary results. J Matern Fetal Neonatal Med. 2014;27(Suppl 2):27–33.
Spevacek AR, Smilowitz JT, Chin EL, Underwood MA, German JB, Slupsky CM. Infant maturity at birth reveals minor differences in the maternal milk metabolome in the first month of lactation. J Nutr. 2015;145(8):1698–1708.
Sundekilde UK, Downey E, O’Mahony JA, et al. The effect of gestational and lactational age on the human milk metabolome. Nutrient. 2016;8(5):304.
Cecatti JG, Souza RT, Sulek K, et al. Use of metabolomics for the identification and validation of clinical biomarkers for preterm birth: preterm SAMBA. BMC Pregnancy Childbirth. 2016; 16(1):212.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gil, A.M., Duarte, D. Biofluid Metabolomics in Preterm Birth Research. Reprod. Sci. 25, 967–977 (2018). https://doi.org/10.1177/1933719118756748
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719118756748