Abstract
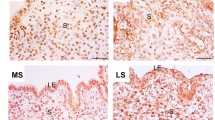
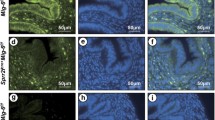
Activin A can stimulate aromatase P450 (P450arom) expression in eutopic endometrial stromal cells (ESCs) of endometriosis by activin type I receptor-Smad pathway. In order to identify Smad3/4 binding to P450arom promoter II that mediates activin A response in ESCs, polymerase chain reaction (PCR) products of a serial truncated deletion of the P450arom promoter II region between —904 and +87 bp were inserted into the pGL3-basic vector to generate the promoter reporter plasmids. Luciferase reporter plasmids were cotransfected into cells with or without activin A (25 ng/mL). The pGL3 —705/+87 revealed a luciferase activity similar to pGL3 —904/+87, whereas progressive truncation to position —464/+87 and — 192/+87, the luciferase activity was significant variation. Chromatin immunoprecipitation assay and Smad4-small interfering RNA (siRNA) testify that Smad3/4 binds to the activin A-responsive aromatase promoter in ESCs. Chromatin immunoprecipitation assay-PCR assay demonstrated anti-Smad3 antibody complexes could interact with the amplified DNA of the activin A-responsive P450arom promoter. Mutations of the binding site (—141/— 138 bp, — 165/— 162 bp) in P450arom promoter II significantly reduced promoter activity of activin A fold-induction to 26% and 28%, respectively. We cotransfected pGL3 —705/+87 with control siRNA and Smad4-siRNA into ESCs in the presence of activin A. Luciferase analysis showed that Smad4-siRNA abolished increased promoter activity of activin A-induced P450arom expression. The effect of activin A on the p-Smad3 accumulation in the cytoplasm and nucleus was significantly abrogated following the pretreatment of ESCs with Smad4-siRNA. In conclusion, activated Smad3 proteins can bind to P450arom promoter —705/+87 bp region, responsive to activin A in ESCs, which can promote P450arom transcription.
Similar content being viewed by others
References
Schreinemacher MH, Backes WH, Slenter JM, et al. Towards endometriosis diagnosis by gadofosveset-trisodium enhanced magnetic resonance imaging. PLoS One. 2012;7(3):e33241.
Barrier BF. Immunology of endometriosis. Clin Obstet Gynecol. 2010;53(2):397–402.
Ferrero S, Remorgida V, Maganza C, et al. Aromatase and endometriosis: estrogens play a role. Ann N Y Acad Sci. 2014;1317: 17–23.
Huhtinen K, Stahle M, Perheentupa A, Poutanen M. Estrogen biosynthesis and signaling in endometriosis. Mol Cell Endocrinol. 2012;358(2):146–154.
Marchais-Oberwinkler S, Wetzel M, Ziegler E, et al. New druglike hydroxyphenylnaphthol steroidomimetics as potent and selective 17beta-hydroxysteroid dehydrogenase type 1 inhibitors for the treatment of estrogen-dependent diseases. J Med Chem. 2011;54(2):534–547.
Rai P, Shivaji S. The role of DJ-1 in the pathogenesis of endometriosis. PLoS One. 2011;6(3):e18074.
Fedele L, Bianchi S, Zanconato G, Tozzi L, Raffaelli R. Gonadotropin-releasing hormone agonist treatment for endometriosis of the rectovaginal septum. Am J Obstet Gynecol. 2000;183(6):1462–1467.
Wolfler MM, Nagele F, Kolbus A, et al. A predictive model for endometriosis. Hum Reprod. 2005;20(6):1702–1708.
Noble LS, Simpson ER, Johns A, Bulun SE. Aromatase expression in endometriosis. J Clin Endocrinol Metab. 1996;81(1):174–179.
Velasco I, Rueda J, Acien P. Aromatase expression in endometriotic tissues and cell cultures of patients with endometriosis. Mol Hum Reprod. 2006;12(6):377–381.
Huhtinen K, Desai R, Stahle M, et al. Endometrial and endometriotic concentrations of estrone and estradiol are determined by local metabolism rather than circulating levels. J Clin Endocrinol Metab. 2012;97(11):4228–4235.
Kaneda H, Arao T, Matsumoto K, et al. Activin A inhibits vascular endothelial cell growth and suppresses tumour angiogenesis in gastric cancer. Br J Cancer. 2011;105(8):1210–1217.
Yoshino O, Izumi G, Shi J, et al. Activin-A is induced by interleukin-1beta and tumor necrosis factor-alpha and enhances the mRNA expression of interleukin-6 and protease-activated receptor-2 and proliferation of stromal cells from endometrioma. Fertil Steril. 2011;96(1):118–121.
Hirata T, Osuga Y, Takamura M, et al. Interleukin-17F increases the secretion of interleukin-8 and the expression of cyclooxygenase 2 in endometriosis. Fertil Steril. 2011;96(1):113–117.
Yoshino O, Hori M, Osuga Y, et al. Myomectomy reduces endometrial T2 relaxation times. Fertil Steril. 2011;95(8):2781–2783.
Wang Y, Libasci V, Bernard DJ. Activin A induction of FSHbeta subunit transcription requires SMAD4 in immortalized gonadotropes. J Mol Endocrinol. 2010;44(6):349–362.
Chen L, Zhang W, Liang HF, et al. Activin A induces growth arrest through a SMAD-dependent pathway in hepatic progenitor cells. Cell Commun Signal. 2014;12:18.
Woodruff TK, Mather JP. Inhibin, activin and the female reproductive axis. Annu Rev Physiol. 1995;57:219–44.
Risbridger GP, Schmitt JF, Robertson DM. Activins and inhibins in endocrine and other tumors. Endocr Rev. 2001;22(6):836–858.
Attisano L, Lee-Hoeflich ST. The Smads. Genome Biol. 2001; 2(8):REVIEWS3010.
Shi Y, Hata A, Lo RS, Massague J, Pavletich NP. A structural basis for mutational inactivation of the tumour suppressor Smad4. Nature. 1997;388(6637):87–93.
Zheng J, Qu J, Lu P, et al. Activin A stimulates aromatase via the ALK4-Smad pathway in endometriosis. Biomed Res Int. 2016; 2016:5791510.
Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67(5):817–821.
Hou Z, Zhou J, Ma X, Fan L, Liao L, Liu J. Role of interleukin-1 receptor type II in the pathogenesis of endometriosis. Fertil Steril. 2008;89(1):42–51.
Noble LS, Takayama K, Zeitoun KM, et al. Prostaglandin E2 stimulates aromatase expression in endometriosis-derived stromal cells. J Clin Endocrinol Metabol. 1997;82(2):600–606.
Zeitoun K, Takayama K, Michael MD, Bulun SE. Stimulation of aromatase P450 promoter (II) activity in endometriosis and its inhibition in endometrium are regulated by competitive binding of steroidogenic factor-1 and chicken ovalbumin upstream promoter transcription factor to the same cis-acting element. Mol Endocrinol. 1999;13(2):239–253.
Simpson ER, Mahendroo MS, Means GD, et al. Aromatase cytochrome P450, the enzyme responsible for estrogen biosynthesis. Endocr Rev. 1994;15(3):342–355.
Chen SA, Besman MJ, Sparkes RS, et al. Human aromatase: cDNA cloning, Southern blot analysis, and assignment of the gene to chromosome 15. DNA. 1988;7(1):27–38.
Bulun SE, Sebastian S, Takayama K, Suzuki T, Sasano H, Shozu M. The human CYP19 (aromatase P450) gene: update on physiologic roles and genomic organization of promoters. J Steroid Biochem Mol Biol. 2003;86(3-5):219–224.
Harada N, Utsumi T, Takagi Y. Tissue-specific expression of the human aromatase cytochrome P-450 gene by alternative use of multiple exons 1 and promoters, and switching of tissue-specific exons 1 in carcinogenesis. Proc Natl Acad Sci U S A. 1993; 90(23):11312–11316.
Harada N. A unique aromatase (P-450AROM) mRNA formed by alternative use of tissue-specific exons 1 in human skin fibroblasts. Biochem Biophys Res Commun. 1992;189(2):1001–1007.
Sebastian S, Bulun SE. A highly complex organization of the regulatory region of the human CYP19 (aromatase) gene revealed by the Human Genome Project. J Clin Endocrinol Metab. 2001;86(10):4600–4602.
Clyne CD, Speed CJ, Zhou J, Simpson ER. Liver receptor homologue-1 (LRH-1) regulates expression of aromatase in pre-adipocytes. J Biol Chem. 2002;277(23):20591–20597.
Mahendroo MS, Mendelson CR, Simpson ER. Tissue-specific and hormonally controlled alternative promoters regulate aromatase cytochrome P450 gene expression in human adipose tissue. J Biol Chem. 1993;268(26):19463–19470.
Zhou D, Chen S. Identification and characterization of a cAMP-responsive element in the region upstream from promoter 1.3 of the human aromatase gene. Arch Biochem Biophys. 1999;371(2):179–190.
Qiu Y, Krishnan V, Pereira FA, Tsai SY, Tsai MJ. Chicken ovalbumin upstream promoter-transcription factors and their regulation. J Steroid Biochem Mol Biol. 1996;56(1-6 Spec No):81–85.
Ritchie HH, Wang LH, Tsai S, O’Malley BW, Tsai MJ. COUP-TF gene: a structure unique for the steroid/thyroid receptor super-family. Nucleic Acids Res. 1990;18(23):6857–6862.
Wang LH, Tsai SY, Cook RG, Beattie WG, Tsai MJ, O’Malley BW. COUP transcription factor is a member of the steroid receptor superfamily. Nature. 1989;340(6229):163–166.
Ladias JA, Karathanasis SK. Regulation of the apolipoprotein AI gene by ARP-1, a novel member of the steroid receptor super-family. Science. 1991;251(4993):561–565.
Miyajima N, Kadowaki Y, Fukushige S, et al. Identification of two novel members of erbA superfamily by molecular cloning: the gene products of the two are highly related to each other. Nucleic Acids Res. 1988;16(23):11057–11074.
Zawel L, Dai JL, Buckhaults P, et al. Human Smad3 and Smad4 are sequence-specific transcription activators. Mol Cell. 1998;1(4):611–617.
Chen X, Weisberg E, Fridmacher V, Watanabe M, Naco G, Whitman M. Smad4 and FAST-1 in the assembly of activin-responsive factor. Nature. 1997;389(6646):85–89.
Labbe E, Silvestri C, Hoodless PA, Wrana JL, Attisano L. Smad2 and Smad3 positively and negatively regulate TGF beta-dependent transcription through the forkhead DNA-binding protein FAST2. Mol Cell. 1998;2(1):109–120.
Itoh S, Ericsson J, Nishikawa J, Heldin CH, ten Dijke P. The transcriptional co-activator P/CAF potentiates TGF-beta/Smad signaling. Nucleic Acids Res. 2000;28(21):4291–4298.
Datto MB, Hu PP, Kowalik TF, Yingling J, Wang XF. The viral oncoprotein E1A blocks transforming growth factor beta-mediated induction of p21/WAF1/Cip1 and p15/INK4B. Mol Cell Biol. 1997;17(4):2030–2037.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qu, J., Zhu, Y., Wu, X. et al. Smad3/4 Binding to Promoter II of P450arom So As to Regulate Aromatase Expression in Endometriosis. Reprod. Sci. 24, 1187–1194 (2017). https://doi.org/10.1177/1933719116681517
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719116681517