Abstract
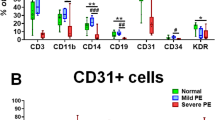
Preeclampsia (PE) is a pregnancy-specific disease characterized by the new onset of hypertension and proteinuria. Mothers with PE are known to develop endothelial dysfunction, but its effect on infants has been understudied, as newborns are often asymptomatic. Recent studies indicate that infants born from preeclamptic pregnancies develop endothelial dysfunction including higher blood pressure during childhood and an increased risk of stroke later in life. We hypothesize that PE reduces the number and function of fetal angiogenic progenitor cells and may contribute to this increased risk. We quantified 2 distinct types of angiogenic progenitors, pro-angiogenic circulating progenitor cells (CPCs) and endothelial colony-forming cells (ECFCs), from the umbilical cord blood of preeclamptic pregnancies and normotensive controls. Pro-angiogenic and non-angiogenic CPCs were enumerated via flow cytometry and ECFCs by cell culture. Additionally, we studied the growth, migration, and tube formation of ECFCs from PE and gestational age–matched normotensive control pregnancies. We found that PE resulted in decreased cord blood pro-angiogenic CPCs and ECFCs. Nonangiogenic CPCs were also decreased. Pre-eclamptic ECFCs demonstrated decreased growth and migration but formed tube-like structures in vitro similar to controls. Our results suggest that the preeclamptic environment alters the number and function of angiogenic progenitor cells and may increase the risk of later vascular disease.
Similar content being viewed by others
References
Wu CS, Nohr EA, Bech BH, Vestergaard M, Catov JM, Olsen J. Health of children born to mothers who had preeclampsia: a population-based cohort study. Am J Obstet Gynecol. 2009; 201(3):269.e1–269.e10.
Ghulmiyyah L, Sibai B. Maternal mortality from preeclampsia/ eclampsia. Semin Perinatol. 2012;36(1):56–59.
Kajantie E, Eriksson JG, Osmond C, Thornburg K, Barker DJ. Pre-eclampsia is associated with increased risk of stroke in the adult offspring: the Helsinki birth cohort study. Stroke. 2009;40(4):1176–1180.
Seidman DS, Laor A, Gale R, Stevenson DK, Mashiach S, Danon YL. Pre-eclampsia and offspring’s blood pressure, cognitive ability and physical development at 17-years-of-age. Br J Obstet Gynaecol. 1991;98(10):1009–1014.
Tenhola S, Rahiala E, Halonen P, Vanninen E, Voutilainen R. Maternal preeclampsia predicts elevated blood pressure in 12-year-old children: evaluation by ambulatory blood pressure monitoring. Pediatr Res. 2006;59(2):320–324.
Vatten LJ, Romundstad PR, Holmen TL, Hsieh C, Trichopoulos D, Stuver SO. Intrauterine exposure to preeclampsia and adoles-cent blood pressure, body size, and age at menarche in female offspring. Obstet Gynecol. 2003;101(3):529–533.
Giannotti G, Doerries C, Mocharla PS, et al. Impaired endothelial repair capacity of early endothelial progenitor cells in prehypertension: relation to endothelial dysfunction. Hypertension. 2010;55(6):1389–1397.
Fadini GP, Miorin M, Facco M, et al. Circulating endothelial progenitor cells are reduced in peripheral vascular complications of type 2 diabetes mellitus. J Am Coll Cardiol. 2005;45(9):1449–1457.
Burnham EL, Taylor WR, Quyyumi AA, Rojas M, Brigham KL, Moss M. Increased circulating endothelial progenitor cells are associated with survival in acute lung injury. Am J Respir Crit Care Med. 2005;172(7):854–860.
Rafat N, Hanusch C, Brinkkoetter PT, et al. Increased circulating endothelial progenitor cells in septic patients: correlation with survival. Crit Care Med. 2007;35(7):1677–1684.
Vasa M, Fichtischerer S, Aicher A, et al. Number and migratory activity of circulating endothelial progenitor cells inversely cor-relate with risk factors for coronary artery disease. Circ Res. 2001; 89(1):E1–E7.
Ghani U, Shuaib A, Salam A, et al. Endothelial progenitor cells during cerebrovascular disease. Stroke. 2005;36(1):151–153.
Marti-Fàbregas J, Delgado-Mederos R, Crespo J, et al. Circulat-ing endothelial progenitor cells and the risk of vascular events after ischemic stroke. PLoS One. 2015;10(4):e0124895.
Boos CJ, Goon PK, Lip GY. Endothelial progenitor cells in the vascular pathophysiology of hypertension: arterial stiffness, age-ing and more. J Hum Hypertens. 2006;20(7):475–477.
Monga R, Buck S, Sharma P, Thomas R, Chouthai NS. Effect of preeclampsia and intrauterine growth restriction on endothelial progenitor cells in human umbilical cord blood. J Matern Fetal Neonatal Med. 2012;25(11):2385–2389.
Luppi P, Powers RW, Verma V, Edmunds L, Plymire D, Hubel CA. Maternal circulating CD34+VEGFR-2+ and CD133+ VEGFR-2+ progenitor cells increase during normal pregnancy but are reduced in women with preeclampsia. Reprod Sci. 2010;17(7):643–652.
Liu X, Luo Q, Zheng Y, et al. Notchl impairs endothelial pro-genitor cell bioactivity in preeclampsia [published online May 18, 2016]. Reprod Sci. 2016.
Case J, Mead LE, Bessler WK, et al. Human CD34+ AC133+VEGFR-2+ cells are not endothelial progenitor cells but distinct, primitive hematopoietic progenitors. Exp Hematol. 2007;35(7):1109–1118.
Yoder MC, Mead LE, Prater D, et al. Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/ progenitor cell principals. Blood. 2007;109(5):1801–1809.
Ingram DA, Caplice NM, Yoder MC. Unresolved questions, changing definitions, and novel paradigms for defining endothe-lial progenitor cells. Blood. 2005;106(5):1525–1531.
Duong HT, Erzurum SC, Asosingh K. Pro-angiogenic hemato-poietic progenitor cells and endothelial colony-forming cells in pathological angiogenesis of bronchial and pulmonary circulation. Angiogenesis. 2011;14(4):411–422.
Richardson MR, Yoder MC. Endothelial progenitor cells: quo vadis? J Mol Cell Cardiol. 2011;50(2):266–272.
Baker CD, Balasubramaniam V, Mourani PM, et al. Cord blood angiogenic progenitor cells are decreased in bronchopulmonary dysplasia. Eur Respir J. 2012;40(6):1516–1522.
Estes ML, Mund JA, Ingram DA, Case J. Identification of endothelial cells and progenitor cell subsets in human peripheral blood. Curr Protoc Cytom. 2010; Chapter 9:Unit 9.33.1–11.
Estes ML, Mund JA, Mead LE, et al. Application of polychro-matic flow cytometry to identify novel subsets of circulating cells with angiogenic potential. Cytometry A. 2010;77(9):831–839.
Acosta JC, Haas DM, Saha CK, Dimeglio LA, Ingram DA, Haneline LS. Gestational diabetes mellitus alters maternal and neonatal circulating endothelial progenitor cell subsets. Am J Obstet Gynecol. 2011;204(3):254.e8–254.e15.
Ingram DA, Mead LE, Tanaka H, et al. Identification of a novel hierarchy of endothelial progenitor cells using human peripheral and umbilical cord blood. Blood. 2004;104(9):2752–2760.
Borghesi A, Massa M, Campanelli R, et al. Circulating endothelial progenitor cells in preterm infants with bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2009;180(6):540–546.
Munoz-Hernandez R, Miranda ML, Stiefel P, et al. Decreased level of cord blood circulating endothelial colony-forming cells in preeclampsia. Hypertension. 2014;64(1):165–171.
von Versen-Höynck F, Brodowski L, Dechend R, Myerski AC, Hubel CA. Vitamin D antagonizes negative effects of preeclamp-sia on fetal endothelial colony forming cell number and function. PLoS One. 2014;9(6):e98990.
Tita ATN, Andrews WW. Diagnosis and management of clinical chorioamnionitis. Clin Perinatol. 2010;37(2):339–354.
ACOG Committee on Practice Bulletins—Obstetrics. ACOG practice bulletin. Diagnosis and management of preeclampsia and eclampsia. Number 33, January 2002. Obstet Gynecol. 2002;99(1):159–167.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381.
Bikfalvi A, Cramer EM, Tenza D, Tobelem G. Phenotypic mod-ulations of human umbilical vein endothelial cells and human dermal fibroblasts using two angiogenic assays. Biol Cell. 1991;72(3):275–278.
Ingram DA, Lien IZ, Mead LE, et al. In vitro hyperglycemia or a diabetic intrauterine environment reduces neonatal endothelial colony-forming cell numbers and function. Diabetes. 2008;57(3):724–731.
Sipos PI, Bourque SL, Hubel CA, et al. Endothelial colony-forming cells derived from pregnancies complicated by intrauterine growth restriction are fewer and have reduced vasculogenic capacity. J Clin Endocrinol Metab. 2013;98(12):4953–4960.
Blue EK, DiGiuseppe R, Derr-Yellin E, et al. Gestational diabetes induces alterations in the function of neonatal endothelial colony-forming cells. Pediatr Res. 2014;75(2):266–272.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gumina, D.L., Black, C.P., Balasubramaniam, V. et al. Umbilical Cord Blood Circulating Progenitor Cells and Endothelial Colony-Forming Cells Are Decreased in Preeclampsia. Reprod. Sci. 24, 1088–1096 (2017). https://doi.org/10.1177/1933719116678692
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719116678692