Abstract
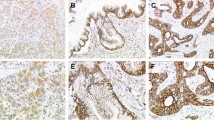
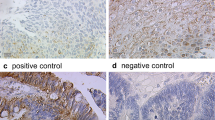
Rationale: Development of cervical squamous carcinoma (CXCA) is accompanied by changes in estrogen receptors (ERs, ERα and ERβ) and ezrin expression; however, reports have been conflicting. Using histologically documented staging of cervical biopsies, we determined ezrin and ER relationships during CXCA development. Methods: Immunoreactive (ir) ezrin, ir-ERα, and ir-ERβ were studied in normal epithelium, carcinoma in situ/cervical intraepithelial neoplasia (CIN) 1 to 3, and local invasion or metastatic CXCA. Results were compared using H scoring. Cultures of Caski metastatic CXCA cells were treated with estradiol and/or tamoxifen and studied for ER-driven ir-ezrin and the morphologic response. Results: Koilocytosis was present and indicated viral presence. The ezrin H score increased from CIN1 to CIN3, reaching significant differences from normal by CIN3 (P = .004) and 2× normal in metastatic CXCA. Estrogen receptor α and ERβ H scores fell, reaching significance by CIN3 (ERα, P = .0001; ERβ, P = .024). During estradiol treatment, ezrin in Caski cells increased and localized to the periphery, in ruffles and processes. The selective ER modulator tamoxifen blocked the estradiol-induced changes. Conclusions: During cervical carcinogenesis, the usual relationship between estrogen and ezrin induction is abridged. This is consistent with the effects of human papilloma virus viral proteins such as E6 and E7 that upregulate SIXI, a protein that induces ezrin. Cervical carcinogenesis is progressive but arrests at the preinvasive stage for varying lengths of time. These studies suggest that changes in ezrin may be associated with the development of the invasive phenotype and penetration of the basement membrane. They also raise the possibility that inhibiting ezrin expression could be a target for the prevention of invasive CXCA.
Similar content being viewed by others
References
American Cancer Society. Cancer Facts & Figures 2015. Atlanta, GA: American Cancer Society; 2015.
Dunleavey R. Incidence, pathophysiology and treatment of cervical cancer. Nurs Times. 2004;100(44):38–41.
Mirkovic J, Howitt BE, Roncarati P, et al. Carcinogenic HPV infection in the cervical squamo-columnar junction. J Pathol. 2015;236(3):265–271.
Fadiel A, Lee HH, Demir N, et al. Ezrin is a key element in the human vagina. Maturitas. 2008;60(1):31–41.
Fehon RG, McClatchey AI, Bretscher A. Organizing the cell cortex: the role of ERM proteins. Nat Rev Mol Cell Biol. 2010;11(4):276–287.
Ohtani K, Sakamoto H, Rutherford T, Chen Z, Satoh K, Naftolin F. Ezrin, a membrane-cytoskeletal linking protein, is involved in the process of invasion of endometrial cancer cells. Cancer Lett. 1999;147(1-2):318.
Fadiel A, Chen Z, Ulukus E, Ohtani K, Hatami M, Naftolin F. Ezrin overexpression by transformed human ovarian surface epithelial cells, ovarian cleft cells, and serous ovarian adenocarcinoma cells. Reprod Sci. 2012;19(8):797–805.
Martin TA, Harrison G, Mansel RE, Jiang WG. The role of the CD44/ezrin complex in cancer metastasis. Crit Rev Oncol Hematol. 2003;46(2):165–186.
Majores M, Schick V, Engels G, et al. Mutational and immunohistochemical analysis of ezrin-, radixin-, moesin (ERM) molecules in epilepsy-associated glioneuronal lesions. Acta Neuropathol. 2005;110(6):537–546.
Clucas J, Valderrama F. ERM proteins in cancer progression. J Cell Sci. 2015;128(6):1253.
Chen Z, Fadiel A, Feng Y, Ohtani K, Rutherford T, Naftolin F. Ovarian epithelial carcinoma tyrosine phosphorylation, cell proliferation, and ezrin translocation are stimulated by interleukin 1alpha and epidermal growth factor. Cancer. 2001;92(12):3068–3075.
Chung SH. Targeting female hormone receptors as cervical cancer therapy. Trends Endocrinol Metab. 2015;26(8):399–401.
Munger K. Are selective estrogen receptor modulators (SERMs) a therapeutic option for HPV-associated cervical lesions and cancers? Am J Pathol. 2014;184(2):358–361.
Spurgeon ME, Chung SH, Lambert PF. Recurrence of cervical cancer in mice after selective estrogen receptor modulator therapy. Am J Pathol. 2014;184(2):530–540.
Song J, Fadiel A, Edusa V, et al. Estradiol-induced ezrin overexpression in ovarian cancer: a new signaling domain for estrogen. Cancer Lett. 2005;220(1):57–65.
Nayar R, Wilbur DC. The pap test and Bethesda 2014. “The reports of my demise have been greatly exaggerated.” (After a quotation from Mark Twain). Acta Cytol. 2015;59(2):121–132.
Ohtani K, Sakamoto H, Rutherford T, et al. Ezrin, a membrane-cytoskeletal linking protein, is highly expressed in atypical endometrial hyperplasia and uterine endometrioid adenocarcinoma. Cancer Lett. 2002;179(1):79–86.
Chung SH, Wiedmeyer K, Shai A, Korach KS, Lambert PF. Requirement for estrogen receptor alpha in a mouse model for human papillomavirus-associated cervical cancer. Cancer Res. 2008;68(23):9928–9934.
Yu Y, Davicioni E, Triche TJ, Merlino G. The homeoprotein SIX1 transcriptionally activates multiple protumorigenic genes but requires ezrin to promote metastasis. Cancer Res. 2006;66(4):1982–1989.
López-Romero R, Garrido-Guerrero E, Rangel-López A, et al. The cervical malignant cells display a down regulation of ER-a but retain the ER-β expression. Int J Clin Exp Pathol. 2013;6(8):1594–602.
Caiazza F, Ryan EJ, Doherty G, Winter DC, Sheahan K. Estrogen receptors and their implications in colorectal carcinogenesis. Front Oncol. 2015;5:19.
Dey P, Barros RP, Warner M, Ström A, Gustafsson JÅ. Insight into the mechanisms of action of estrogen receptor β in the breast, prostate, colon, and CNS. J Mol Endocrinol. 2013;51(3):T61–T74.
Fogarty EA, Matulis CK, Kraus WL. Activation of estrogen receptor a by raloxifene through an activating protein-1-dependent tethering mechanism in human cervical epithelial cancer cells: a role for c-Jun N-terminal kinase. Mol Cell Endocrinol. 2012;348(1):331–338.
Chung SH, Lambert PF. Prevention and treatment of cervical cancer in mice using estrogen receptor antagonists. Proc Natl Acad Sci U S A. 2009;106(46):19467–19472.
Saito Y, Sakamoto H, MacLusky NJ, Naftolin F. Gap junctions and myometrial steroid hormone receptors in pregnant and postpartum rats: a possible cellular basis for the progesterone withdrawal hypothesis. Am J Obstet Gynecol. 1985;151(6):805–812.
Lowery AJ, Miller N, Dwyer RM, Kerin MJ. Dysregulated miR-183 inhibits migration in breast cancer cells. BMC Cancer. 2010;10:502.
Mathe A, Scott RJ, Avery-Kiejda KA. miRNAs and other epigenetic changes as biomarkers in triple negative breast cancer. Int J Mol Sci. 2015;16(12):28347–28376.
Murad N, Kokkinaki M, Gunawardena N, et al. miR-184 regulates ezrin, LAMP-1 expression, affects phagocytosis in human retinal pigment epithelium and is downregulated in age-related macular degeneration. FEBS J. 2014;281(23):5251–5264.
Zhu X, Leav I, Leung YK, et al. Dynamic regulation of estrogen receptor-beta expression by DNA methylation during prostate cancer development and metastasis. Am J Pathol. 2004;164(6):2003–2012.
Auvinen E, Carpen O, Korpela T, Ronty M, Vaheri A, Tarkkanen J. Altered expression of ezrin, E-cadherin and β-catenin in cervical neoplasia. Neoplasma. 2013;60(1):56–61.
Kong J, Li Y, Liu S, et al. High expression of ezrin predicts poor prognosis in uterine cervical cancer. BMC Cancer. 2013;13:520.
Shen ZY, Xu LY, Chen MH, et al. Upregulated expression of Ezrin and invasive phenotype in malignantly transformed esophageal epithelial cells. World J Gastroenterol. 2003;9(6):1182–1186.
Díaz L, Ceja-Ochoa I, Restrepo-Angulo I, et al. Estrogens and human papilloma virus oncogenes regulate human ether-a-go-go-1 potassium channel expression. Cancer Res. 2009;69(8):3300–3307.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fadiel, A., Choi, S.D., Park, B. et al. Expression of Ezrin and Estrogen Receptors During Cervical Carcinogenesis. Reprod. Sci. 24, 706–712 (2017). https://doi.org/10.1177/1933719116667222
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719116667222