Abstract
Objectives
To investigate whether maternal plasma glycogen phosphorylase BB (GPBB) levels were altered in early pregnancy and/or at the time of diagnosis of disease in preeclampsia (term and preterm <37 weeks’ gestation) or small for gestational age (SGA).
Methods
We conducted 6 nested case–control studies within the Screening of Pregnancy Endpoint (SCOPE) Ireland cohort. Blood samples from women with preeclampsia or SGA were analyzed both from the time of disease presentation and at 15 and 20 weeks’ gestation. These were compared with control samples obtained from SCOPE women with healthy uncomplicated pregnancies matched for age, ethnicity, parity, body mass index, and gestational age. Glycogen phosphorylase BB levels were measured using the Diacordon GPBB enzyme-linked immunosorbent assay (Diagenics, Germany).
Results
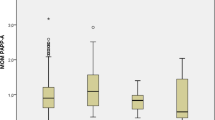
Glycogen phosphorylase BB levels were higher in women with preeclampsia compared with controls at the time of disease (term preeclampsia median [interquartile range (IQR)]: 22.2 [15.1-39.8] ng/mL vs 16.9 [10.4-19.1] ng/mL; P = .04; N = 14 and preterm preeclampsia median [IQR]: 23.1 [11.2-30.9] ng/mL vs 17.2 [9.8-19.1] ng/mL; P = .04; N = 11) and at 20 weeks’ gestation (median [IQR]: 23.0 [15.6-31.4] ng/mL vs 17.0 [13.4-23.6] ng/mL; N = 39; P = .04). Glycogen phosphorylase BB levels were also significantly higher in women with SGA compared with normal controls at the time of disease detection (median [IQR]: 22.7 [12.6-25.5] ng/mL vs 17.0 [9.8-18.0] ng/mL; N = 23; P = .03) but significantly less than controls at 15 weeks’ gestation prior to disease detection (median [IQR]: 16.0 [12.1-23.2] ng/mL vs 22.2 [17.0-28.9] ng/mL; N = 25; P = .02).
Conclusion
Glycogen phosphorylase BB alone has modest predictive abilities for the development of preeclampsia or SGA. Further research may examine its use in combination with other markers.
Similar content being viewed by others
References
Apple FS, Wu AH, Mair J, et al. Future biomarkers for detection of ischemia and risk stratification in acute coronary syndrome. Clin Chem. 2005;51(5):810–824.
Kato K, Shimizu A, Kurobe N, Takashi M, Koshikawa T. Human brain-type glycogen phosphorylase: quantitative localization in human tissues determined with an immunoassay system. J Neurochem. 1989;52(5):1425–1432.
Krause EG, Rabitzsch G, Noll F, Mair J, Puschendorf B. Glycogen phosphorylase isoenzyme BB in diagnosis of myocardial ischaemic injury and infarction. Mol Cell Biochem. 1996; 160–161:289–295.
Newgard CB, Hwang PK, Fletterick RJ. The family of glycogen phosphorylases: structure and function. Crit Rev Biochem Mol Biol. 1989;24(1):69–99.
Peetz D, Post F, Schinzel H, et al. Glycogen phosphorylase BB in acute coronary syndromes. Clin Chem Lab Med. 2005;43(12): 1351–1358.
Lippi G, Mattiuzzi C, Comelli I, Cervellin G. Glycogen phosphorylase isoenzyme BB in the diagnosis of acute myocardial infarction: a meta-analysis. Biochem Med (Zagreb). 2013; 23(1):78–82.
Rabitzsch G, Mair J, Lechleitner P, et al. Isoenzyme BB of glycogen phosphorylase b and myocardial infarction. Lancet. 1993; 341(8851):1032–1033.
Cubranic Z, Madzar Z, Matijevic S, et al. Diagnostic accuracy of heart fatty acid binding protein (H-FABP) and glycogen phosphorylase isoenzyme BB (GPBB) in diagnosis of acute myocardial infarction in patients with acute coronary syndrome. Biochem Med (Zagreb). 2012;22(2):225–236.
Robson SC, Hunter S, Boys RJ, Dunlop W. Serial study of factors influencing changes in cardiac output during human pregnancy. Am J Physiol. 1989;256(4 pt 2):h1060–h1065.
Gilbert JS, Ryan MJ, LaMarca BB, et al. Pathophysiology of hypertension during preeclampsia: linking placental ischemia with endothelial dysfunction. Am J Physiol Heart Circ Physiol. 2008;294(2):h541–h550.
Kanasaki K, Kalluri R. The biology of preeclampsia. Kidney Int. 2009;76(8):831–837.
LaMarca B, Wallace K, Herse F, et al. Hypertension in response to placental ischemia during pregnancy: role of B lymphocytes. Hypertension. 2011;57(4):865–871.
Gagnon R. Placental insufficiency and its consequences. Eur J Obstet Gynecol Reprod Biol. 2003;110(suppl 1):s99–s107.
Burton GJ, Jauniaux E, Watson AL. Maternal arterial connections to the placental intervillous space during the first trimester of human pregnancy: the Boyd collection revisited. Am J Obstet Gynecol. 1999;181(3):718–724.
Genbacev O, Zhou Y, Ludlow JW, Fisher SJ. Regulation of human placental development by oxygen tension. Science. 1997;277(5332):1669–1672.
Huppertz B, Peeters LL. Vascular biology in implantation and placentation. Angiogenesis. 2005;8(2):157–167.
Sibai B, Dekker G, Kupferminc M. Pre-eclampsia. Lancet. 2005; 365(9461):785–799.
Hutcheon JA, Lisonkova S, Joseph KS. Epidemiology of preeclampsia and the other hypertensive disorders of pregnancy. Best Pract Res Clin Obstet Gynaecol. 2011;25(4):391–403.
Kenny L, Baker PN. Maternal pathophysiology in pre-eclampsia. Baillieres Best Pract Res Clin Obstet Gynaecol. 1999;13(1):59–75.
Robb AO, Mills NL, Din JN, et al. Influence of the menstrual cycle, pregnancy, and preeclampsia on arterial stiffness. Hypertension. 2009;53(6):952–958.
Craici I, Wagner S, Garovic VD. Preeclampsia and future cardiovascular risk: formal risk factor or failed stress test? Ther Adv Cardiovasc Dis. 2008;2(4):249–259.
Vikse BE, Irgens LM, Leivestad T, Skjaerven R, Iversen BM. Preeclampsia and the risk of end-stage renal disease. N Engl J Med. 2008;359(8):800–809.
Bellamy L, Casas JP, Hingorani AD, Williams DJ. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: systematic review and meta-analysis. BMJ. 2007;335(7627):974.
Kenny LC, Black MA, Poston L, et al. Early pregnancy prediction of preeclampsia in nulliparous women, combining clinical risk and biomarkers: the Screening for Pregnancy Endpoints (SCOPE) International Cohort Study. Hypertension. 2014; 64(3):644–652.
Lee J, Romero R, Dong Z, et al. Glycogen phosphorylase isoenzyme BB plasma concentration is elevated in pregnancy and preterm preeclampsia. Hypertension. 2012;59(2):274–282.
North RA, McCowan LM, Dekker GA, et al. Clinical risk prediction for pre-eclampsia in nulliparous women: development of model in international prospective cohort. BMJ. 2011;342:d1875.
McCarthy FP, O’Keeffe LM, Khashan AS, et al. Association between maternal alcohol consumption in early pregnancy and pregnancy outcomes. Obstet Gynecol. 2013;122(4):830–837.
McCarthy FP, Khashan AS, North RA, et al. Pregnancy loss managed by cervical dilatation and curettage increases the risk of spontaneous preterm birth. Hum Reprod. 2013;28(12):3197–3206.
McCowan L, North R, Taylor R. Screening nulliparous women to identify the combinations of clinical risk factors and/or biomarkers required to predict preeclampsia, small for gestational age babies and spontaneous preterm birth. ACTRN12607000551493. Australian New Zealand Clinical Trials Registry: The University of Sydney. 2007. Web site. http://www.anzctr.org.au/trial_view.aspx?ID=82254. Accessed November 4, 2015.
McCowan LM, Dekker GA, Chan E, et al. Spontaneous preterm birth and small for gestational age infants in women who stop smoking early in pregnancy: prospective cohort study. BMJ. 2009;338:b1081.
Brown MA, Hague WM, Higgins J, et al. The detection, investigation and management of hypertension in pregnancy: executive summary. Aust N Z J Obstet Gynaecol. 2000;40(2):133–138.
Gardosi J, Francis A. Customised Centile Calculator-GROW 6.12 Bulk. Gestation Network; 2007. Web site. http://www.gestation.net/birthweight_centiles/birthweight_centiles.htm.
McCowan LM, Thompson JM, Taylor RS, et al. Clinical prediction in early pregnancy of infants small for gestational age by customised birthweight centiles: findings from a healthy nulliparous cohort. PloS One. 2013;8(8):e70917.
Evans DJ, Murray R, Kissebah AH. Relationship between skeletal muscle insulin resistance, insulin-mediated glucose disposal, and insulin binding. Effects of obesity and body fat topography. J Clin Invest. 1984;74(4):1515–1525.
Laviola L, Perrini S, Belsanti G, et al. Intrauterine growth restriction in humans is associated with abnormalities in placental insulin-like growth factor signaling. Endocrinology. 2005; 146(3):1498–1505.
Fu Q, Yu X, Callaway CW, Lane RH, McKnight RA. Epigenetics: intrauterine growth retardation (IUGR) modifies the his-tone code along the rat hepatic IGF-1 gene. FASEB J. 2009; 23(8):2438–2449.
Hiden U, Glitzner E, Hartmann M, Desoye G. Insulin and the IGF system in the human placenta of normal and diabetic pregnancies. J Anat. 2009;215(1):60–68.
Thorn SR, Regnault TR, Brown LD, et al. Intrauterine growth restriction increases fetal hepatic gluconeogenic capacity and reduces messenger ribonucleic acid translation initiation and nutrient sensing in fetal liver and skeletal muscle. Endocrinology. 2009;150(7):3021–3030.
Limesand SW, Rozance PJ, Smith D, Hay WW Jr. Increased insulin sensitivity and maintenance of glucose utilization rates in fetal sheep with placental insufficiency and intrauterine growth restriction. Am J Physiol Endocrinol Metab. 2007;293(6):e1716–e1725.
Lin YS, Tang CH, Yang CY, et al. Effect of pre-eclampsia-eclampsia on major cardiovascular events among peripartum women in Taiwan. Am J Cardiol. 2011;107(2):325–330.
Chappell LC, Duckworth S, Seed PT, et al. Diagnostic accuracy of placental growth factor in women with suspected preeclampsia: a prospective multicenter study. Circulation. 2013;128(19):2121–2131.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McCarthy, F.P., Doyle, A., Khashan, A.S. et al. Altered Maternal Plasma Glycogen Phosphorylase Isoenzyme BB as a Biomarker for Preeclampsia and Small for Gestational Age. Reprod. Sci. 23, 738–747 (2016). https://doi.org/10.1177/1933719115616495
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719115616495