Abstract
Introduction
Altered angiogenesis has been implicated in the pathogenesis of various pregnancy complications, particularly preeclampsia. At present, there is a lack of data on the possible role of angiogenesis and its molecular mechanism in preterm pregnancy. We have previously reported reduced placental global DNA methylation levels in preterm pregnancy. Now, we have extended the study to examine plasma levels of angiogenic factors from maternal and cord blood and correlate them with placental promoter CpG methylation and messenger RNA expression of these angiogenic genes in preterm pregnancies.
Methods
We recruited 99 women delivering at term and 90 women delivering preterm. Plasma levels of angiogenic factors, vascular endothelial growth factor (VEGF), placental growth factor (PlGF), fms-related tyrosine kinase 1 (FLT-1), and kinase insert domain receptor (KDR) were analyzed by enzyme-linked immunosorbent assay. Expression levels and promoter CpG methylation of angiogenic genes in placentae were determined by quantitative real-time polymerase chain reaction and by the Sequenom EpiTYPER technology, respectively.
Results
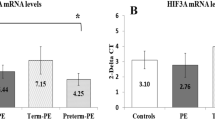
Maternal VEGF and PlGF levels (P < .01 for both) were lower but soluble FLT-1 (sFLT-1) levels and sFLT-1–PlGF ratio (P < .05 for both) were higher in the preterm group. Placental VEGF expression (P < .05) was lower, and CpG site 14 in the VEGF promoter was hypermethylated (P < .05) in the preterm group. The KDR expression (P < .05) was higher in women delivering preterm.
Conclusions
Our study provides first evidence of differential placental CpG methylation patterns and expression of VEGF, FLT-1, and KDR genes in women delivering preterm. This may explain the possible mechanism for angiogenic imbalance in the pathophysiology of preterm pregnancy.
Similar content being viewed by others
References
Demir R, Yaba A, Huppertz B. Vasculogenesis and angiogenesis in the endometrium during menstrual cycle and implantation. Acta Histochem. 2010;112(3):203–214.
Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9(6):669–676.
He Y, Smith SK, Day KA, Clark DE, Licence DR, Charnock-Jones DS. Alternative splicing of vascular endothelial growth factor (VEGF)-R1 (FLT-1) pre-mRNA is important for the regulation of VEGF activity. Mol Endocrinol. 1999;13(4):537–545.
Ebos JM, Bocci G, Man S, et al. A naturally occurring soluble form of vascular endothelial growth factor receptor 2 detected in mouse and human plasma. Mol Cancer Res. 2004;2(6):315–326.
Munaut C, Lorquet S, Pequeux C, et al. Differential expression of VEGFR-2 and its soluble form in preeclampsia. PLoS One. 2012; 7(3):e33475.
Bergers G, Benjamin LE. Tumorigenesis and the angiogenic switch. Nat Rev Cancer. 2003;3(6):401–410.
Romero R, Nien JK, Espinoza J, et al. A longitudinal study of angiogenic (placental growth factor) and anti-angiogenic (soluble endoglin and soluble vascular endothelial growth factor receptor-1) factors in normal pregnancy and patients destined to develop preeclampsia and deliver a small for gestational age neonate. J Matern Fetal Neonatal Med. 2008;21(1):9–23.
Kulkarni AV, Mehendale SS, Yadav HR, Kilari AS, Taralekar VS, Joshi SR. Circulating angiogenic factors and their association with birth outcomes in preeclampsia. Hypertens Res. 2010;33(6): 561–567.
Harris LK. IFPA Gabor Than Award lecture: Transformation of the spiral arteries in human pregnancy: key events in the remodelling timeline. Placenta. 2011;32(suppl 2):S154–S158.
Kim YM, Chaiworapongsa T, Gomez R, et al. Failure of physiologic transformation of the spiral arteries in the placental bed in preterm premature rupture of membranes. Am J Obstet Gynecol. 2002;187(5):1137–1142.
Kim YM, Bujold E, Chaiworapongsa T, et al. Failure of physiologic transformation of the spiral arteries in patients with preterm labor and intact membranes. Am J Obstet Gynecol. 2003;189(4): 1063–1069.
Ambrose JA, Barua RS. The pathophysiology of cigarette smoking and cardiovascular disease: an update. J Am Coll Cardiol. 2004;43(10):1731–1737.
Chaiworapongsa T, Romero R, Tarca A, et al. A subset of patients destined to develop spontaneous preterm labor has an abnormal angiogenic/anti-angiogenic profile in maternal plasma: evidence in support of pathophysiologic heterogeneity of preterm labor derived from a longitudinal study. J Matern Fetal Neonatal Med. 2009;22(12):1122–1139.
Sert T, Kırzıoǧlu FY, Fentoǧlu O, Aylak F, Mungan T. Serum placental growth factor, vascular endothelial growth factor, soluble vascular endothelial growth factor receptor-1 and -2 levels in periodontal disease, and adverse pregnancy outcomes. J Periodontol. 2011;82(12):1735–1748.
Andraweera PH, Dekker GA, Laurence JA, Roberts CT. Placental expression of VEGF family mRNA in adverse pregnancy outcomes. Placenta. 2012;33(6):467–472.
Daneshmand SS, Chmait RH, Moore TR, Bogic L. Preterm premature rupture of membranes: vascular endothelial growth factor and its association with histologic chorioamnionitis. Am J Obstet Gynecol. 2002;187(5):1131–1136.
Marvin KW, Keelan JA, Eykholt RL, Sato TA, Mitchell MD. Expression of angiogenic and neurotrophic factors in the human amnion and choriodecidua. Am J Obstet Gynecol. 2002;187(3): 728–734.
Galjaard S, Devlieger R, Van Assche FA. Fetal growth and developmental programming. J Perinat Med. 2013;41(1):101–105.
Joshi SR, Mehendale SS, Dangat KD, Kilari AS, Yadav HR, Taralekar VS. High maternal plasma antioxidant concentrations associated with preterm delivery. Ann Nutr Metab. 2008;53(3–4):276–282.
Kilari AS, Mehendale SS, Dangat KD, et al. Long chain polyunsaturated fatty acids in mothers of preterm babies. J Perinat Med. 2010;38(6):659–664.
Dhobale MV, Wadhwani N, Mehendale SS, Pisal HR, Joshi SR. Reduced levels of placental long chain polyunsaturated fatty acids in preterm deliveries. Prostaglandins Leukot Essent Fatty Acids. 2011;85(3–4):149–153.
Dhobale M, Chavan P, Kulkarni A, Mehendale S, Pisal H, Joshi S. Reduced folate, increased vitamin B12 and homocysteine concentrations in women delivering preterm. Ann Nutr Metab. 2012;61(1):7–14.
Dhobale M, Mehendale S, Pisal H, Nimbargi V, Joshi S. Reduced maternal and cord nerve growth factor levels in preterm deliveries. Int J Dev Neurosci. 2012;30(2):99–103.
Kulkarni A, Dangat K, Kale A, Sable P, Chavan-Gautam P, Joshi S. Effects of altered maternal folic acid, vitamin B12 and docosahexaenoic acid on placental global DNA methylation patterns in Wistar rats. PLoS One. 2011;6(3):e17706.
Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107(9):1058–1070.
Kale A, Naphade N, Sapkale S, et al. Reduced folic acid, vitamin B12 and docosahexaenoic acid and increased homocysteine and cortisol in never-medicated schizophrenia patients: implications for altered one-carbon metabolism. Psychiatry Res. 2010;175(1– 2):47–53.
Davis CD, Uthus EO. DNA methylation, cancer susceptibility, and nutrient interactions. Exp Biol Med (Maywood). 2004; 229(10):988–995.
Roberts CT. IFPA Award in placentology lecture: complicated interactions between genes and the environment in placentation, pregnancy outcome and long term health. Placenta. 2010;31 suppl:S47–S53.
Chavan-Gautam P, Sundrani D, Pisal H, Nimbargi V, Mehendale S, Joshi S. Gestation-dependent changes in human placental global DNA methylation levels. Mol Reprod Dev. 2011;78(3):150.
Sundrani DP, Reddy US, Joshi AA, et al. Differential placental methylation and expression of VEGF, FLT-1 and KDR genes in human term and preterm preeclampsia. Clin Epigenetics. 2013; 5(1):6.
Barut F, Barut A, Gun BD, et al. Intrauterine growth restriction and placental angiogenesis. Diagn Pathol. 2010;5:24.
Helske S, Vuorela P, Carpén O, Hornig C, Weich H, Halmesmäki E. Expression of vascular endothelial growth factor receptors 1, 2 and 3 in placentas from normal and complicated pregnancies. Mol Hum Reprod. 2001;7(2):205–210.
Savasan ZA, Romero R, Chaiworapongsa T, et al. Evidence in support of a role for anti-angiogenic factors in preterm prelabor rupture of membranes. J Matern Fetal Neonatal Med. 2010; 23(8):828–841.
Morgan TK, Tolosa JE, Mele L, et al. Placental villous hypermaturation is associated with idiopathic preterm birth. J Matern Fetal Neonatal Med. 2013;26(7):647–653.
Novakovic B, Yuen RK, Gordon L, et al. Evidence for widespread changes in promoter methylation profile in human placenta in response to increasing gestational age and environmental/stochastic factors. BMC Genomics. 2011;12:529.
Yuen RK, Penaherrera MS, von Dadelszen P, McFadden DE, Robinson WP. DNA methylation profiling of human placentas reveals promoter hypomethylation of multiple genes in early-onset preeclampsia. Eur J Hum Genet. 2010;18(9): 1006–1012.
Jośko J, Mazurek M. Transcription factors having impact on vascular endothelial growth factor (VEGF) gene expression in angiogenesis. Med Sci Monit. 2004;10(4):RA89–RA98.
Farré D, Roset R, Huerta M, et al. Identification of patterns in biological sequences at the ALGGEN server: PROMO and MALGEN. Nucleic Acids Res. 2003;31(13):3651–3653.
Messeguer X, Escudero R, Farré D, Núñez O, Martínez J, Albà MM. PROMO: detection of known transcription regulatory elements using species-tailored searches. Bioinformatics. 2002; 18(2):333–334.
Gibbs JR, van der Brug MP, Hernandez DG, et al. Abundant quantitative trait loci exist for DNA methylation and gene expression in human brain. PLoS Genet. 2010;6(5):e1000952.
Maunakea AK, Nagarajan RP, Bilenky M, et al. Conserved role of intragenic DNA methylation in regulating alternative promoters. Nature. 2010;466(7303):253–257.
Zhang D, Cheng L, Badner JA, et al. Genetic control of individual differences in gene-specific methylation in human brain. Am J Hum Genet. 2010;86(3):411–419.
Brenet F, Moh M, Funk P, et al. DNA methylation of the first exon is tightly linked to transcriptional silencing. PLoS One. 2011;6(1): e14524.
Sundrani D, Khot V, Pisal H, et al. Gestation dependant changes in angiogenic factors and their associations with fetal growth measures in normotensive pregnancy. PLoS One. 2013;8(1):e54153.
Weinberger B, Anwar M, Hegyi T, Hiatt M, Koons A, Paneth N. Antecedents and neonatal consequences of low Apgar scores in preterm newborns: a population study. Arch Pediatr Adolesc Med. 2000;154(3):294–300.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sundrani, D.P., Reddy, U.S., Chavan-Gautam, P.M. et al. Altered Methylation and Expression Patterns of Genes Regulating Placental Angiogenesis in Preterm Pregnancy. Reprod. Sci. 21, 1508–1517 (2014). https://doi.org/10.1177/1933719114532838
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719114532838