Abstract
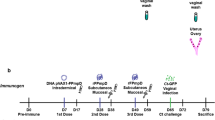
We recently demonstrated that co-administration of mouse zona pellucida 3 (mZP3) DNA and protein vaccine enhanced the contraception of mice by increasing humoral immune responses. In this study, we try to use granulocyte—macrophage colony-stimulating factor (GM-CSF) to further improve the humoral immune responses induced by mZP3 DNA and protein co-administration. BALB/c mice were intranasally pre-injected with GM-CSF 4 days before co-administration. Compared to DNA and protein coadministration without GM-CSF, the combination of GM-CSF and coadministration significantly enhances humoral immune responses, especially the level of secretory immunoglobulin A (sIgA) in vaginal washes. The enhanced antibody responses are correlated with the upregulated level of interleukin 4 (IL-4) and enhanced maturation of dendritic cells (DCs). Thus, GM-CSF is a potential candidate adjuvant to be used for the development of a safe and effective contraceptive vaccine.
Similar content being viewed by others
References
Aitken RJ, Paterson M, van Duin M. The potential of the zona pellucida as a target for immunocontraception. Am J Reprod Immunol. 1996;35(3):175–180.
Greenhouse S, Castle PE, Dean J. Antibodies to human ZP3 induce reversible contraception in transgenic mice with ‘humanized’ zonae pellucidae. Hum Reprod. 1999;14(3):593–600.
Bleil JD, Wassarman PM. Structure and function of the zona pellucida: identification and characterization of the proteins of the mouse oocyte’s zona pellucida. Dev Biol. 1980;76(1):185–202.
Shimizu S, Tsuji M, Dean J. In vitro biosynthesis of three sulfated glycoproteins of murine zonae pellucidae by oocytes grown in follicle culture. J Biol Chem. 1983;258(9):5858–5863.
Paterson M, Koothan PT, Morris KD, et al. Analysis of the contraceptive potential of antibodies against native and deglycosylated porcine ZP3 in vivo and in vitro. Biol Reprod. 1992;46(4): 523–534.
Paterson M, Wilson MR, Morris KD, van Duin M, Aitken RJ. Evaluation of the contraceptive potential of recombinant human ZP3 and human ZP3 peptides in a primate model: their safety and efficacy. Am J Reprod Immunol. 1998;40(3):198–209.
Clydesdale G, Pekin J, Beaton S, Jackson RJ, Vignarajan S, Hardy CM. Contraception in mice immunized with recombinant zona pellucida subunit 3 proteins correlates with Th2 responses and the levels of interleukin 4 expressed by CD4+ cells. Reproduction. 2004;128(6):737–745.
Jackson RJ, Maguire DJ, Hinds LA, Ramshaw IA. Infertility in mice induced by a recombinant ectromelia virus expressing mouse zona pellucida glycoprotein 3. Biol Reprod. 1998;58(1): 152–159.
Lloyd ML, Shellam GR, Papadimitriou JM, Lawson MA. Immunocontraception is induced in BALB/c mice inoculated with murine cytomegalovirus expressing mouse zona pellucida 3. Biol Reprod. 2003;68(6):2024–2032.
Hasegawa A, Hamada Y, Shigeta M, Koyama K. Contraceptive potential of synthetic peptides of zona pellucida protein (ZPA). J Reprod Immunol. 2002;53(1–2):91–98.
Sivapurapu N, Upadhyay A, Hasegawa A, Koyama K, Gupta SK. Native zona pellucida reactivity and in-vitro effect on human sperm-egg binding with antisera against bonnet monkey ZP1 and ZP3 synthetic peptides. J Reprod Immunol. 2002;56(1–2):77–91.
Ringleb J, Rohleder M, Jewgenow K. Impact of feline zona pellucida glycoprotein B-derived synthetic peptides on in vitro fertilization of cat oocytes. Reproduction. 2004;127(2):179–186.
Borillo J, Coonrod SA, Wu J, Zhou C, Lou Y. Antibodies to two ZP3 B cell epitopes affect zona pellucida assembly. J Reprod Immunol. 2008;78(2):149–157.
Hardy CM, Beaton S, Hinds LA. Immunocontraception in mice using repeated, multi-antigen peptides: immunization with purified recombinant antigens. Mol Reprod Dev. 2008;75(1):126–135.
Li J, Jin H, Zhang A, Li Y, Wang B, Zhang F. Enhanced contraceptive response by co-immunization of DNA and protein vaccines encoding the mouse zona pellucida 3 with minimal oophoritis in mouse ovary. J Gene Med. 2007;9(12):1095–1103.
Zhang A, Li J, Zhao G, et al. Intranasal co-administration with the mouse zona pellucida 3 expressing construct and its coding protein induces contraception in mice. Vaccine. 2011;29(39): 6785–6792.
Perales MA, Yuan J, Powel S, et al. Phase I/II study of GM-CSF DNA as an adjuvant for a multipeptide cancer vaccine in patients with advanced melanoma. Mol Ther. 2008;16(12):2022–2029.
Qing Y, Chen M, Zhao J, et al. Construction of an HBV DNA vaccine by fusion of the GM-CSF gene to the HBV-S gene and examination of its immune effects in normal and HBV-transgenic mice. Vaccine. 2010;28(26):4301–4307.
Hirata Y, Egea L, Dann SM, Eckmann L, Kagnoff MF. GM-CSF-facilitated dendritic cell recruitment and survival govern the intestinal mucosal response to a mouse enteric bacterial pathogen. Cell Host Microbe. 2010;7(2):151–163.
Jones T, Stern A, Lin R. Potential role of granulocyte-macrophage colony-stimulating factor as vaccine adjuvant. Eur J Clin Microbiol Infect Dis. 1994;13 (suppl 2):S47–S53.
Inaba K, Inaba M, Romani N, et al Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J Exp Med. 1992;176(6):1693–1702.
Kusakabe K, Xin KQ, Katoh H, et al. The timing of GM-CSF expression plasmid administration influences the Th1/Th2 response induced by an HIV-1-specific DNA vaccine. J Immunol. 2000;164(6):3102–3111.
Illum L, Jabbal-Gill I, Hinchcliffe M, Fisher AN, Davis SS. Chitosan as a novel nasal delivery system for vaccines. Adv Drug Deliv Rev. 2001;51(1–3):81–96.
Nishi E, Kume N, Ueno Y, Ochi H, Moriwaki H, Kita T. Lysophosphatidylcholine enhances cytokine-induced interferon gamma expression in human T lymphocytes. Circ Res. 1998;83(5): 508–515.
Perrin-Cocon L, Agaugue S, Coutant F, et al. Lysophosphatidylcholine is a natural adjuvant that initiates cellular immune responses. Vaccine. 2006;24(9):1254–1263.
Sung CM, Yeh CT, Shiau SS, Liang CK, Chang ML. Hydrodynamics-based transfection of the combination of betacellulin and neurogenic differentiation 1 DNA ameliorates hyperglycemia in mice with streptozotocin-induced diabetes. Diabetes Technol Ther. 2011;13:519–525.
Hardy CM, ten Have JF, Mobbs KJ, Hinds LA. Assessment of the immunocontraceptive effect of a zona pellucida 3 peptide antigen in wild mice. Reprod Fertil Dev. 2002;14(3–4):151–155.
Mellstedt H, Fagerberg J, Osterborg A. Local low-dose of soluble GM-CSF significantly augments an immune response against tumour antigens in man. Eur J Cancer. 1999;35 (suppl 3): S29–S32.
Hardy CM, ten Have JF, Pekin J, Beaton S, Jackson RJ, Clydesdale G. Contraceptive responses of mice immunized with purified recombinant mouse zona pellucida subunit 3 (mZP3) proteins. Reproduction. 2003;126(1):49–59.
Naz RK, Chauhan SC. Human sperm-specific peptide vaccine that causes long-term reversible contraception. Biol Reprod. 2002;67(2):674–680.
Lou YH, Park KK, Agersborg S, Alard P, Tung KS. Retargeting T cell-mediated inflammation: a new perspective on autoantibody action. J Immunol. 2000;164(10):5251–5257.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kadir, Z., Ma, X., Li, J. et al. Granulocyte—Macrophage Colony-Stimulating Factor Enhances the Humoral Immune Responses of Mouse Zona Pellucida 3 Vaccine Strategy Based on DNA and Protein Coadministration in BALB/c Mice. Reprod. Sci. 20, 400–407 (2013). https://doi.org/10.1177/1933719112459236
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719112459236