Abstract
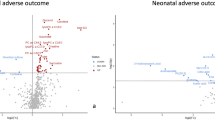
Metabolomics offers a powerful holistic approach to examine the metabolite composition of biofluids to identify disruptions present in disease. We used ultra performance liquid chromatography–mass spectroscopy on the maternal serum obtained in the third trimester to address the hypothesis that pregnancies ending in poor outcomes (small for gestational age infant, preterm birth, or neonatal intensive care admission, n = 40) would have a different maternal serum metabolic profiles to matched healthy pregnancies (n = 40). Ninety-eight identified metabolic features differed between normal and poor pregnancy outcomes. Classes of metabolites perturbed included free fatty acids, glycerolipids, progesterone metabolites, sterol lipids, vitamin D metabolites, and sphingolipids; these highlight potential molecular mechanisms associated with pregnancy complications in the third trimester linked by placental dysfunction. In this clinical setting, metabolomics has the potential to describe differences in fetoplacental and maternal metabolites in pregnancies with poor pregnancy outcomes compared with controls.
Similar content being viewed by others
References
Brosens I, Pijnenborg R, Vercruysse L, Romero R. The "Great Obstetrical Syndromes" are associated with disorders of deep pla-centation. Am J Obstet Gynecol. 2011;204(3):193–201.
Confidential Enquiry into Maternal and Child Health. Perinatal Mortality 2007: England, Wales and Northern Ireland. London: Confidential Enquiry into Maternal and Child Health; 2009.
Gardosi J, Kady SM, McGeown P, Francis A, Tonks A. Classification of stillbirth by relevant condition at death (ReCoDe): population based cohort study. BMJ. 2005 ;331(7525): 1113–1117.
Haws RA, Yakoob MY, Soomro T, Menezes EV, Darmstadt GL, Bhutta ZA. Reducing stillbirths: screening and monitoring during pregnancy and labour. BMC Pregnancy Childbirth. 2009; 9(suppl 1):S5.
Dunn WB, Broadhurst DI, Atherton HJ, Goodacre R, Griffin JL. Systems level studies of mammalian metabolomes: the roles of mass spectrometry and nuclear magnetic resonance spectroscopy. Chem Soc Rev. 2011;40(l):387–426.
Heazell AE, Brown M, Worton SA, Dunn WB. Review: the effects of oxygen on normal and pre-eclamptic placental tissue—insights from metabolomics. Placenta. 32(suppl 2):S119–S124.
Kenny LC, Broadhurst D, Brown M, et al. Detection and identification of novel metabolomic biomarkers in preeclampsia. Reprod Sci. 2008;15(6):591–597.
Kenny LC, Broadhurst DI, Dunn W, et al. Robust early pregnancy prediction of later preeclampsia using metabolomic biomarkers. Hypertension. 2010;56(4):741–749.
Odibo AO, Goetzinger KR, Odibo L, et al. First-trimester prediction of preeclampsia using metabolomic biomarkers: a discovery phase study. Prenat Diagn. 2011;31(10):990–994.
Romero R, Mazaki-Tovi S, Vaisbuch E, et al. Metabolomics in premature labor: a novel approach to identify patients at risk for preterm delivery. J Matern Fetal Neonatal Med. 2010;23(12): 1344–1359.
Graca G, Duarte IF, Barros AS, et al. Impact of prenatal disorders on the metabolic profile of second trimester amniotic fluid: a nuclear magnetic resonance metabonomic study. J Proteome Res. 2010;9(ll):6016–6024.
Heazell AE, Brown M, Dunn WB, et al. Analysis of the metabolic footprint and tissue metabolome of placental villous explants cultured at different oxygen tensions reveals novel redox biomarkers. Placenta. 2008;29(8):691–698.
Dunn WB, Brown M, Worton SA, et al. Changes in the metabolic footprint of placental explant-conditioned culture medium identifies metabolic disturbances related to hypoxia and pre-eclampsia. Placenta. 2009;30(ll):974–980.
Horgan RP, Broadhurst DI, Dunn WB, et al. Changes in the metabolic footprint of placental explant-conditioned medium cultured in different oxygen tensions from placentas of small for gestational age and normal pregnancies. Placenta. 2009;31(10): 893–901.
Warrander LK, Kroll J, Greenwood SL, Sibley CP, Jones RL, Heazell AEP. Placentally-derived factors may be used to predict poor pregnancy outcome in reduced fetal movements. Arch Dis Child Fetal Neonatal Ed. 201 l;96:Fal2.
Dunn WB, Broadhurst D, Begley P, et al. Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry. Nat Protoc. 2011;6(7):1060–1083.
Smith CA, Want EJ, O'Maille G, Abagyan R, Siuzdak G. XCMS: processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Anal Chem. 2006;78(3):779–787.
Sumner LW, Amberg A, Barrett D, et al. Proposed minimum reporting standards for chemical analysis. Metabolomics. 2007; 3:211–221.
Brown M, Wedge DC, Goodacre R, et al. Automated workflows for accurate mass-based putative metabolite identification in LC/ MS-derived metabolomic datasets. Bioinformatics. 2011;27(8): 1108–1112.
Redline RW. Clinically and biologically relevant patterns of placental inflammation. Pediatr Dev Pathol. 2002;5(4):326–328.
Heazell AE, Moll SJ, Jones CJ, Baker PN, Crocker IP. Formation of syncytial knots is increased by hyperoxia, hypoxia and reactive oxygen species. Placenta. 2007;28(suppl A):S33–S40.
Vcelakova H, Hill M, Lapcik O, Parizek A. Determination of 17alpha-hydroxypregnenolone sulfate and its application in diagnostics. Steroids. 2007;72(4):323–327.
Salas SP, Marshall G, Gutierrez BL, Rosso P. Time course of maternal plasma volume and hormonal changes in women with preeclampsia or fetal growth restriction. Hypertension. 2006; 47(2):203–208.
Ligam P, Manuelpillai U, Wallace EM, Walker D. Localisation of indoleamine 2,3-dioxygenase and kynurenine hydroxylase in the human placenta and decidua: implications for role of the kynurenine pathway in pregnancy. Placenta. 2005;26(6):498–504.
Kamimura S, Eguchi K, Yonezawa M, Sekiba K. Localization and developmental change of indoleamine 2,3-dioxygenase activity in the human placenta. Acta Med Okayama. 1991 ;45(3): 135–139.
Kudo Y, Boyd CA. Human placental indoleamine 2,3-dioxygenase: cellular localization and characterization of an enzyme preventing fetal rejection. Biochim Biophys Acta. 2000; 1500(1):119–124.
Dunn WB, Brown M, Worton SA, et al. The metabolome of human placental tissue: investigation of first trimester tissue and changes related to preeclampsia in late pregnancy. Metabolomics. 2011;In Press. DOI: 10.1007/sl 1306-011-0348-6
How HY, Cook CR, Cook VD, et al. The pattern of change in the lecithin/sphingomyelin ratio in patients with preterm premature rupture of membranes between 24 and 34 weeks' gestation. J Perinatol. 2002;22(l):21–25.
Torrance HL, Voorbij HA, Wijnberger LD, van Bel F, Visser GH. Lung maturation in small for gestational age fetuses from pregnancies complicated by placental insufficiency or maternal hypertension. Early Hum Dev. 2008;84(7):465–469.
Wymann MP, Schneiter R. Lipid signalling in disease. Nat Rev Mol Cell Biol. 2008;9(2):162–176.
Robinson CJ, Wagner CL, Hollis BW, Baatz JE, Johnson DD. Maternal vitamin D and fetal growth in early-onset severe preeclampsia. Am J Obstet Gynecol. 2011;204(6):556 e551–e554.
Baker AM, Haeri S, Camargo CA JrStuebe AM, Boggess KA. A nested case-control study of first-trimester maternal vitamin D status and risk for spontaneous preterm birth. Am J Perinatol. 2011;28(9):667–672.
Specker BL. Does vitamin D during pregnancy impact offspring growth and bone? Proc Nutr Soc. 2012;71(l):38–45.
Bodnar LM, Catov JM, Zmuda JM, et al. Maternal serum 25-hydroxyvitamin D concentrations are associated with small-for-gestational age births in white women. J Nutr. 2010;140(5): 999–1006.
Brough L, Rees GA, Crawford MA, Morton RH, Dorman EK. Effect of multiple-micronutrient supplementation on maternal nutrient status, infant birth weight and gestational age at birth in a low-income, multi-ethnic population. Br J Nutr. 2010; 104(3):437–445.
Horgan RP, Clancy OH, Myers JE, Baker PN. An overview of proteomic and metabolomic technologies and their application to pregnancy research. BJOG. 2009;116(2):173–181.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heazell, A.E.P., Bernatavicius, G., Warrander, L. et al. A Metabolomic Approach Identifies Differences in Maternal Serum in Third Trimester Pregnancies That End in Poor Perinatal Outcome. Reprod. Sci. 19, 863–875 (2012). https://doi.org/10.1177/1933719112438446
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719112438446