Abstract
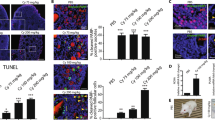
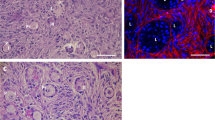
This study investigated the cytoprotective effects of amifostine against the adverse effects of cyclophosphamide relying on ovarian cell death markers and the fertilization rate of the surviving follicles as a late outcome of the study. Combined pretreatment of amifostine with cyclophosphamide enabled partial recovery of antiapoptotic messenger RNA (mRNA) expression levels while a decrease in expression of BAX and Casp3 were identified. The pretreatment of amifostine to cyclophosphamide significantly reduced the proportion of terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL)-positive primary and preantral follicles (P < .001 and P < .05, respectively). These findings were comparable with the results obtained from protein expression of cleaved caspase 3. The fertilization rate showed a significant capability of amifostine to improve fertilization potency of oocytes exposed to cyclophosphamide (P < .01). In conclusion, administration of amifostine prior to cyclophosphamide might serve as a promising protocol to improve female fertility.
Similar content being viewed by others
References
Bukowski R. Cytoprotection in the treatment of pediatric cancer: review of current strategies in adults and their application to children. Med Pediatr Oncol. 1999;32(2):124–134.
Colvin OM. An overview of cyclophosphamide development and clinical applications. Curr Pharm Des. 1999;5(8):555–560.
Wilkinson A. Progress in the clinical application of immunosuppressive drugs in renal transplantation. Curr Opin Nephrol Hyper-tens. 2001;10(6):763–770.
Meirow D, Assad G, Dor J, Rabinovici J. The GnRH antagonist cetrorelix reduces cyclophosphamide-induced ovarian follicular destruction in mice. Hum Reprod. 2004;19(6):1294–1299.
Plowchalk DR, Meadows MJ, Mattison DR. Reproductive toxicity of cyclophosphamide in the C57BL/6N mouse: 2. Effects on uterine structure and function. Reprod Toxicol. 1992;6(5):423–429.
Lopez SG, Luderer U. Effects of cyclophosphamide and buthio-nine sulfoximine on ovarian glutathione and apoptosis. Free Radic Biol Med. 2004;36(11):1366–1377.
Howell S, Shalet S. Gonadal damage from chemotherapy and radiotherapy. Endocrinol Metab Clin North Am. 1998;27(4):927–943.
Kumar R, Biggart JD, McEvoy J, McGeown MG. Cyclophosphamide and reproductive function. Lancet. 1972;1(7762):1212–1214.
Barekati Z, Gourabi H, Valojerdi MR, Yazdi PE. Previous maternal chemotherapy by cyclophosphamide (Cp) causes numerical chromosome abnormalities in preimplantation mouse embryos. Reprod Toxicol. 2008;26(3–4):278–281.
Pydyn EF, Ataya KM. Effect of cyclophosphamide on mouse oocyte in vitro fertilization and cleavage: recovery. Reprod Toxicol. 1991;5(1):73–78.
Ataya KM, Pydyn EF, Sacco AG. Effect of “activated” cyclophosphamide on mouse oocyte in vitro fertilization and cleavage. Reprod Toxicol. 1988;2(2):105–109.
Partridge AH, Gelber S, Peppercorn J, et al. Web-based survey of fertility issues in young women with breast cancer. J Clin Oncol. 2004;22(20):4174–4183.
Anchan RM, Ginsburg ES. Fertility concerns and preservation in younger women with breast cancer. Crit Rev Oncol Hematol. 2010;74(3):175–192.
Kim SS. Fertility preservation in female cancer patients: current developments and future directions. Fertil Steril. 2006;85(1):1–11.
Nguyen NP, Levinson B, Dutta S, et al. Amifostine and curative intent chemoradiation for compromised cancer patients. Anticancer Res. 2003;23(2C):1649–1656.
Brizel DM, Wasserman TH, Henke M, et al. Phase III randomized trial of amifostine as a radioprotector in head and neck cancer. J Clin Oncol. 2000;18(19):3339–3345.
Culy CR, Spencer CM. Amifostine: an update on its clinical status as a cytoprotectant in patients with cancer receiving chemotherapy or radiotherapy and its potential therapeutic application in myelodysplastic syndrome. Drugs. 2001;61(5):641–684.
Links M, Lewis C. Chemoprotectants: a review of their clinical pharmacology and therapeutic efficacy. Drugs. 1999;57(3):293–308.
Calabro-Jones PM, Aguilera JA, Ward JF, Smoluk GD, Fahey RC. Uptake of WR-2721 derivatives by cells in culture: identification of the transported form of the drug. Cancer Res. 1988;48(13):3634–3640.
Smoluk GD, Fahey RC, Calabro-Jones PM, Aguilera JA, Ward JF. Radioprotection of cells in culture by WR-2721 and derivatives: form of the drug responsible for protection. Cancer Res. 1988;48(13):3641–3647.
List AF, Heaton R, Glinsmann-Gibson B, Capizzi RL. Amifostine protects primitive hematopoietic progenitors against chemotherapy cytotoxicity. Semin Oncol. 1996;23(4 suppl 8):58–63.
Peters GJ, van der Vijgh WJ. Protection of normal tissues from the cytotoxic effects of chemotherapy and radiation by amifostine (WR-2721): preclinical aspects. Eur J Cancer. 1995;31(suppl 1):S1–S7.
Santini V, Giles FJ. The potential of amifostine: from cytoprotectant to therapeutic agent. Haematologica. 1999;84(11):1035–1042.
Yuhas JM, Culo F. Selective inhibition of the nephrotoxicity of cis-dichlorodiammineplatinum(II) by WR-2721 without altering its antitumor properties. Cancer Treat Rep. 1980;64(1):57–64.
Wasserman TH, Phillips TL, Ross G, Kane LJ. Differential protection against cytotoxic chemotherapeutic effects on bone marrow CFUs by Wr-2721. Cancer Clin Trials. 1981;4(1):3–6.
Valeriote F, Tolen S. Protection and potentiation of nitrogen mustard cytotoxicity by WR-2721. CancerRes. 1982;42(11):4330–4331.
Jahnukainen K, Jahnukainen T, Salmi TT, Svechnikov K, Eksborg S, Soder O. Amifostine protects against early but not late toxic effects of doxorubicin in infant rats. Cancer Res. 2001;61(17):6423–6427.
Andrieu MN, Kurtman C, Hicsonmez A, Ozbilgin K, Eser E, Erdemli E. In vivo study to evaluate the protective effects of amifostine on radiation-induced damage of testis tissue. Oncology. 2005;69(1):44–51.
Hou M, Chrysis D, Nurmio M, et al. Doxorubicin induces apoptosis in germ line stem cells in the immature rat testis and amifostine cannot protect against this cytotoxicity. Cancer Res. 2005;65(21):9999–10005.
Vendramini V, Sasso-Cerri E, Miraglia SM. Amifostine reduces the seminiferous epithelium damage in doxorubicin-treated prepubertal rats without improving the fertility status. Reprod Biol Endocrinol. 2010;8:3.
Lirdi LC, Stumpp T, Sasso-Cerri E, Miraglia SM. Amifostine protective effect on cisplatin-treated rat testis. Anat Rec (Hoboken). 2008;291(7):797–808.
Yoon YD, Kim JH, Lee KH, Kim JK. Amifostine has an inhibitory effect on the radiation-induced p53-branched cascade in the immature mouse ovary. In Vivo. 2005;19(3):509–514.
Meirow D, Lewis H, Nugent D, Epstein M. Subclinical depletion of primordial follicular reserve in mice treated with cyclophosphamide: clinical importance and proposed accurate investigative tool. Hum Reprod. 1999;14(7):1903–1907.
Nagy A GM, Vintersten K, Behringer R, eds. Manipulating the mouse embryo. CSHL press. 2003;3rd ed.
Vandesompele J, De Preter K, Pattyn F, et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002;3:RESEARCH0034.
Brady HJ, Salomons GS, Bobeldijk RC, Berns AJ. T cells from baxalpha transgenic mice show accelerated apoptosis in response to stimuli but do not show restored DNA damage-induced cell death in the absence of p53. EMBO J. 1996;15(6):1221–1230.
Familiari G, Caggiati A, Nottola SA, Ermini M, Di Benedetto MR, Motta PM. Ultrastructure of human ovarian primordial follicles after combination chemotherapy for Hodgkin’s disease. Hum Reprod. 1993;8(12):2080–2087.
Bedaiwy MA, Shahin AY, Falcone T. Reproductive organ transplantation: advances and controversies. Fertil Steril. 2008;90(6):2031–2055.
Crow MT, Mani K, Nam YJ, Kitsis RN. The mitochondrial death pathway and cardiac myocyte apoptosis. Circ Res. 2004;95(10):957–970.
Strauss G, Westhoff MA, Fischer-Posovszky P, et al. 4-hydroperoxy-cyclophosphamide mediates caspase-independent T-cell apoptosis involving oxidative stress-induced nuclear relocation of mitochondrial apoptogenic factors AIF and EndoG. Cell Death Differ. 2008;15(2):332–343.
Zhao XJ, Huang YH, Yu YC, Xin XY. GnRH antagonist cetrorelix inhibits mitochondria-dependent apoptosis triggered by chemotherapy in granulosa cells of rats. Gynecol Oncol. 2010;118(1):69–75.
Ishida H. The research method for investigating the role of the mitochondrial permeability transition pore in cell death [in Japanese]. Nihon Yakurigaku Zasshi. 2004;123(5):329–334.
Crompton M. The mitochondrial permeability transition pore and its role in cell death. Biochem J. 1999;341(pt 2):233–249.
Huang YH, Zhao XJ, Zhang QH, Xin XY. The GnRH antagonist reduces chemotherapy-induced ovarian damage in rats by suppressing the apoptosis. Gynecol Oncol. 2009;112(2):409–414.
Fu X, He Y, Xie C, Liu W. Bone marrow mesenchymal stem cell transplantation improves ovarian function and structure in rats with chemotherapy-induced ovarian damage. Cytotherapy. 2008;10(4):353–363.
Jarrell JF, Bodo L, YoungLai EV, Barr RD, O’Connell GJ. The short-term reproductive toxicity of cyclophosphamide in the female rat. Reprod Toxicol. 1991;5(6):481–485.
Bokser L, Szende B, Schally AV. Protective effects of D-Trp6-luteinising hormone-releasing hormone microcapsules against cyclophosphamide-induced gonadotoxicity in female rats. Br J Cancer. 1990;61(6):861–865.
Tsai-Turton M, Luong BT, Tan Y, Luderer U. Cyclophosphamide-induced apoptosis in COV434 human granulosa cells involves oxidative stress and glutathione depletion. Toxicol Sci. 2007;98(1):216–230.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barekati, Z., Golkar-Narenji, A., Totonchi, M. et al. Effects of Amifostine in Combination With Cyclophosphamide on Female Reproductive System. Reprod. Sci. 19, 539–546 (2012). https://doi.org/10.1177/1933719111426599
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719111426599