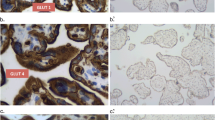
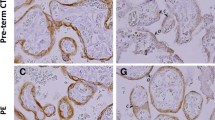
Abstract
Glucose transport from the maternal to fetal side of the placenta is critical for fetal growth and development due to the absence of fetal gluconeogenesis. Human GLUT9, existing as 2 isoforms, is a novel member of the transporter family. This study investigated the localization and relative expression levels of these isoforms in the human term placenta from both control and diabetic patients. Placenta samples were collected from normal pregnancies and those complicated by maternal diabetes (White classifications A1, A2, and B). Antibodies specific for the different isoforms were used to detect expression. Both forms of the protein are expressed in syncytiotrophoblast cells. Subcellular fractionation revealed an asymmetrical expression pattern with GLUT9a on basal membranes, whereas GLUT9b localizes to microvillus membranes. Expression of both isoforms is significantly increased in placental tissue from diabetic pregnancies. Altered expression of GLUT9 in the placenta may play a role in the fetal pathophysiology associated with diabetes-complicated pregnancies.
Similar content being viewed by others
References
Barros LF, Yudilevich DL, Jarvis SM, Beaumont N, Baldwin SA. Quantitation and immunolocalization of glucose transporters in the human placenta. Placenta. 1995;16(7):623–633.
Jansson T, Wennergren M, Illsley NP. Glucose transporter protein expression in human placenta throughout gestation and in intrauterine growth retardation. J Clin Endocrinol Metab. 1993;77(6):1554–1562.
Takata K, Kasahara T, Kashahara M, Ezaki O, Hirano H. Localization of erythrocyte/HepG2-type glucose transporter (GLUT1) in human placental villi. Cell Tissue Res. 1992;267(3):407–412.
Hauguel-de Mouzon S, Challier JC, Kacemi A, Cauzac M, Malek A, Girard J. The GLUT3 glucose transporter isoform is differentially expressed within human placental cell types. J Clin Endocrinol Metab. 1997;82(8):2689–2694.
Xing AY, Challier JC, Lepercq J, et al. Unexpected expression of glucose transporter 4 in villous stromal cells of human placenta. J Clin Endocrinol Metab. 1998;83(11):4097–4101.
Gude NM, Stevenson JL, Rogers S, et al. GLUT12 expression in human placenta in first trimester and term. Placenta. 2003;24(5):566–570.
Scheepers A, Schmidt S, Manolescu A, et al. Characterization of the human SLC2A11 (GLUT11) gene: alternative promoter usage, function, expression, and subcellular distribution of three isoforms, and lack of mouse orthologue. Mol Membr Biol. 2005;22(4):339–351.
Gude NM, Stevenson JL, Murthi P, et al. Expression of GLUT12 in the fetal membranes of the human placenta. Placenta. 2005;26(1):67–72.
Augustin R, Carayannopoulos MO, Dowd LO, Phay JE, Moley JF, Moley KH. Identification and characterization of human glucose transporter-like protein-9 (GLUT9): alternative splicing alters trafficking. J Biol Chem. 2004;279(16):16229–16236.
Manolescu A, Augustin R, Moley KH, Cheeseman CI. A highly conserved hydrophobic motif in the exofacial vestibule of fructose transporting SLC2A proteins acts as a critical determinant of their substrate selectivity. Mol Membr Biol. 2007;24(5–6):1–9.
Ben-Haroush A, Yogev Y, Hod M. Epidemiology of gestational diabetes mellitus and its association with Type 2 diabetes. Diabet Med. 2004;21(2):103–113.
Vohr BR, McGarvey ST, Tucker R. Effects of maternal gestational diabetes on offspring adiposity at 4–7 years of age. Diabetes Care. 1999;22(8):1284–1291.
Lindsay RS, Dabelea D, Roumain J, Hanson RL, Bennett PH, Knowler WC. Type 2 diabetes and low birth weight: the role of paternal inheritance in the association of low birth weight and diabetes. Diabetes. 2000;49(3):445–449.
Silverman BL, Rizzo TA, Cho NH, Metzger BE. Long-term effects of the intrauterine environment. The Northwestern University Diabetes in Pregnancy Center. Diabetes Care. 1998;21(suppl 2):B142–B149.
Gaither K, Quraishi AN, Illsley NP. Diabetes alters the expression and activity of the human placental GLUT1 glucose transporter. J Clin Endocrinol Metab. 1999;84(2):695–701.
Carayannopoulos MO, Schlein A, Wyman A, Chi M, Keembiyehetty C, Moley KH. GLUT9 is differentially expressed and targeted in the preimplantation embryo. Endocrinology. 2004;145(3):1435–1443.
Evans SA, Doblado M, Chi MM, Corbett JA, Moley KH. Facilitative glucose transporter 9 expression affects glucose sensing in pancreatic beta-cells. Endocrinology. 2009;150(12):5302–5310.
Kim ST, Moley KH. The expression of GLUT8, GLUT9a, and GLUT9b in the mouse testis and sperm. Reprod Sci. 2007;14(5):445–455.
Kim ST, Moley KH. Regulation of facilitative glucose transporters and AKT/MAPK/PRKAA signaling via estradiol and progesterone in the mouse uterine epithelium. Biol Reprod. 2009;81(1):188–198.
Jansson T, Powell TL, Illsley NP. Non-electrolyte solute permeabilities of human placental microvillous and basal membranes. J Physiol. 1993;468:261–274.
Baumann MU, Deborde S, Illsley NP. Placental glucose transfer and fetal growth. Endocrine. 2002;19(1):13–22.
Jansson T, Wennergren M, Powell TL. Placental glucose transport and GLUT 1 expression in insulin-dependent diabetes. Am J Obstet Gynecol. 1999;180(1 pt 1):163–168.
Keembiyehetty C, Augustin R, Carayannopoulos MO, et al. Mouse glucose transporter 9 splice variants are expressed in adult liver and kidney and are up-regulated in diabetes. Mol Endocrinol. 2006;20(3):686–697.
Hagerman DD, Villee CA. The transport of fructose by human placenta. J Clin Invest. 1952;31(10):911–913.
Bertolini M, Wallace C, Anderson G. Expression profile and protein levels of placental products as indirect measures of placental function in in vitro-derived bovine pregnancies. Reproduction. 2006;131(1):163–173.
Rand EB, Depaoli AM, Davidson NO, Bell GI, Burant CF. Sequence, tissue distribution, and functional characterization of the rat fructose transporter GLUT5. Am J Physiol. 1993;264(6 pt 1):G1169–G1176.
Kawasaki T, Akanuma H, Yamanouchi T. Increased fructose concentrations in blood and urine in patients with diabetes. Diabetes Care. 2002;25(2):353–357.
Meznarich HK, Hay WW Jr. Sparks JW, Meschia G, Battaglia FC. Fructose Disposal and oxidation rates in the ovine fetus. Exp Physiol. 1987;72(4):617–625.
Pere MC. Maternal and fetal blood levels of glucose, lactate, fructose and insulin in the conscious pig. J Anim Sci. 1995;73(10):2994–2999.
Morris ED, Wood C, Archer GD. The effect on cord blood glucose levels of the intravenous administsration of fructose to the mother. J Obstet Gynecol Br Commonw. 1964;71:766–767.
Thorburn AW, Storlien LH, Jenkins AB, Khouri S, Kraegen EW. Fructose-induced in vivo insulin resistance and elevated plasma triglyceride levels in rats. Am J Clin Nutr. 1989;49(6):1155–1163.
Lee MK, Miles PD, Khoursheed M, Gao KM, Moossa AR, Olefsky JM. Metabolic effects of troglitazone on fructose-induced insulin resistance in the rat. Diabetes. 1994;43(12):1435–1439.
Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54(6):1615–1625.
Delbosc S, Paizanis E, Magous R, et al. Involvement of oxidative stress and NADPH oxidase activation in the development of cardiovascular complications in a model of insulin resistance, the fructose-fed rat. Atherosclerosis. 2005;179(1):43–49.
Zhao W, Devamanoharan PS, Varma SD. Fructose induced deactivation of antioxidant enzymes: preventive effect of pyruvate. Free Radic Res. 2000;33(1):23–30.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bibee, K.P., Illsley, N.P. & Moley, K.H. Asymmetric Syncytial Expression of GLUT9 Splice Variants in Human Term Placenta and Alterations in Diabetic Pregnancies. Reprod. Sci. 18, 20–27 (2011). https://doi.org/10.1177/1933719110380276
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719110380276