Abstract
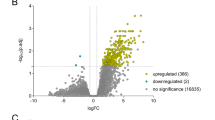
Human endometrium undergoes modifications in preparation for embryonic implantation. This study investigated in vivo the endocrine effects of pregnancy on the endometrium, using the model of ectopic pregnancy. Endometrial biopsies from 9 subjects with ectopic pregnancy (Preg) were compared with 8 and 6 samples of mid and late secretory endometrium, respectively. After hybridizing with Affymetrix HGU133 Plus 2 chips, data were analyzed using GeneSpring GX and Ingenuity Pathways Analysis. From 54 675 genes, 3021 genes were significantly differentiated when mid-secretory endometrium was compared with the Preg (Volcano plot; P < .05, ≥2-fold change). The complement and coagulation cascade, phospholid degradation, glycosphingolipid biosynthesis (globoseries), retinol metabolism, antigen presentation pathway, glycosphingolipid biosynthesis, and O-glycan biosynthesis were main significant canonical pathways found in Preg samples. Validation was done with reverse transcriptase polymerase chain reaction. In conclusion, the ectopic embryo has a significant impact, by an endocrine mechanism, on endometrium, when compared with the window of implantation.
Similar content being viewed by others
References
Noyes RW, Hertig AT, Rock J. Dating the endometrial biopsy. Fertil Steril. 1950;1:3–25.
Lessey BA, Damjanovich L., Coutifaris C., Castelbaum A., Albelda SM, Buck CA Integrins adhesion molecules in the human endometrium. Correlation with the normal and abnormal menstrual cycle. J Clin Invest. 1992;90:188–195.
Achache H., Revel A. Endometrial receptivity markers, the journey to successful embryo implantation. Hum Reprod Update. 2006;12:731–746.
Giudice LC, Irwin JC Roles of the insulinlike growth factor family in nonpregnant human endometrium and at the decidual:trophoblast interface. Semin Reprod Endocrinol. 1999;17:13–21.
Mooney SB, Giudice LC Fetal Endocrinology. Endotext.com; 2002.
Carson DD, Lagow E., Thathiah A., et al. Changes in gene expression during the early to mid-luteal (receptive phase) transition in human endometrium detected by high-density microarray screening. Mol Hum Reprod. 2002;8:871–879.
Horcajadas JA, Riesewijk A., Martín J., et al. Global gene expression profiling of human endometrial receptivity. J Reprod Immunol. 2004;63:41–49.
Kao LC, Tulac S., Lobo S., et al. Global gene profiling in human endometrium during the window of implantation. Endocrinology. 2002;143:2119–2138.
Ponnampalam AP, Weston GC, Trajstman AC, Susil B., Rogers PA Molecular classification of human endometrial cycle stages by transcriptional profiling. Mol Hum Reprod. 2004;10:879–893.
Hess AP, Hamilton AE, Talbi S., et al. Decidual stromal cell response to paracrine signals from the trophoblast: amplification of immune and angiogenic modulators. Biol Reprod. 2007; 76:102–117.
Popovici RM, Betzler NK, Krause MS, et al. Gene expression profiling of human endometrial-trophoblast interaction in a coculture model. Endocrinology. 2006;147:5662–5675.
Sherwin JR, Sharkey AM, Cameo P., et al. Identification of novel genes regulated by chorionic gonadotropin in baboon endometrium during the window of implantation. Endocrinology. 2007;148:618–626.
Chen HW, Chen JJ, Tzeng CR, et al. Global analysis of differentially expressed genes in early gestational decidua and chorionic villi using a 9600 human cDNA microarray. Mol Hum Reprod. 2002;8:475–484.
Talbi S., Hamilton AE, Vo KC, et al. Molecular phenotyping of human endometrium distinguishes menstrual cycle phases and underlying biological processes in normo-ovulatory women. Endocrinology. 2006;147:1097–1121.
Liu P., Hwang JT Quick calculation for sample size while controlling false discovery rate with application to microarray analysis. Bioinformatics. 2007;23:739–746.
Breitling R. Biological microarray interpretation: the rules of engagement. Biochim Biophys Acta. 2006;1759:319–327.
Cui X., Churchill GA Statistical tests for differential expression in cDNA microarray experiments. Genome Biol. 2003;4:210.
Viera AJ, Garrett JM Understanding interobserver agreement: the kappa statistic. Fam Med. 2005;37:360–363.
Soriano D., Hugol D., Quang NT, Darai E. Serum concentrations of interleukin-2R (IL-2R), IL-6, IL-8, and tumor necrosis factor alpha in patients with ectopic pregnancy. Fertil Steril. 2003;79:975–980.
Sallam HN, Sallam A., Ezzeldin F., Agamia AF, Abou-Ali A. Reference values for the midluteal plasma progesterone concentration: evidence from human menopausal gonadotropin-stimulated pregnancy cycles. Fertil Steril. 1999;71:711–714.
Critchley HO, Jones RL, Lea RG, et al. Role of inflammatory mediators in human endometrium during progesterone withdrawal and early pregnancy. J Clin Endocrinol Metab. 1999;84:240–248.
Tierney EP, Tulac S., Huang ST, Giudice LC Activation of the protein kinase A pathway in human endometrial stromal cells reveals sequential categorical gene regulation. Physiol Genomics. 2003;16:47–66.
Burney RO, Talbi S., Hamilton AE, et al. Gene expression analysis of endometrium reveals progesterone resistance and candidate susceptibility genes in women with endometriosis. Endocrinology. 2007;148:3814–3826.
Tabibzadeh S., Kong QF, Babaknia A., May LT Progressive rise in the expression of interleukin-6 in human endometrium during menstrual cycle is initiated during the implantation window. Hum Reprod. 1995;10:2793–2799.
Otani T., Minami S., Kokawa K., Shikone T., Yamoto M., Nakano R. Immunohistochemical localization of activin A in human endometrial tissues during the menstrual cycle and in early pregnancy. Obstet Gynecol. 1998;91(5 Pt 1):685–692.
Jones RL, Salamonsen LA, Zhao YC, Ethier JF, Drummond AE, Findlay JK Expression of activin receptors, follistatin and betaglycan by human endometrial stromal cells; consistent with a role for activins during decidualization. Mol Hum Reprod. 2002;8:363–374.
Grinius L., Kessler C., Schroeder J., Handwerger S. Forkhead transcription factor FOXO1A is critical for induction of human decidualization. J Endocrinol. 2006;189:179–187.
Labied S., Kajihara T., Madureira PA, et al. Progestins regulate the expression and activity of the forkhead transcription factor FOXO1 in differentiating human endometrium. Mol Endocrinol. 2006;20:35–44.
Sasaki A., Shinkawa O., Yoshinaga K. Placental corticotropinreleasing hormone may be a stimulator of maternal pituitary adrenocorticotropic hormone secretion in humans. J Clin Invest. 1989;84:1997–2001.
Rogatsky I., Wang JC, Derynck MK, et al. Target-specific utilization of transcriptional regulatory surfaces by the glucocorticoid receptor. Proc Natl Acad Sci U S A. 2003;100: 13845–13850.
Tulac S., Nayak NR, Kao LC, et al. Identification, characterization, and regulation of the canonical Wnt signaling pathway in human endometrium.J Clin Endocrinol Metab. 2003;88:3860–3866.
Xu C., Mao D., Holers VM, Palanca B., Cheng AM, Molina H. A critical role for murine complement regulator crry in fetomaternal tolerance. Science. 2000;287:498–501.
Young SL, Lessey BA, Fritz MA, et al. In vivo and in vitro evidence suggest that HB-EGF regulates endometrial expression of human decay-accelerating factor. J Clin Endocrinol Metab. 2002;87:1368–1375.
Low JM, Moore TL A role for the complement system in rheumatoid arthritis. Curr Pharm Des. 2005;11:655–670.
Markiewski MM, Lambris JD The role of complement in inflammatory diseases from behind the scenes into the spotlight. Am J Pathol. 2007;171:715–727.
Borregaard N., Theilgaard-Mönch K., Cowland JB, Ståhle M., Sørensen OE Neutrophils and keratinocytes in innate immunity-cooperative actions to provide antimicrobial defense at the right time and place.J Leukoc Biol. 2005;77:439–443.
Bratt T. Lipocalins and cancer. Biochim Biophys Acta. 2000; 1482:318–326.
Sørensen OE, Cowland JB, Theilgaard-Mönch K., Liu L., Ganz T., Borregaard N. Wound healing and expression of antimicrobial peptides/polypeptides in human keratinocytes, a consequence of common growth factors. J Immunol. 2003;170: 5583–5589.
Seth P., Porter D., Lahti-Domenici J., Geng Y., Richardson A., Polyak K. Cellular and molecular targets of estrogen in normal human breast tissue. Cancer Res. 2002;62:4540–4544.
Yao MW, Lim H., Schust DJ, et al. Gene expression profiling reveals progesterone-mediated cell cycle and immunoregulatory roles of Hoxa-10 in the preimplantation uterus. Mol Endocrinol. 2003;17:610–627.
Ziegler S., Röhrs S., Tickenbrock L., et al. Lipocalin 24p3 is regulated by the Wnt pathway independent of regulation by iron. Cancer Genet Cytogenet. 2007;174:16–23.
Berger T., Togawa A., Duncan GS, et al. Lipocalin 2-deficient mice exhibit increased sensitivity to Escherichia coli infection but not to ischemia-reperfusion injury. Proc Natl Acad Sci U S A. 2006;103:1834–1839.
Huang HL, Chu ST, Chen YH Ovarian steroids regulate 24p3 expression in mouse uterus during the natural estrous cycle and the preimplantation period. J Endocrinol. 1999;162:11–19.
King A., Burrows T., Verma S., Hiby S., Loke YW Human uterine lymphocytes. Hum Reprod Update. 1998;4:480–485.
Huddleston H., Schust DJ Immune interactions at the maternal-fetal interface: a focus on antigen presentation. Am J Reprod Immunol. 2004;51:283–289.
Lee N., Llano M., Carretero M., et al. HLA-E is a major ligand for the natural killer inhibitory receptor CD94/NKG2A. Proc Natl Acad Sci U S A. 1998;95:5199–5204.
Le BP, Tabiasco J. Killers become builders during pregnancy. Nat Med. 2006;12:991–992.
Kobayashi Y. The role of chemokines in neutrophil biology. Front Biosci. 2008;13:2400–2407.
Foell D., Wittkowski H., Vogl T., Roth J. S100 proteins expressed in phagocytes: a novel group of damage-associated molecular pattern molecules. J Leukoc Biol. 2007;81:28–37.
Dong JC, Dong H., Campana A., Bischof P. Matrix metalloproteinases and their specific tissue inhibitors in menstruation. Reproduction. 2002;123:621–631.
Zhai Y., Wu R., Schwartz DR, et al. Role of beta-catenin/ T-cell factor-regulated genes in ovarian endometrioid adenocarcinomas. Am J Pathol. 2002;160:1229–1238.
Raouf A., Li V., Kola I., Watson DK, Seth A. The Ets1 proto-oncogene is upregulated by retinoic acid: characterization of a functional retinoic acid response element in the Ets1 promoter. Oncogene. 2000;19:1969–1974.
Takeshita S., Kikuno R., Tezuka K., Amann E. Osteoblast-specific factor 2: cloning of a putative bone adhesion protein with homology with the insect protein fasciclin I. Biochem J. 1993;294(Pt 1):271–278.
Gillan L., Matei D., Fishman DA, Gerbin CS, Karlan BY, Chang DD Periostin secreted by epithelial ovarian carcinoma is a ligand for alpha(V)beta(3) and alpha(V)beta(5) integrins and promotes cell motility. Cancer Res. 2002;62:5358–5364.
Horiuchi K., Amizuka N., Takeshita S., et al. Identification and characterization of a novel protein, periostin, with restricted expression to periosteum and periodontal ligament and increased expression by transforming growth factor beta. J Bone Miner Res. 1999;14:1239–1249.
Sasaki H., Lo KM, Chen LB, et al. Expression of periostin, homologous with an insect cell adhesion molecule, as a prognostic marker in non-small cell lung cancers. Jpn J Cancer Res. 2001;92:869–873.
Litvin J., Chen X., Keleman S., Zhu S., Autieri M. Expression and function of periostin-like factor in vascular smooth muscle cells. Am J Physiol Cell Physiol. 2007;292:C1672–C1680.
Grigoriadis A., Mackay A., Reis-Filho JS, et al. Establishment of the epithelial-specific transcriptome of normal and malignant human breast cells based on MPSS and array expression data. Breast Cancer Res. 2006;8:R56.
Kudo Y., Siriwardena BS, Hatano H., Ogawa I., Takata T. Periostin: novel diagnostic and therapeutic target for cancer. Histol Histopathol. 2007;22:1167–1174.
Oka T., Xu J., Kaiser RA, et al. Genetic manipulation of periostin expression reveals a role in cardiac hypertrophy and ventricular remodeling. Circ Res. 2007;101:313–321.
Yoshiba N., Yoshiba K., Hosoya A., et al. Association of TIMP-2 with extracellular matrix exposed to mechanical stress and its co-distribution with periostin during mouse mandible development. Cell Tissue Res. 2007;330:133:145.
Floridon C., Nielsen O., Hølund B., et al. Does plasminogen activator inhibitor-1 (PAI-1) control trophoblast invasion? A study of fetal and maternal tissue in intrauterine, tubal and molar pregnancies. Placenta. 2000;21:754–762.
Lockwood CJ, Krikun G., Papp C., et al. The role of progestationally regulated stromal cell tissue factor and type-1 plasminogen activator inhibitor (PAI-1) in endometrial hemostasis and menstruation. Ann N Y Acad Sci. 1994;734:57–79.
Dossenbach-Glaninger A., van Trotsenburg M., Dossenbach M., et al. Plasminogen activator inhibitor 1 4G/5G polymorphism and coagulation factor XIII Val34Leu polymorphism: impaired fibrinolysis and early pregnancy loss. Clin Chem. 2003;49:1081–1086.
Smith SK Regulation of angiogenesis in the endometrium. Trends Endocrinol Metab. 2001;12:147–151.
Strieter RM, Kunkel SL, Elner VM, et al. Interleukin-8. A corneal factor that induces neovascularization. Am J Pathol. 1992;141:1279–1284.
Iozzo RV The family of the small leucine-rich proteoglycans: key regulators of matrix assembly and cellular growth. Crit Rev Biochem Mol Biol. 1997;32:141–174.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the NICHD/NIH through cooperative agreement 1U54HD055764-01 (LCG) and U54-HD35041 (BAL) as part of the Specialized Cooperative Centers Program in Reproduction and Infertility Research (LCG) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) BEX3656-06-3 (RFS). Samples were collected at Hospital de Clínicas de Porto Alegre. Data analysis and RT-PCR were performed at University of California, San Francisco. Immunohistochemistry was performed at Greenville Hospital System.
Rights and permissions
About this article
Cite this article
Savaris, R.F., Hamilton, A.E., Lessey, B.A. et al. Endometrial Gene Expression in Early Pregnancy: Lessons From Human Ectopic Pregnancy. Reprod. Sci. 15, 797–816 (2008). https://doi.org/10.1177/1933719108317585
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719108317585