Abstract
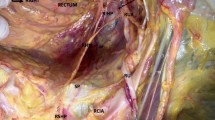
The authors previously demonstrated nerve trunks and autonomic ganglia of the hypogastric plexus within the uterosacral ligament (USL) and the cardinal ligaments. The nerve content of these ligaments is greatest closer to the pelvic sidewalls and diminishes toward the insertion of the ligaments into the uterus, with the greater nerve content in the USL. Here the authors determine whether the nerve content of the superficial and deep portion of the USLs, where they are divided at a radical hysterectomy, differ. Biopsies were taken from the right and left superficial and deep USL in 6 patients during radical hysterectomy for early-stage cervical cancer. Indirect immunofluorescence was performed using primary antibodies to (1) the panneuronal marker PGP 9.5, (2) the parasympathetic marker vasoactive intestinal peptide, (3) the sympathetic markers tyrosine hydroxylase and neuropeptide-Y, (4) the sensory and nociceptive nerve marker substance P, and (5) the sensory and sensory-motor nerve marker calcitonin gene-related peptide. The percentage area of immunoreactivity (PAI) was determined using a computer-assisted image analyzer as an objective measure of nerve content. There was a lower nerve content in the superficial USL compared with the deep USL. The PAI of the deep USL was greater than that of the superficial USL for all the nerve markers (P < .05). The PAI was greatest for sympathetic and sensory/nociceptive nerve markers. There were relatively more sympathetic nerve fibers than parasympathetic nerve fibers in the deep USL. These data provide further indirect evidence that pelvic dysfunction following radical hysterectomy is associated with division of the deep portion of the USL.
Similar content being viewed by others
References
Butler-Manuel SA, Summerville K., Ford AM, et al. Self-assessment of morbidity following radical hysterectomy for cervical cancer. J Obstet Gynaecol. 1999;19:180–183.
Pieterse QD, Maas CP, ter Kuile MM, et al. An observational longitudinal study to evaluate micturition, defaecation, and sexual function after radical with pelvic lymphadenectomy for early stage cancer. Int J Gynecol Cancer. 2006;16:1119–1129.
Last R. Anatomy, Regional and Applied. 7th ed. Edinburgh, UK: Churchill Livingstone; 1984.
Mundy AR An anatomical explanation for bladder dysfunction following rectal and uterine surgery. Br J Urol. 1982;54: 501–504.
Smith PH, Ballantyne B. The neuroanatomical basis of the urinary bladder dysfunction following major pelvic surgery. Br J Surg. 1968;55:929–933.
Maas CP, DeRuiter MC, Kenter GG, Trimbos JB The inferior hypogastric plexus in gynecologic surgery. J Gynecol Technol. 1999;5:55–62.
Butler-Manuel SA, Buttery LDK, Polak JM, A’Hern R, Barton DPJ. Pelvic nerve plexus trauma at radical hysterectomy and simple hysterectomy: the nerve content of the uterine supporting ligaments. Cancer. 2000;89:834–841.
Butler-Manuel SA, Buttery LDK, Polak JM, A’Hern R, Barton DPJ. Pelvic nerve plexus trauma at radical hysterectomy and simple hysterectomy: a quantitative study of nerve types within the uterosacral and cardinal ligaments. J Soc Gynecol Invest. 2002;9:47–56.
Zamboni L., De Martino C. Buffered picric acid formaldehyde: a new rapid fixative for electron microscopy. J Cell Biol. 1967;35:148A.
Marron K., Yacoub MH, Polak JM, et al. Innervation of human atrioventricular and arterial valves. Circulation. 1996; 94:368–375.
Takahashi T., Ueno M., Azekura K., Ohta H. Lateral ligament: its anatomy and clinical importance. Semin Surg Oncol. 2000;19:386–395.
Kato T., Murakami G., Yabuki Y. Does the cardinal ligament of the uterus contain a nerve that should be preserved in radical hysterectomy? Anat Sci Int. 2002;77:161–168.
Tamakawa M., Murakami G., Kato T., Hareyama M. Fascial structures and autonomic nerves in the female pelvis: a study using macroscopic slices and their corresponding histology. Anat Sci Int. 2003;78:228–242.
Baader B., Herrmann M. Topography of the pelvic autonomic nervous system and its potential impact on surgical intervention in the pelvis. Clin Anat. 2003;16:119–130.
Yabuki Y., Asamoto A., Hoshiba T., Nishimoto H., Nishiwara Y., Nakajima T. Radical hysterectomy: an anatomic evaluation of parametrial dissection. Gynecol Oncol. 2000;77:155–163.
Scambia G., Ferrandina G., Distefano M., et al. Is there a place for a less extensive radical surgery in locally advanced cervical cancer patients? Gynecol Oncol. 2001;83:319–324.
Winter R., Haas J., Reich O., et al. Parametrial spread of cervical cancer in patients with negative pelvic lymph nodes. Gynecol Oncol. 2002;84:252–257.
Benedetti-Panici P., Maneschi F., D’Andrea G., et al. Early cervical cancer: the natural history of lymph node involvement redefined on the basis of the thorough parametrectomy and giant section study. Cancer. 2000;88:2267–2274.
Piver MS, Rutledge F., Smith JP Five classes of extended hysterectomy for women with cervical cancer. Obstet Gynecol. 1974;44:265–272.
Höckel M., Konerding MA, Heussel CP Liposuction-assisted nerve-sparing extended radical hysterectomy: oncological rationale, surgical anatomy and feasibility study. Am J Obstet Gynecol. 1998;178:971–976.
Trimbos JB, Maas CP, Deruiter MC, Peters AAW, Kenter GG A nerve-sparing radical hysterectomy: guidelines and feasibility in Western patients. Int J Gynecol Cancer. 2001;11:180–186.
Kato T., Murakami G., Yabuki Y. A new perspective on nerve-sparing radical hysterectomy: nerve topography and over preservation of the cardinal ligament. Jpn J Clin Oncol. 2003;33:589–591.
Ito E., Saito T. Nerve-sparing techniques for radical hysterectomy. Eur J Surg Oncol. 2004;30:1137–1140.
Sakuragi N., Todo Y., Kudo M., Yamamoto R., Sato T. A systematic nerve-sparing radical hysterectomy technique in invasive cervical cancer for preserving postsurgical bladder function. Int J Gynecol Cancer. 2005;15:389–397.
Possover M., Stober S., Plaul K., Schneider A. Identification and preservation of the motoric innervation of the bladder in radical hysterectomy type III. Gynecol Oncol. 2000;79:154–157.
Ercoli A., Delmas V., Gadonneix P., et al. Classical and nerve-sparing radical hysterectomy: an evaluation of the risk of injury to the autonomous pelvic nerves. Surg Radiol Anat. 2003;25:200–206.
Katahira A., Niikura H., Kaiho Y., et al. Intraoperative electrical stimulation of the pelvic splanchnic nerves during nerve-sparing radical hysterectomy. Gynecol Oncol. 2005;98:462–466.
Katahira A., Niikura H., Ito K., et al. Vesicouterine ligament contains abundant autonomic nerve ganglion dells: the distribution in histology concerning nerve-sparing radical hysterectomy. Int J Gynecol Cancer. In press.
Sato K., Sato K. The vascular and neuronal composition of the lateral ligament of the rectum and the rectosacral fascia. Surg Radiol Anat. 1991;13:17–22.
Mauroy B., Demondion X., Bizet B., Claret A., Mestdagh P., Hurt C. The female inferior hypogastric plexus: anatomical and radiological description of the plexus and its afferences—applications to pelvic surgery. Surg Radiol Anat. 2007;29:55–66.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Butler-Manuel, S.A., Buttery, L.D.K., Polak, J.M. et al. Autonomic Nerve Trauma at Radical Hysterectomy: The Nerve Content and Subtypes Within the Superficial and Deep Uterosacral Ligaments. Reprod. Sci. 15, 91–96 (2008). https://doi.org/10.1177/1933719107309648
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719107309648