Abstract
Objective
CD44 is a cell surface glycoprotein widely distributed in the extracellular matrix. CD44 isoforms arising from alternative mRNA splicing are implicated in tumor metastases. The aim of this study is to investigate the expression of CD44s and two splice variants, CD44-9v and CD44-10v, in squamous cell carcinoma (SCC) of the vulva as well as its correlation with lymph node (LN) metastases and disease-free survival.
Methods
Thirty-five SCC vulvar tumors were evaluated for CD44s, CD44-9v and CD44-10v expression by immunoctyochemistry. One nonmetastatic LN was studied also. In cases with LN metastases, the metastatic LN as well as a nonmetastatic LN from the same patient were evaluated.
Results
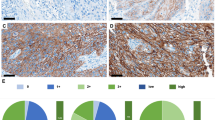
CD44s and CD449v were expressed in all epithelia—normal, dysplastic, and SCC. However, intensity and distribution of expression of 9v isoforms changed within the tissue containing invasive cancer. CD44-9v expression was downregulated in the most differentiated cells within the carcinoma, mainly in patients who had disease recurrence or eventually died of disease (P = .031). All metastatic tumor to LNs was immunoreactive also for CD44-9v. CD44-10v expression was present in 78% of tumors and 56% of normal epithelium. Interestingly, CD44-10v membrane expression, but not cytoplasmic expression, correlated with disease recurrence (P = .035).
Conclusion
Our findings warrant larger multi-institutional studies to determine the potential of CD44-9v and CD44-10v as molecular markers of disease recurrence in vulvar carcinoma. We propose to test the use of anti-CD44-9v monoclonal antibody for radioimmunoimaging of a occult LN metastases.
Similar content being viewed by others
References
Bracke ME, Charlier C, Bruyneel EA. Tamoxifen restores the E-cadherin function in human breast cancer MCF-7/6 cells and suppresses their invasive phenotype. Cancer Res 1994;54:4607.
Liotta LA, Steeg PS, Stetler-Stevenson WG. Cancer metastasis and angiogenesis: An imbalance of positive and negative regulation. Cell 1991;64:327.
Underhill CB, Chi-Rosso G, Toole BP. Effects of detergent solubilization on the hyaluronate-binding protein from membranes of simian virus 40-transformed 3T3 cells. J Biol Chem 1983;258:8086–91.
Naor D, Sionov RV, Ish-Shalom D. CD44: Structure, function and association with the malignant process. In: Vande Woude GF, Klein G, eds. Advances in cancer research. San Diego: Academic Press, 1997;71:241–318.
Soukka T, Salmi M, Joensuu H, et al. Regulation of CD44v6-containing isoforms during proliferation of normal and malignant epithelial cells. Cancer Res 1997;57:2281–9.
Teder P, Bergh J, Heldin P. Functional hyaluronan receptors are expressed on a squamous cell lung carcinoma cell line but not other lung carcinoma cell lines. Cancer Res 1995;55:3908–14.
Friedrichs K, Folker F, Björn-Wieland L, et al. CD44 isoforms correlate with cellular differentiation but not with prognosis in human breast cancer. Cancer Res 1995;55:5424–33.
Rodriguez C, Monges G, Rouanet P, et al. CD44 expression patterns in breast and colon tumors: A PCR-based study of splice variants. Int J Cancer 1995;64:347–54.
Li H, Hamou MF, de Tribolet N, et al. Variant CD44 adhesion molecules are expressed in human brain metastases but not in glioblastomas. Cancer Res 1993;53:5345–9.
Tempfer C, Gitsch G, Haeusler G, et al. Prognostic value of immunohistochemically detected CD44 expression in patients with carcinoma of the vulva. Cancer 1996;78:273–7.
Tempfer C, Gitsch G, Hanzal E, et al. Expression of the adhesion molecule CD44v3 is a prognostic factor in vulvar carcinoma. Anticancer Res 1996;16:2029–32.
Ambros RA, Kallakury BV, Malfetano JH, et al. Cytokine, cell adhesion receptor, and tumor suppressor gene expression in vulvar squamous carcinoma: Correlation with prominent fibromyxoid stromal response. Int J Gynecol Pathol 1996;15:320–5.
Rodríguez-Rodríguez L, Sancho-Torres I, Mesonero C, et al. CD44 splice variant expression in clear cell carcinoma of the ovary. J Gynecol Oncol 1998;71:223–9.
Sturgeon SR, Brinton LA, Devesa SS, et al. In situ and invasive vulvar cancer incidence trends (1973 to 1987). Am J Obstet Gynecol 1992;166:1482.
Dall P, Heider KH, Hekele A, et al. Surface protein expression and messenger RNA-splicing analysis of CD44 in uterine cervical cancer and normal cervical epithelium. Cancer Res 1994;54:3337–41.
Kainz C, Kohlberger P, Sliutz G, et al. Splice variants of CD44 in human cervical cancer stage IB to IIB. Gvnecol Oncol 1995;57:383–7.
Delias A, Schultheiss E, Almendral AC, et al. Expression of CD44 and variant isoforms in cervical intraepithelial neoplasia. Gynecol Oncol 1996;62:218–25.
Lagorce-Pages C, Paraf F, Dubois J, et al. Expression of CD44 in premalignant and malignant barrett’s oesophagus. Histopathology 1998;32:7–14.
Ingle R, Jennings T, Goodman M, et al. CD44 expression in sinonalsal inverted papillomas and associated squamous cell carcinoma. ACJP 1998;109:309–14.
Steelentag W, Gunthert U, Saremaslani P, et al. CD44 standard and variant isaform expression in human epidermal skin tumors is not correlated with tumor aggressiveness but down-regulated during proliferation and tumor de-differentiation. Int J Cancer 1996;69:218–24.
Seiter S, Tilgen W, Herrmann K, et al. Expression of CD44 splice variants in human skin and epidermal tumours. Virchows Arch 1996;428:141–9.
Author information
Authors and Affiliations
Additional information
The research was supported by the Mae Stone Goode Trust Foundation and National Cancer Institute grant no. POCA11198G.
The authors thank Ms. Diana Scott and Mr. Albert Paglialunga for their technical support, and Ms. Carine Polliotti for her excellent editorial assistance.
Rights and permissions
About this article
Cite this article
Rodríguez-Rodríguez, L., Sancho-Torres, I., Gibbon, D.G. et al. CD44-9v and CD44-10v Are Potential Molecular Markers for Squamous Cell Carcinoma of the Vulva. Reprod. Sci. 7, 70–75 (2000). https://doi.org/10.1177/107155760000700111
Published:
Issue Date:
DOI: https://doi.org/10.1177/107155760000700111