Abstract
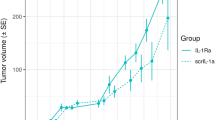
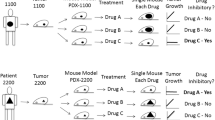
Tumor growth inhibition data in in vivo anticancer experiments are commonly analyzed using the treatment-to-control ratio (TCR). Parametric and nonparametric confidence interval approaches for this ratio are introduced, enabling a quantitative statistical decision. The growth curves are characterized by the area-under-the-curve technique, adjusted for animal-specific survival. Simple simultaneous approaches are proposed for complex designs, including several treatment or dose groups. This implementation makes decision making easier for the pharmacologists through the use of simple diagrams for the treatment-to-control ratios and their confidence intervals. Tumor inhibition and regression can be appropriately statistically analyzed by treatment-to-control ratios and their confidence intervals.
Similar content being viewed by others
References
Shusterman S, Grupp SA, Barr R, Carpentieri D, Zhao H, Maris JM. The angiogenesis inhibitor TNP-470 effectively inhibits human neuroblastoma xenograft growth, especially in the setting of subclinical disease. Clin Cancer Res. 2001;7:977–984.
Freedman LS, Midthune DN, Brown CC, Steele V, Kelloff GJ. Statistical analysis of animal cancer chemoprevention experiments. Biometrics. 1993;49:259–268.
Kokoska SM, Hardin JM, Grubbs CJ, Hsu C. The statistical analysis of cancer inhibition/promotion experiments. Anticancer Res. 1993;13:1357–1363.
Dunson DB, Haseman JK. Modeling tumor onset and multiplicity using transition models with latent variables. Biometrics. 1999;55:965–70.
Koziol JA, Maxwell DA. A distribution-free test for paired growth curve analyses with application to an animal tumour immunotherapy experiment. Stat. Med. 1992;1:83–89.
Hanfelt JJ. Statistical approaches to experimental design and data analysis of in vivo studies. Breast Cancer Res Treat. 1997; 46:279–302.
Liang H, Sha NJ. Modeling antitumor activity by using a non-linear mixed-effects model. Math Biosci. 2004;89:61–73.
Tan M, Fang HB, Tian GL, Houghton PJ. Small-sample inference for incomplete longitudinal data with truncation and censoring in tumor xenograft models. Biometrics. 2002;58:612–620.
Tan T, Fang HB, Tian GL Repeated-measures models with constrained parameters for incomplete data in tumour xenograft experiments. Stat. Med. 2005;24:109–119.
Pham B, Cranney A, Boers M, Verhoeven AC, Wells G, Tugwell P. Validity of area-under-the-curve analysis to summarize effect in rheumatoid arthritis clinical trials. J Rheumatol. 1999;26:712–716.
Frison L. Pocock SJ. Repeated measures in clinical trials: analysis using mean summary statistics and its implications for design. Stat Med. 1992;11:1685–1704.
Qian W, Parmar MKB. Analysis of messy longitudinal data from a randomized clinical trial. Stat Med. 2000;19:2657–2674.
Matthews JN, Campbell MJ. Adjusting for baseline: change or percentage change. Stat Med. 1992;11:1624–1626.
Vardi Y, Ying Z, Zhang G. Two-sample tests for growth curves under dependent right censoring. Biometrika. 2001;80:949–960.
Heitjan DF, Kunselman S. Time until first significant difference in in vivo tumor growth experiments. In Vivo. 1995:9:7–9.
Re RN, Cook JL, Giardina JF. The inhibition of tumor growth by triplex-forming oligonuceotides. Cancer Lett. 2004;209:51–53.
Harrison G, Richards S, Lawson S. et al. Comparison of allogeneic transplant versus chemotherapy for relapsed childhood acute lymphoblastic leukaemia in the MRC UKALL R1 trial. Ann Oncol. 2000;11:999–1006.
Roehmel J. Therapeutic equivalence investigations: statistical considerations. Stat Med. 1998;17:1703–1714.
Feuerstein TJ, Rossner R, Schumacher M. How to express an effect mean as percentage of a control mean? J Pharmacol Toxicol Methods. 1997;37:187–190.
Liu JP, Weng CS. Evaluation of log-transformation in assessing bioequivalence. Commun Stat. 1994;A23:421–434.
Fieller E. Some problems in interval estimation. J R Stat Soc. 1954;B16:175–185.
Corbett Th. Valeriote F. Tumor models and the discovery and secondary evaluation of solid tumor active agents. Int J Pharmacognosy. 1995;33:102–122.
Hauschke D, Kieser M, Hothorn LA. Proof of safety in toxicology based on the ratio of two means for normally distributed data. Biometr J. 1999;41:295–304.
Iversen PW. Comparison of in vivo tumor growth models. Proc Annu Meeting Am Stat Assoc. 2001:5–9.
Hothorn T, Munzel U. Non-parametric confidence interval for the ratio. Report University of Erlangen. Department Medical Statistics. 2002. Available at: www.uni-erlangen.de.
Hodges JL, Lehmann EL. Estimates of location based on rank tests. Ann Math Stat 1963;34:598–611.
Dilba G, Bretz F, Guiard V, Hothorn LA. Simultaneous confidence intervals for ratios for the comparison of several treatments with a control. Meth Inf Med. 2004;43:465–469.
Bretz F, Hothorn LA, Hsu J. Identifying effective and/or safe doses by stepwise confidence intervals for ratios. Stat Med. 2003;22:847–858.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hothorn, L.A. Statistical Analysis of in Vivo Anticancer Experiments: Tumor Growth Inhibition. Ther Innov Regul Sci 40, 229–238 (2006). https://doi.org/10.1177/009286150604000212
Published:
Issue Date:
DOI: https://doi.org/10.1177/009286150604000212