Abstract
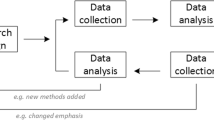
This paper describes a method for designing a clinical trial to combine aspects of Phases 2 and 3 of the clinical development program. Specifically, a group sequential design is presented, which incorporates treatment selection based upon a short-term “provisional” endpoint, as is often undertaken in Phase 2 trials, followed by a comparison of the selected treatment with control in terms of a longer-term primary endpoint. An example is given illustrating the methodology and we discuss how this approach may reduce the total number of patients required in the evaluation process without compromising its integrity, leading to more ethical and efficient clinical studies.
Similar content being viewed by others
References
Stallard N, Todd S. Sequential designs for phase III clinical trials incorporating treatment selection. Stat Med. 2003;22:689–703.
Paulson E. A selection procedure for selecting the population with the largest mean from k normal populations. Ann Math Stat. 1964;35:174–180.
Bechhofer RE, Kiefer J, Sobel M. Sequential Identification and Ranking Problems. Chicago: University of Chicago Press; 1968.
Thall PF, Simon R, Ellenberg SS. Two-stage selection and testing designs for comparative clinical trials. Biometrika. 1988;75:303–310.
Thall PF, Simon R, Ellenberg SS. A two-stage design for choosing among several experimental treatments and a control in clinical trials. Biometrics. 1989;45:537–547.
Schaid DJ, Wieand S, Therneau TM. Optimal two-stage screening designs for survival comparisons. Biometrika. 1990:77:659–663.
Follman DA, Proschan MA, Geller NL Monitoring pairwise comparisons in multi-armed clinical trials. Biometrics. 1994;50:325–336.
Inoue LYT, Thall PF, Berry DA. Seamlessly expanding a raodomised phase II trial to phase III. Biometrics. 2002;58:823–831.
Berry DA, Müller P, Grieve AP, Smith M, Parke T. Blazek R, Mitchard N, Krams M. Adaptive Bayesian Designs for Dose-Ranging Drug Trials. In Case Studies in Bayesian Statistics, Lecture Notes in Statistics. New York: Springer; 2002;162, 99–181.
Lin DY, Shen L, Ying Z, Breslow NE. Group sequential designs for monitoring survival probabilities. Biometrics. 1996;52:1033–1041.
Whitehead J. The Design and Analysis of Sequential Clinical Trials. Revised Second Edition. Chichester. England: Wiley; 1997.
Lan KKG, DeMets DL. Discrete sequential boundaries for clinical trials. Biometrika. 1983; 70:659–663.
Kim K, DeMets DL. Design and analysis of group sequential tests based on the type I error spending rate function. Biometrika. 1987;74:149–154.
Stallard N, Facey KM. Comparison of the spending function method and the Christmas tree correction for group sequential tests. J Biopharma Stat. 1996;6:361–373.
Pampallona S, Tsiatis AA, Kim K. Spending functions for the type I and type II error probabilities of group sequential tests. Technical Report, Department of Biostatistics. Boston. MA: Harvard School of Public Health; 1995.
Scharfstein DO, Tsiatis AA, Robins JM. Semiparametric efficiency and its implication on the design and analysis of group-sequential studies. J Am Stat Assoc. 1997;92:1342–1350.
Todd S. A flexible information-based approach to the design and interim monitoring of bivariate group sequential clinical trials. J Biopharma Stat. 2003;13:605–619.
Whitehead J. Sample sizes for phase II and phase III clinical trials: an integrated approach. Stat Med. 1986;5:459–464.
Shen L. An improved method of evaluating drug effect in a multiple dose clinical trial. Stat Med. 2001;20:1913–1929.
Stallard N, Todd S. Point estimates and confidence regions for sequential trials involving selection. J Stat Plan Inference. 2005; in press.
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented at the DIA 39th Annual Meeting, June 15–19, 2003, San Antonio, Texas.
Rights and permissions
About this article
Cite this article
Todd, S., Stallard, N. A New Clinical Trial Design Combining Phases 2 and 3: Sequential Designs with Treatment Selection and a Change of Endpoint. Ther Innov Regul Sci 39, 109–118 (2005). https://doi.org/10.1177/009286150503900201
Published:
Issue Date:
DOI: https://doi.org/10.1177/009286150503900201