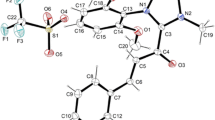
Abstract
The reactions of the dithio substituted carbenium ions 2a-c with allylsilanes, allylstannanes (3), and silylated enol ethers (4) which yield dithioacetal protected β, ψ-unsaturated carbonyl compounds (7) and selectively protected 1,3-dicarbonyl compounds (8), have been studied kinetically. The second-order rate constants have been used to determine the electrophilicity parameters for the [1,3]dithiolan-2-ylium ion 2a (E=−6.25) and the [1,3]dithian-2-ylium ions 2b (E=−6.82) and 2c (E=−2.17). It is shown how these parameters can be used to predict the electrophilic potential of 2a-c.
Similar content being viewed by others
References
G.A. Olah, Angew. Chem., 85, 183 (1973); Angew. Chem. Int. Ed. Engl., 12, 173 (1973); b) G. A. Olah, G.K.S. Prakash, and J. Sommer, Superacids, John Wiley & Sons, 1985, p. 113.
This has been employed for the [1,3]dithian-2-ylium ion (2c generated from 2-methoxy-[1,3]dithiane and TiCl4) and indole by P. Stütz and P. A. Stadler, Helv. Chim. Acta 55, 75, (1972).
H. Mayr and M. Patz, Angew. Chem., 106, 990 (1994); Angew. Chem. Int. Ed. Engl., 33, 938 (1994).
Y. Yamamoto and N. Asao, Chem. Rev., 93, 2207 (1993); b) I. Fleming, J. Dunogues, and R. Smithers, Org. React., 37, 57 (1989); c) W.P. Weber. Silicon Reagents for Organic Synthesis, Springer. Berlin, 1983; d) I. Fleming in Comprehensive Organic Chemistry, D.H.R. Barton and W.D. Ollis, Eds., Pergamon Press, Oxford, 1979, Vol. 3, p. 541; e) G. Hagen and H. Mayr, J. Am. Chem. Soc., 113, 4954 (1991).
M. Pereyre, J.P. Quintard, and A. Rahm, Tin in Organic Synthesis, Butterworths, London, 1987.
I. Paterson and L.G. Price, Tetrahedron Lett., 22, 2829 (1981).
H. Mayr, R. Schneider, C. Schade, J. Bartl, and R. Bederke, J. Am. Chem. Soc., 112, 4446 (1990).
H. Mayr, Angew. Chem., 102, 1415 (1990); Angew. Chem. Int. Ed. Engl., 29, 1371 (1990).
H. Mayr, N. Basso, and G. Hagen, J. Am. Chem. Soc., 114, 3060 (1992).
H. Mayr, G. Lang unpublished results.
I. Stahl, Chem. Ber., 118, 4857 (1985).
T. Okuyama, N. Haga, S-y. Takane, K. Ueno and T. Fueno, Bull. Chem. Soc. Jpn., 64, 2751 (1991).
H. Mayr, M. F. Gotta unpublished results.
I. Stahl, Chem. Ber., 118, 1798 (1985).
K. Hatanaka, S. Tanimoto, T. Sugimoto, and M. Okano, Tetrahedron Lett., 22, 3243 (1981); b) M. Kodpinid, T. Siwapinyoyos, and Y. Thebtaranonth, J. Am. Chem. Soc., 106, 4862 (1984); c) I. Paterson and L.G. Price, Tetrahedron Lett., 22, 2833 (1981).
I. Stahl, Chem. Ber., 118, 3159 (1985).
C. Westerlund, Tetrahedron Lett., 23, 4835 (1982).
E.A. Hill, M.L. Gross, M. Stasiewicz, and M. Manion, J. Am. Chem. Soc., 91, 7381 (1969).
E.Akgün, M. Tunali, and U. Pindur, Liebigs Ann. Chem., 1628 (1986); b) P. Stütz and P.A. Stadler, Org. Synth., Coll. Vol. 6, 109 (1988).
see ref. [2]— Helv. Chim. Acta 55, 75, (1972).
R. Taylor, Electrophilic Aromatic Substitution, John Wiley & Sons, Chichester, 1990, p. 458.
I. Stahl, Chem. Ber., 120, 135 (1987).
D.D. Perrin, W.L.F. Armarego, and D.R. Perrin, Purification of Labatory Chemicals, 2nd ed., Pergamon Press, Oxford, 1980.
H.O. House, L.J. Czuba, M. Gall, and H.D. Olmstead, J. Org. Chem., 34, 2324 (1969).
J. Klaveness and K. Undheim, Acta Chem. Scand. Ser. B, 37, 687 (1983); b) J. Klaveness, F. Rise, and K. Undheim, Acta Chem. Scand. Ser. B, 40, 398 (1986).
A. Padwa, M. Akiba, L.A. Cohen, and J.G. MacDonald, J. Org. Chem., 48, 695, (1983).
W.A. Donaldson and M.A. Hossain, Tetrahedron Lett., 33, 4107, (1992).
G. Picotin and P. Migniac, J. Org. Chem., 50, 1299, (1985).
K. Narasaka, N. Arai, and T. Okauchi, Bull. Chem. Soc. Jpn., 66, 2995 (1993).
R.C. Chambers and C.L. Hill, J. Am. Chem. Soc., 112, 8427 (1990).
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. George A. Olah who imbued carbocations with immortality.
Rights and permissions
About this article
Cite this article
Mayr, H., Henninger, J. & Siegmund, T. Quantification of the electrophilicities of dithiocarbenium ions. Res. Chem. Intermed. 22, 821–838 (1996). https://doi.org/10.1163/156856796X00502
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1163/156856796X00502